El término glomerulonefritis membranoproliferativa denota un patrón general de daño glomerular fácilmente reconocido por microscopía óptica. Con estudios adiciones de microscopía electrónica e inmunofluorescencia, la clasificación en subgrupos es posible. El estudio por microscopía electrónica resuelve las diferencias según la localización de los depósitos electrodensos, mientras que la inmunofluorescencia detecta la composición de los depósitos electrodensos. La glomerulopatía C3 es una entidad descrita de forma reciente, una glomerulonefritis proliferativa (normalmente, pero no siempre), con un patrón de glomerulonefritis membranoproliferativa en la microscopía óptica y con depósitos de C3 aislados en el estudio de inmunofluorescencia, implicando una hiperactividad de la vía alternativa del complemento. La evaluación de un paciente con glomerulopatía C3 debe centrarse en la cascada del complemento, en la desregulación de la vía alternativa del complemento y en la cascada terminal del complemento. Aunque no hay actualmente tratamientos específicos para las glomerulopatías C3, una mejor comprensión de la patogénesis sentaría las bases para el posible uso de drogas anticomplemento como terapia de elección, como el eculizumab. En la presente revisión, se resume la patogenia de las glomerulopatías C3, centrándonos en el papel del complemento, las series de casos recientemente publicados y las opciones terapéuticas hasta el momento actual.
Membranoproliferative glomerulonephritis denotes a general pattern of glomerular injury that is easily recognised by light microscopy. With additional studies, MPGN subgrouping is possible. For example, electron microscopy resolves differences in electron-dense deposition location, while immunofluorescence typically detects the composition of electron-dense deposits. A C3 glomerulopathy (C3G) is a recently described entity, a proliferative glomerulonephritis (usually but not always), with a MPGN pattern on light microscopy, with C3 staining alone on immunofluorescence, implicating hyperactivity of the alternative complement pathway. The evaluation of C3G in a patient should focus on the complement cascade, as deregulation of the alternative pathway and terminal complement cascade underlies pathogenesis. Although there are no specific treatments currently available for C3G, a better understanding of their pathogenesis would set the stage for the possible use of anti-complement drugs, as eculizumab. In this review, we summarise the pathogenesis of the C3 glomerulopathies, focusing on the role of complement, the patient cohorts recently reported and options of treatment up to the current moment.
INTRODUCTION
