Hereditary renal amyloidosis is an autosomal dominant condition with considerable overlap with other amyloidosis types. Differential diagnosis is complicated, but is relevant for prognosis and treatment. We describe a patient with nephrotic syndrome and progressive renal failure, who had a mother with renal amyloidosis.
Renal biopsy revealed amyloid deposits in glomerular space, with absence of light chains and protein AA. We suspected amyloidosis with fibrinogen A alpha chain deposits, which is the most frequent cause of hereditary amyloidosis in Europe, with a glomerular preferential affectation. However, the genetic study showed a novel mutation in apolipoprotein AI. On reviewing the biopsy of the patient's mother similar glomerular deposits were found, but there were significant deposits in the renal medulla as well, which is typical in APO AI amyloidosis. The diagnosis was confirmed by immunohistochemistry.
Apo AI amyloidosis is characterised by slowly progressive renal disease and end-stage renal disease occurs approximately 3–15 years from initial diagnosis. Renal transplantation offers an acceptable graft survival and in these patients with hepatorenal involvement simultaneous liver and kidney transplantation could be considered.
La amiloidoisis renal hereditaria es un trastorno autosómico dominante cuya clínica se solapa con la de otros tipos de amiloidoisis. Hacer un adecuado diagnóstico diferencial puede ser difícil, pero tiene una gran relevancia respecto al pronóstico y tratamiento, que difiere según sea el origen de la enfermedad. Presentamos el caso clínico de un paciente con síndrome nefrótico e insuficiencia renal progresiva, con antecedente familiar de madre con amiloidosis renal.
En la biopsia renal se observó depósito de amiloide a nivel glomerular, con negatividad para cadenas ligeras y proteína AA. La sospecha clínica inicial fue la de amiloidosis por depósito de cadena A alfa de fibrinógeno, que es la causa más frecuente de amiloidosis hereditaria en Europa, con afectación preferentemente glomerular. Sin embargo, el estudio genético determinó una nueva mutación previamente no descrita de la Apolipoproteina AI (APO AI). En la biopsia de la madre se detectó depósito glomerular, pero también depósito masivo en médula, lo que caracteriza a la amiloidosis por depósito de APO AI. El diagnóstico se confirmó mediante inmunohistoquímica.
La amiloidosis por depósito de Apo AI progresa a enfermedad renal crónica terminal en el plazo de de 3 a 15 años. Se diferencia clínicamente de la amiloidosis AL por su menor afectación extrarrenal y su mejor pronóstico. El trasplante renal ofrece una supervivencia del injerto aceptable y el trasplante hepato-renal se podría tener en cuenta en pacientes con disfunción significativa de ambos órganos.
A 40-year-old male came to the emergency room complaining of oedemas. Patient presented family history of mother with non-affiliated renal amyloidosis. The mother's renal biopsy showed permanganate resistance, which seemed to rule out AA amyloidosis, and led to consider the possibility of AL amyloidosis.