C3 glomerulopathy is a recently-described entity whose principal characteristic is the exclusive deposition of C3 at glomerular level in immunofluorescence (IF) with an absence of immunoglobulin (Ig) deposits and markers for classical complement pathway activation (C1q and C4). In light microscopy (LM), the majority of the cases described show lesions typical of membranoproliferative glomerulonephritis (MPGN), although other histological patterns have been described. Electron microscopy (EM) has shown electron-dense, mesangial, intramembranous and/subendothelial deposits.
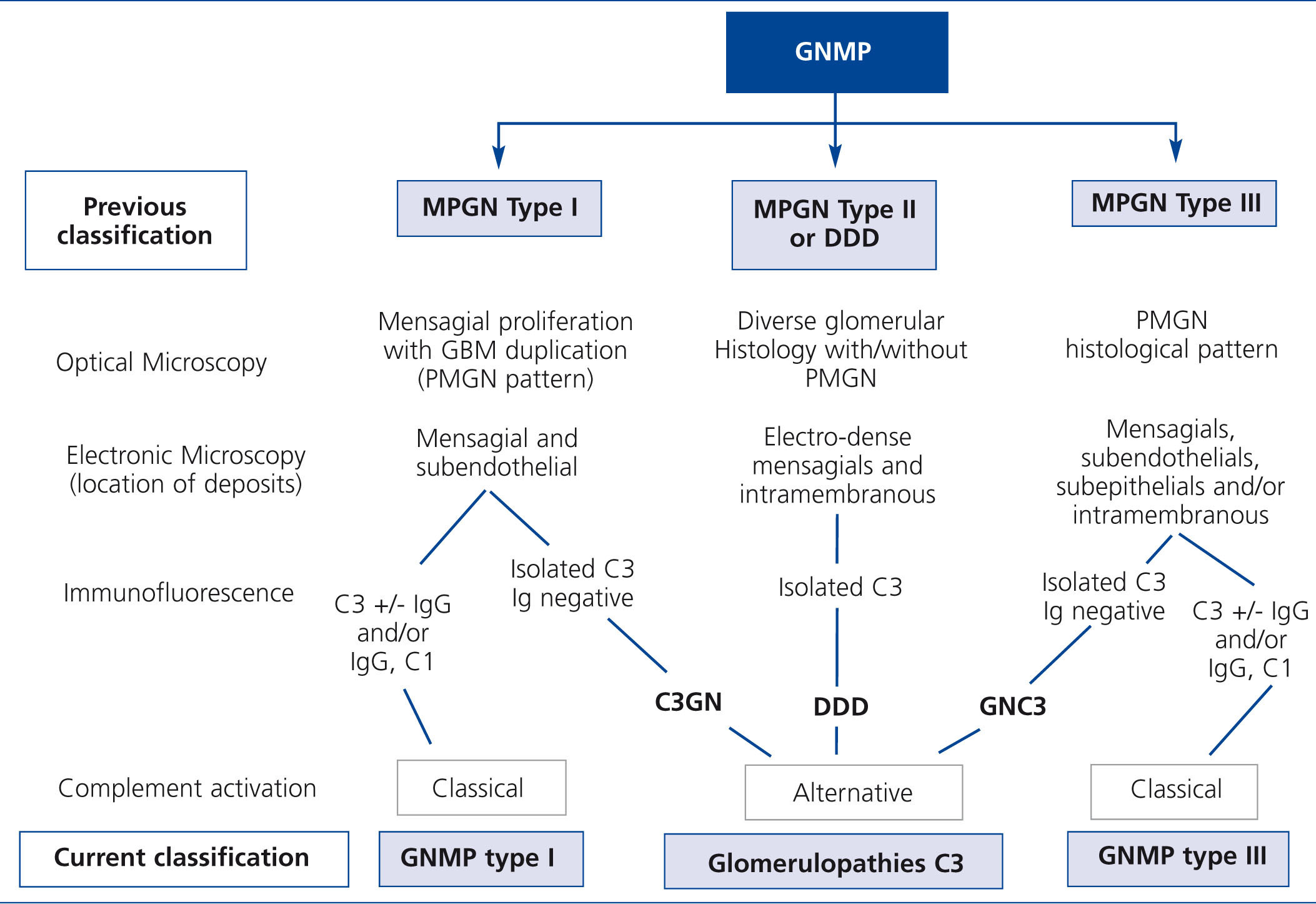
Until recently, the main classification of MPGN made a distinction between secondary causes when it was possible to identify an underlying aetiology (such as, for example, cryoglobulinemia, autoimmune diseases and infections) or idiopathic forms when such aetiology did not exist. Additionally, the traditional histological classification - MPGN type I (MPGN I), type 2 (MPGN II) and type 3 (MPGN III) - was based on ultrastructural appearance and the location of electron-dense deposits1. Both classifications presented problems, as neither was wholly based on pathogenesis.
In recent years, advances in knowledge regarding the role played by the complement system in its pathogenesis has recently made it possible to describe this new entity characterised by the presence of C3 in the absence of significant quantities of Ig or classical complement pathway activation components and to name it C3 glomerulopathy. This new advance allows, in turn, to focus on physiopathology, making it possible to decide how to assess and treat this new entity.
HISTORICAL DEVELOPMENT OF THE CLASSIFICATION OF MEMBRANOPROLIFERATIVE GLOMERULONEPHRITIS
The histological pattern of MPGN is characterised by mesangial expansion and the duplication of glomerular baseline membranes, typically associated with immune deposits on the walls of the glomerular capillaries. Traditionally, MPGN have been classified according to histological and ultrastructural findings. This pattern of glomerular lesion is easily identified by LM, making the diagnosis of MPGN relatively simple.
EM studies provide a classification by location of electron-dense deposits: subendothelial and mesangial deposits in MPGN I; intramembranous and mesangial deposits in MPGN II, also known as dense deposit disease (DDD)2. In MPGN III, deposits may be subendothelial and subepithelial or intramembranous, subepithelial and subendothelial, disorganising the lamina densa. However, this classification is not without its problems and, for example, in a single biopsy it is possible to see both subtypes overlapping.
A major advance in our knowledge brought about a subsequent reclassification, giving particular attention to the composition of the deposits detected using IF. MPGN I deposits typically contain Ig (IgM and/or IgG) as well as complement (C3 and/or C4), unlike MPGN II or DDD, which is characterised by isolated C3 deposits. MPGN III usually has Ig deposits and complement but nonetheless may have isolated C3 deposits.3
By the 1980's, some pathologists had already begun to point out the presence of isolated C3 deposits in MPGN types I and III, filtering out these cases from the more common variants containing Ig.3,4 These Ig-negative lesions, initially named idiopathic MPGN with isolated C3 deposits, came to be known as C3 glomerulonephritis (C3GN).
IMMUNE COMPLEX-MEDIATED MEMBRANOPROLIFERATIVE GLOMERULONEPHRITIS COMPARED WITH MEMBRANOPROLIFERATIVE GLOMERULONEPHRITIS MEDIATED BY ALTERNATIVE COMPLEMENT PATHWAY ACTIVATION
The complement system is divided into three initial pathways: classical, lectin and alternative. In cascade form, all three converge in C3 to create an enzymatic complex called C3 convertase which cleaves C3 into C3a and C3b. The association of C3b with C3 convertase produces C5 convertase, which activates the terminal complement cascade (TCC) and the formation of the membrane attack complex on cell surfaces, resulting in cell lysis.5
Classical complement pathway activation by antigen-antibody (Ag-Ab) immune complexes (IC) causes both IC and complement factors to deposit in the mesangium and the glomerular capillaries, as detected by IF studies. However, on rare occasions, when a proliferative glomerulonephritis is marked by the presence of complement, usually C3 without Ig, this IF pattern involves activation of the alternative complement pathway and the isolated deposit of complement factors in the tissues.6
The merging of histopathological observations of isolated C3 deposits with the advances in knowledge of the defects of the alternative complement pathway in these lesions led to proposals for a new nomenclature.7 The term C3G came about as a recently-discovered subgroup encompassing DDD and C3GN, underlining the pathogenic importance of deregulation of the alternative complement pathway.5 The current approach, therefore, distinguishes those forms of MPGN with isolate C3 deposits (including DDD and C3GN) as alternative complement pathway-mediated C3G from those cases of MPGN that are mediated by the classical complement pathway with deposits of Ig and complement (Figure 1).8
PATHOGENESIS OF C3 GLOMERULOPATHIES
In C3G, defects in complement regulatory proteins promote excessive activation of the alternative complement pathway in the fluid phase, causing deposition of complement product, including C3b-degradation products and components of the terminal complement cascade on the wall of the glomerular capillaries.9 The alternative pathway maintains constant low-level activity in the circulation (the tick-over mechanism). In order to prevent self-damage, this activation is tightly regulated by both activator and inhibitor proteins. One important advance was the discovery of genetic mutations or deficits in these regulatory factors in subjects with isolated deposits of C3.10
A similar defect of frenzy activity might also be caused by acquired autoantibodies, the best-known being C3 nephritic factor (C3Neph), which directly stabilizes the activation of C3 complex convertase and prevents the inhibiting action of complement regulatory factor H (CFH).11
Recently, a number of family case studies have been published providing unique knowledge of the pathogenesis and presentation of DDD and C3GN, making clear at what specific level the dysfunction occurs in the complement system. These family anomalies could bring clarity to therapies (both existing and developing) that would be beneficial for this illness.
The main familiar example of C3G was recently described by Gale et al.,12who identified two families in Cyprus with autosomal-dominant microscopic hematuria whose renal biopsy turned up a C3G with lesions different from MPGN. The genomic study revealed an abnormality in the CFH/CFHR region of the chromosome 1q31-32. The researchers identified in these families a mutation of the protein CFHR5, which is less effective than native CFHR5, suggesting a dominant-negative mechanism of action in the control of C3 and C5 convertase activity. This familiar glomerulopathy, endemic to Cyprus, has been named CFHR5 nephropathy.
This defect illustrates the importance of CFHR5 in the processing of complement in the kidney, making identification of new mutations of CFHR5 in DDD and C3GN patients an interesting prospect. Another family, described by Habbig et al.,9 includes two brothers of related parents with hematuria and childhood-onset proteinuria. Studies of complement showed a drop in serum C3 and complement regulatory factor B, with normal C4 and increased C3d. The biopsy revealed mesangial C3 and C5b-9 deposits, while EM showed numerous mesangial, intramembranous and subendothelial deposits. Screening of CFH mutations showed that both boys were homozygous for deletion of an amino acid, lysine, in position 224. This deletion leads to complement control defects, severely reducing the cofactor with C3b activity and deteriorating integrity due to the mutated factor H. In addition, both patients and their healthy mother were positive for C3Neph, underlining the role of the factor H defect in this family's pathogenesis.