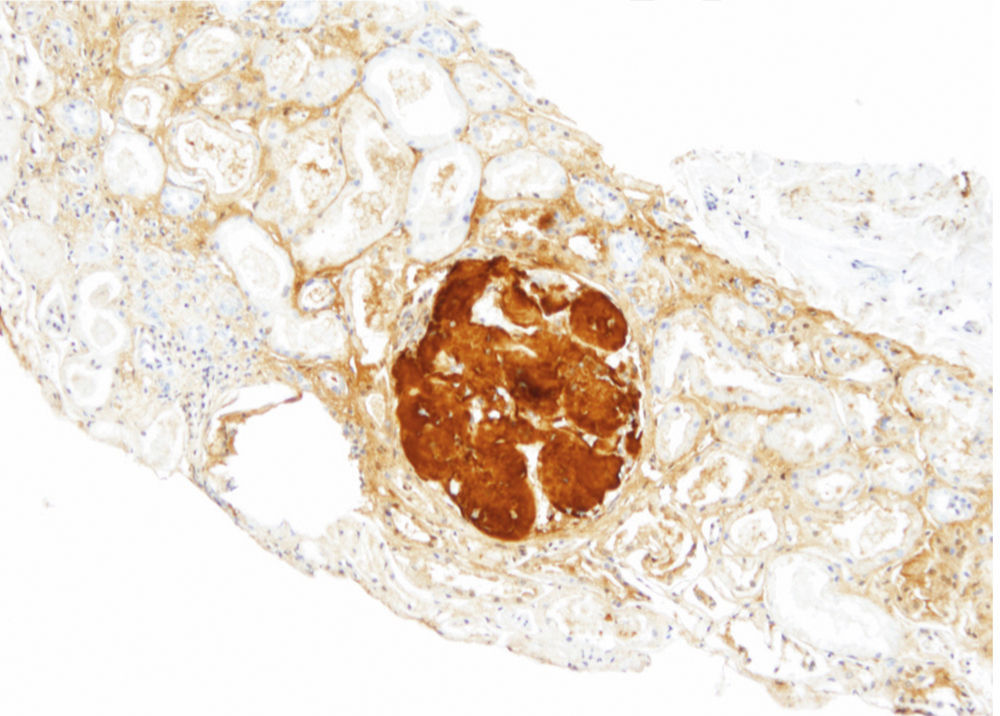
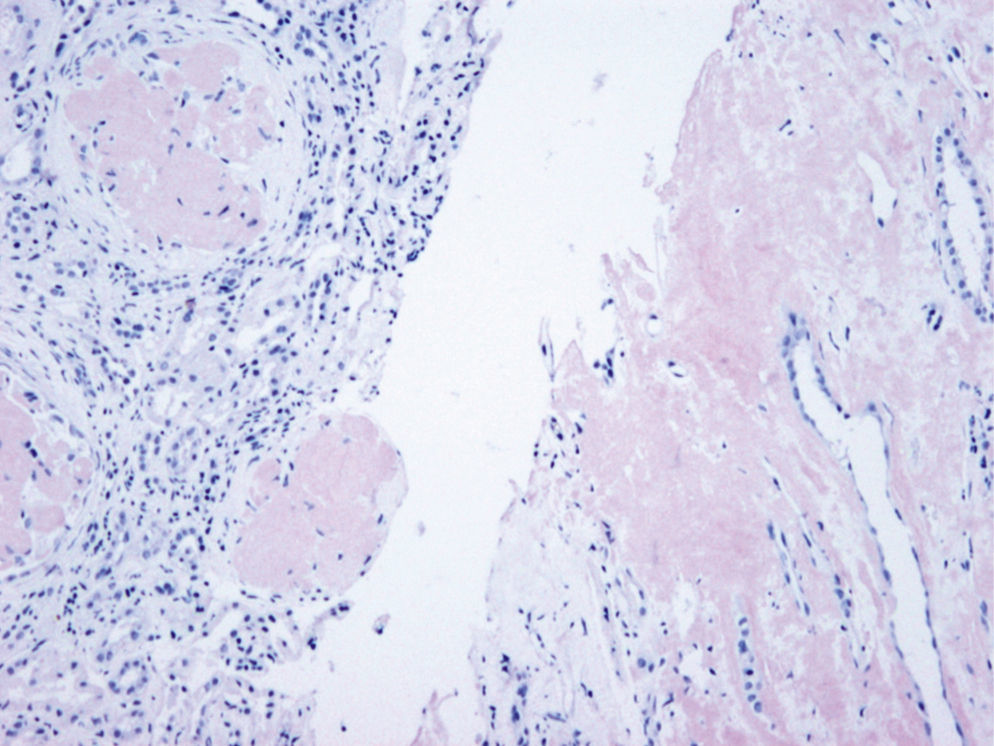
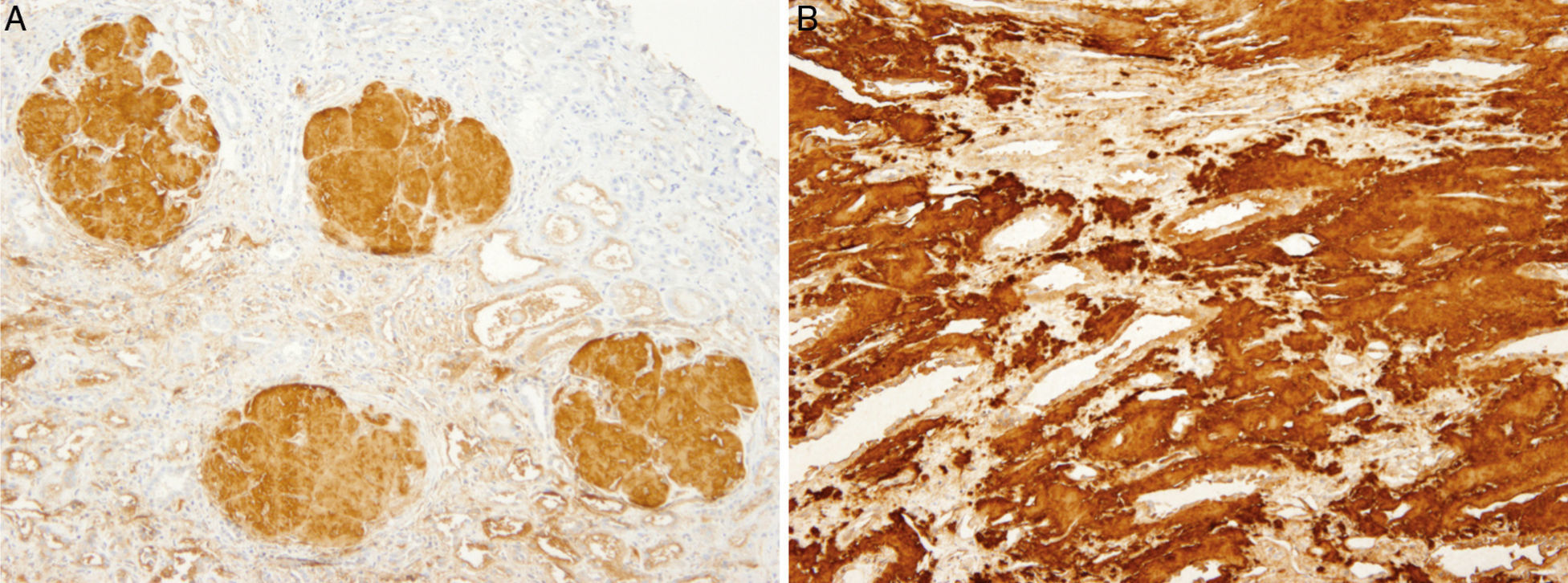
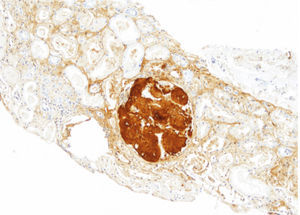
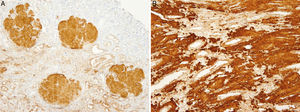
The patient did not receive any pharmacological treatment. Upon physical examination, the patient presented blood pressure of 135/75mmHg and bimalleolar oedemas, without other data of interest. Laboratory tests confirmed the presence of nephrotic syndrome (serum albumin of 2.5g/dl, cholesterolaemia of 275mg/dl and urine protein of 6g/24h), without alterations in the sediment and with normal glomerular filtration rate. Electrophoretic spectrum in blood and urine, immunological study and renal ultrasound scan were normal. A renal biopsy was performed that presented 19 glomeruli with fully distorted structure by a hyaline deposit in nodular disposition, with positivity for Congo red and thioflavin. Tubules and vessels did not show significant alterations and therefore the findings were compatible with amyloidosis. The study of a paraffin sample showed a marked positivity for AP protein, which is a part of the amyloid substance common in all amyloidosis types, with negativity for light chains and AA protein, B2 microglobulin and transthyretin. A supplementary study was performed with antibodies to fibrinogen and lysozyme and the amyloid substance deposits resulted positive for the fibrinogen A-alpha chain (AFib). However, the genetic study did not show any mutation in the AFib chain gene but there was a previously undescribed mutation in the Apolipoprotein A1 gene (Apo AI). Faced with this discrepancy, the mother's biopsy was analysed. The biopsy contained a glomerular deposit in the cortex and a massive deposit at the medullary level. The deposit at the medullary level was not described in the AFib amyloidosis, being very characteristic of the Apo AI. The conclusion is that the first results of the immunohistochemistry were not completely reliable as it was not performed under adequate conditions. The presence of Apo AI was later confirmed using the same technique.
An extension study was performed, with echocardiogram and electromyography, without signs of heart condition or autonomic neuropathy. The clinical evolution showed a progressive increase in transaminases, suggesting hepatic involvement. These results were in agreement with this type of familial amyloidosis which, preferably, involves the kidney and liver. Although the intila GFR was normal, renal function subsequently deteriorated progressively until renal replacement therapy was required after 2 years. The patient received treatment with peritoneal dialysis for 4 years, thereafter the patient underwent undergoing renal transplantation from a deceased donor. The patient presented a complicated postoperative period with splenic rupture, in the context of his disease, with amyloid deposit in the spleen. An urgent splenectomy was indicated. Finally, the patient was discharged in stable condition (Figs. 1–4).
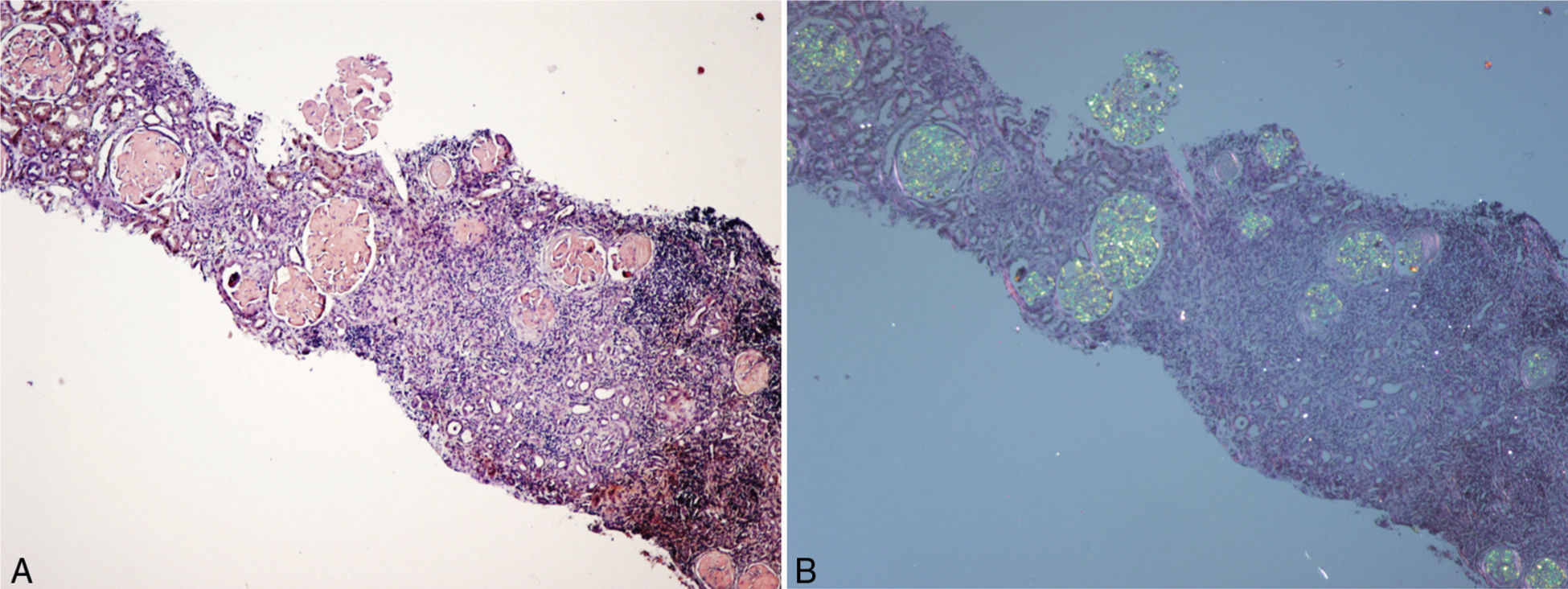
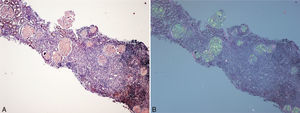
(A) Renal biopsy (son). Cortical parenchyma with massive glomerular deposit of amyloid substance stained with Congo red. (B) Renal biopsy (son). Cortical parenchyma with massive glomerular deposit of amyloid substance observed with polarised light. There is also fibrosis and interstitial inflammation (×40).