In 2010, Martínez-Barricarte et al.13 identified a small family core with DDD with the C3 mutation. This mutation made C3 resistant to cleavage by C3 convertase, consequently making the formation of C3b impossible. This case provided conclusive proof that the illness in this family was attributable exclusively to deregulation in the fluid phase of the alternative pathway without any contribution from the TCC. In contrast, the C3G found in Cypriot families described by Gale et al.12 and the brothers described by Habbig et al.9 are associated with the deregulation of C5 convertase and subsequent activation of the TCC.
Taken together, these family cases suggest that C3G is a disease spectrum, seeming to be less homogenous in reality than the nomenclature suggests. The presentation and prognosis of the disease depend on the level and degree of the dysfunction of the alternative pathway and the terminal complement cascade. The histopathology also illustrates the heterogeneity of the pathogenesis. Cases have been published of post-infectious glomerulonephritis progressing to C3GN or DDD.14 With regard to histology, the morphological expression of the glomerular damage caused by deregulation of the alternative complement pathway can be remarkably diverse: from mild mensangial and endocapillary proliferation to lesions compatible with MPGN and even glomerulonephritis with extracapillary proliferation.15
There is an ever greater number of published cases that attempt to clarify the pathogeny of the term "C3 glomerulopathy". In a recently published cohort, Servais et al.16 describe a protein analysis and genetic screening of regulatory proteins of the alternative complement pathway in a series of 134 patients (including cases of DDD, C3GN and MPGN I) that found a defect in the control of the alternative complement pathway due either to antibodies or mutations in 71% of the cases. In this study, over 53% of the MPGN I patients were found to have dysfunction of the alternative complement pathway. The role these dysfunctions play in the pathogenesis of MPGN I is yet to be resolved, as it is a IC disease and might be mediated in some way by deregulation of the alternative pathway. The American series published by Sethi et al.17 involves 12 subjects (C3GN cases only) and the complement assessment revealed a dysfunction in the alternative complement pathway in all. In the American cohort, 9 of the 12 patients with C3GN had initially been classified as MPGN I (n=4), MPGN III (n=1) and postinfectious glomerulonephritis (n=4). One possible explanation for the French cohort may be that these patients were previously classified incorrectly as MPGN I, as was the case with the American cohort. The demonstration by Sethi et al.17 that the proteomic profile of C3GN by mass spectrometry is similar to that of DDD (with a predominance of C3 and final complement pathway components) supports the theory that the presence of isolated C3 is the "major marker" for dysfunction of the alternative complement pathway in C3G, irrespective of histology and ultrastructural features.
ASSESSING PATIENTS WITH C3 GLOMERULOPATHY
While assessment of an IC-mediated MPGN is centered on identification of the underlying etiology in order to begin a specific treatment, assessment of C3G has to focus on identifying the underlying defect in the alternative complement pathway. This assessment should include: genetic tests, antibody studies and assays of the activity of the alternative complement pathway.1 The genetic test should look for mutation in genes codifying complement regulatory proteins, principally CFH, CFI and membrane cofactor protein, but it should also include CFHR1, CFHR2, CFHR3, CFHR4 and CFHR5 if available. It should include autoantibodies such as C3Neph and antibodies that block the action of inhibitory mechanisms, such as factor H autoantibodies.18 Other alternative pathway screening tests used in recent C3G studies are: haemolysis assay, functional assay of the alternative pathway and serum levels of membrane attack complex (sMAC).17
TREATMENT AND PROGNOSIS
The information available concerning the development, long-term prognosis and the various treatments employed for C3 glomerulopathies is scarce. As the number of cases identified rises, the hope is to correlate the screening findings on complement functions and mutations with the natural progression of the disease, development kidney disease and long-term prognosis. As with all glomerular diseases, it is important to take into account clinical parameters such as the degree of renal dysfunction and proteinuria at the moment of diagnosis.