Hereditary non-neuropathic renal amyloidosis was first described by Ostertag in 1932.1 This name was given to emphasise the absence of peripheral nervous system involvement, with the purpose of differentiating this pathology from other types of amyloidosis. However, this term is confusing and is not currently appropriate because several variants related to the apolipoprotein A1 associated with neuropathy have been described since then.2 In regards to this disease, more than 25 mutations that alter the structure of different proteins, causing the deposit in amyloid form, have been identified. These proteins are lysozyme,3 Apo AI,4,5 Apolipoprotein A II (Apo II)6,7 and AFib chain.8–12 Clinically, all of them manifest themselves with renal failure at middle age and they have a dominant autosomic heritage, although with highly variable penetrance and some specific clinic characteristics, depending on the protein that originates the deposit.
Apo AI is the main protein that forms the high-density lipoprotein (HDL) structure. It is secreted in the liver and the small intestine and catabolised mainly in the liver and kidneys.13–15 It is a co-factor for lecithin-cholesterol acetyltransferase (LCAT), and its function is linked to cholesterol elimination.13,15,16
To date, at least 19 amyloidogenic mutations have been described in the APO AI gene. A different clinical presentation of the disease has been observed depending on the region in which the mutation is located. In this way, mutations that affect the amino-terminal region manifest themselves with amyloid deposit at hepatic and renal level, whereas mutations that affect the carboxi-terminal region manifest themselves with cutaneous, laryngeal and cardiac amyloidosis.2,15,17
The Apo AI amyloidosis phenotype is very heterogeneous. Some of the published cases describe extensive visceral amyloidosis and end-stage renal failure, whereas others include larynx and skin involvement, with minimal clinical consequences. Clinical expression does not only depend on the mutation location, since even patients with the same mutation may experience different clinical expression. Phenotypic heterogeneity has also been observed between lineages with the same Apo AI mutation. There are publications including series of patients with an elevated number of relatives affected and fast progression to end-stage renal disease (10 out of 16 cases affected),15 which suggests the existence of a more aggressive phenotype or other genetic and environmental factors interacting or having an influence on clinical manifestations. The possible amyloid deposit has been described in several organs, among them the kidney, gastrointestinal tract, spleen, liver, heart, peripheral nervous system, larynx and skin. The most common clinical presentation consists of high blood pressure, mild proteinuria and slowly progressive renal failure deterioration between the ages of 18 and 55.
The first mutation described, and the most frequent in patients with Apo AI amyloidosis of Irish ancestry, is Gly26Arg. It is characterised by the progressive accumulation of amyloid fibrils composed by the amino-terminal polypeptide fragments of the Apo AI and it appears in young patients, aged 20–46. The amyloid deposit is located in the peripheral nerves, kidneys, liver and gastrointestinal tract,2,17 manifesting itself clinically with peripheral neuropathy, peptic ulcer and nephropathy.2,18
At least up to 19 amyloidogenic mutations of Apo AI have been described since. Here we describe a new mutation that we did not find in the literature, the c.220T>C (p.Trp74Arg). In our patient's case, we were able to observe kidney involvement, with renal function impairment within two years, as well as liver and spleen involvement.
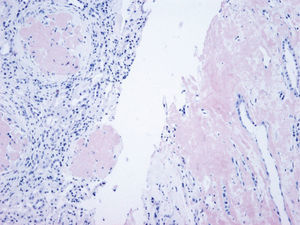
The histological analysis of Apo AI amyloidosis shows predominant amyloid deposits at the medullary level with tubulointerstitial nephritis pattern, characterised by tubular atrophy and interstitial fibrosis, associated with secondary focal glomeruloesclerosis.15,19 Although interstitial involvement is the most frequent, there are also cases in which a deposit at glomerular level has been detected.19–21 One case has even been published with predominant glomerular involvement, associated with nephrotic syndrome, as a result of Apo AI Leu64Pro amyloidosis.22
The preferred selectiveness of Apo AI amyloidosis for the renal medulla allows us to differentiate it from other types of hereditary and acquired systemic amyloidosis,19,23 since this preference for the medullary compartment is only shared with transthyretin amyloidosis.19,24
In our case, the patient's renal biopsy only contained cortex and the glomerular compartment was the only place where amyloid deposit was found. These findings are in agreement with the clinical symptoms at diagnosis, consistent nephrotic syndrome with posterior progressive worsening of renal function. Initially, this particular characteristic made us suspect fibrinogen A alpha chain amyloidosis, since it is the most frequent cause of hereditary renal amyloidosis in Europe, and its deposit is characteristically glomerular. However, the mother's biopsy had glomerular deposit in cortex, but it also had medulla with massive deposit. Finally, the genetic study determined a new, previously undescribed, Apo AI mutation, and the immunochemistry confirmed the diagnosis.
A correct differential diagnosis in systemic amyloidosis is important since disease management and prognosis may be completely different depending on its origin. This is the reason why it is particularly relevant to determine the protein that originates the deposit. In accordance with a study performed in the United Kingdom National Amyloidosis Centre, almost 10% of the patients with a presumptive diagnosis of AL systemic amyloidosis actually suffer from a hereditary amyloidosis.25 This usually happens because it is quite common to consider AL amyloidosis as a diagnosis of exclusion, due to the clinical overlapping between different types of amyloidosis, the relatively frequent finding of a monoclonal protein in patients over 50, and the difficulty to determine the type of fibril, even when the AL amyloidosis diagnosis is correct, since in up to 20% of the cases the lambda or kappa light chain antibodies are not bound to it, probably due to the alteration in their structure. Moreover, it is not always possible to verify the presence of family history of hereditary amyloidosis due to the variable penetrance of the disease and the diagnosis of new mutations.
Among the methods available to determine the protein composition of fibrils, we find immunochemistry and direct protein sequencing. In order to perform this last technique, a considerable amount of tissue is required which is not always possible to obtain. The recent development of proteomic techniques constitutes an advance in the diagnosis of amyloidosis since in order to identify the protein that forms the amyloid fibril only a small amount of fixed tissue is required.26
Regarding disease treatment, renal transplant due to Apo AI amyloidosis has had varied success.19,27,28 There has been recurrence of amyloidosis in grafts, but in most of the published cases graft survival was acceptable. A study performed in the United Kingdom National Amyloidosis Centre revealed that out of 10 renal transplant recipients with APO AI amyloidosis, five showed documented evidence of amyloid deposit in the transplanted kidney. However, only one patient, after 25 years, progressed to end-stage renal disease, initiating dialysis again.21,29
Liver transplant is the final treatment for other types of hereditary amyloidosis, since the amyloidogenic protein is synthesised in the liver. However, in the APO AI amyloidosis, the amyloidogenic protein is also synthesised in the small intestine. Although there have been cases like that of an Irish patient with the Apo AI Gly26Arg mutation, who underwent hepatorenal transplant and two years later evidenced a regression of the amyloid deposits,19,30 double hepatorenal transplant is only considered in patients with significant impairment in both organs.
In our patient's case, we considered the best option was renal transplant since hepatic function was not very impaired. However, post-transplant evolution has become complicated by splenic rupture due to amyloid deposit.
We could assert that hereditary renal amyloidosis constitutes a diagnostic challenge due to the difficulty to identify the protein that originates the deposit, and the importance of this aspect in prognosis and treatment. In APO AI amyloidosis, the indices cases diagnosis requires high clinical suspicion and a renal medulla biopsy is important to observe the amyloid deposit at this level. The possibility of this diagnosis has to be considered in family cases of tubulointerstitial nephritis with hepatic involvement.
Questions- 1)
Dr. Esther Rosello (Valencia). The diagnostic yield of amyloidosis often forces specialists to perform studies in fat, rectal mucosa and, in the end, a renal biopsy. What is the decision followed in the extraction of biopsies upon suspicion of amyloidosis?
Answer (Kelly Samillán): Upon suspicion of amyloidosis, the diagnosis can frequently be confirmed through abdominal fat aspiration, which is a mildly invasive technique. The literature states that this technique provides high sensitivity for the diagnosis of AL amyloidosis (between 80 and 90%) and a lower sensitivity (between 65 and 75%) for AA amyloidosis diagnosis. However, in the case of familial amyloidosis, the diagnostic yield is very low and the absence of positivity to Congo red in an abdominal fat biopsy does not exclude the diagnosis.31 In regards to the salivary gland and rectal mucosa biopsies, they are also used as non-invasive diagnostic methods. We believe that the diagnostic method in cases of amyloidosis must be individualised in accordance with the patient's characteristics and comorbidity. In general, if a patient presents apparent renal involvement, particularly if the patient has nephrotic syndrome, our group indicates the performance of a renal biopsy, except in the exceptional cases in which the renal biopsy would not be decisive to take decisions or in which the patient's comorbidity does not allow it. In the cases of Apo AI amyloidosis with renal involvement, in our opinion, it is advisable to perform the renal biopsy directly.
- 2)
Dr. Esther Roselló (Valencia). Do you think biopsy samples could be sent to referral centers for special amyloidosis techniques?
Answer (Manuel Solé): The diagnosis of amyloidosis presents difficulties at two levels: the basic one, the diagnosis with Congo red stain, should be undertaken in the centre itself, through adequate technique control, although we are aware of the many problems this poses. If typification is in question, it is important to use an antibodies panel and the interpretation requires experience. In this case, it would be adequate to refer the sample to an amyloidosis unit.
- 3)
Dr. Miguel Ángel Martínez (Madrid): I would like to mention that Congo red techniques are only successful if the cut sessions have a minimum of 6μm. Does that coincide with your experience?
Answer (Manuel Solé): This is a very important technical aspect. It is usually recommended to perform the stain on 8-μm sections. However, this is not always possible, particularly in renal biopsies in which routine unstained cut sections are performed for different techniques. If the cut section is thin, the stain will be pale, but the green birefringence will remain, although it may be more difficult to visualise.
- 4)
Dr. Julia Blanco (Madrid). I have noticed that you have obtained an excellent apple green in Congo red techniques, do you have a secret formula?
Answer (Manuel Solé): It is essential that the observation with polarised light be made with a powerful light source. Not just any microscope will do. And remember that the main feature of Congo red is dichroism, that is, the change from red to golden and green when rotating the polarising filter.
The authors have no conflicts of interest to declare.
Please cite this article as: Samillán-Sosa KR, Sención-Martínez G, Lopes-Martín V, Martínez-González MA, Solé M, Arostegui JL, et al. Amiloidosis renal hereditaria por depósito de apolipoproteína AI: un reto diagnóstico. Nefrologia. 2015;35:322–327.