The diversity of C3G physiopathology probably explains the varying responses obtained from the treatments. For example, in the family described by Habbig et al.,9 factor H repletion could be beneficial. Factor H replacement is currently done by means of plasma exchange, although direct application of the factor itself may be available for therapeutic use in the near future.19 However, there are patients who do not respond to plasma exchange or factor H substitution. An example of this is the family with EDD described by Martínez-Barricarte et al.,13 which may have a mutation in C3 convertase that makes it resistant to factor H, consequently requiring specific treatments to restore C3 convertase control and activity and eliminate C3 degradation products in the bloodstream.20
An extremely significant advance in relation to diseases caused by complement deregulation has been the availabilty of eculizumab, a monoclonal antibody that binds with great affinity to C5 and prevents the production of membrane attack complex. As C3G are marked by the deposition of complement products, including C3b and TCC degradation products, treaments targeting the membrane attack complex could be beneficial in treating the disease.21 Until very recently, only individual cases had been described,22-25 three with DDD and another diagnosed with treatment-resistant MPGN I. In these four cases, proteinuria and creatinine both went down after treatment with eculizumab. Recently, Bomback et al.26 published a series of 6 cases (3 with DDD and 3 with C3GN) treated with eculizumab over the course of a year. Their findings suggest that eculizumab might be a beneficial treatment for the EDD and C3GN subgroup. After a year of treatment, two of the patients showed an improvement in renal function, another had remission of nephrotic syndrome and another, whose laboratory parameters had remained stable, showed a histopathological improvement in a new kidney biopsy. However, the two remaining patients showed a deterioration of renal function during follow-up. In this case series, the elevated sMAC levels were normalised once the treatment began, demonstrating a relationship between this normalization and improvement of renal function and a drop in proteinuria. These data suggest that elevated levels of sMAC prior to treatment can be a predictor for response to treatment with eculizumab.26
In the family published by Martínez-Barricarte et al.13, the DDD was caused by a deregulation in the fluid phase of the alternative complement pathway, without any contribution from TCC. In this family, therefore, the sMAC levels would not be elevated and eculizumab would be of no use. In the families described by Gale and Habbig,9,12 the disease is associated with deregulation of C5 convertase and TCC activation, giving reason to expect a beneficial response to eculizumab. Therefore, the variability in the responses to treatment will depend on the physiopathology of these two diseases. In a review recently published by Zuber et al.21 on the use of eculizumab in atypical haemolytic-uremic syndrome and C3 glomerulopathies, the authors are of the opinion that "optimal candiates for treatment with eculizumab in C3G would be patients who have had the disease for a short time and have active inflammatory lesions (endocapillary proliferation and crescents) and minimal instersitial fibrosis in the kidney biopsy with a recent increase in serum creatinine and/or proteinuria and elevated sMAC levels".
At present, the prognosis for C3G is poor, progressing to terminal chronic kidney disease in the majority of patients. The data recently published on the Servais et al. cohort16 reveal worse results for ten-year renal survival rates for DDD patients than those with MPGN I and C3GN. In their cohort, Sethi et al.17 also describe worse results for DDD than C3GN, suggesting that C3GN might be a less aggressive entity. One explanation might be the higher rate of C3Neph in DDD - 80% versus 45-50 % in C3GN. However, case series will undoubtedly be necessary in order to assess the long-term prognosis of this new entity.
CONCLUSIONS AND THE GLOSEN COLLABORATIVE WORK PROPOSAL
1. The recent description of C3G opens a new path for the pathogenic and therapeutic approach to cases that are today catalogued as primary or idiopathic. Although most of the cases described involve MPGN, it should be remembered that there are cases with other patterns of histological damage and that the clinical manifestations can be considerably diverse.
2. The available information on the progress, long-term prognosis and response to the various treatments used on C3G is scarce, as the number of patients identified is small and the clinical progress data published thus far is poor. From what has been described to date, it seems that there are remarkable divergences in development, ranging from very aggressive cases with rapid progression to chronic dialysis to cases of indolent clinical course or even spontaneous remission.
3. The in-depth knowledge of the complement system gained over the last few years makes the study of C3G patients particularly interesting for the identification of the pathogenic bases of each case (mutations in complement regulatory genes, antibodies against regulatory factors). In addition, complement plays a pivotal role in the pathogenesis and progress of renal damage in numerous cases of primary and secondary glomerulonephritis, meaning that the lessons we learn from these 3CG may produce knowledge applicable to other glomerular entities.
4. Undoubtedly, the availability of complement-blocking drugs has been a decisive spur to interest in glomerular entities directly mediated by complement. Eculizumab is a specific inhibitor of terminal complement complex (C5b-9) recently approved by the American and European regulatory agencies for treatment of haemolytic-uraemic syndrome owing to its efficacy and good tolerance. There are already clinical cases of C3G (membranoproliferative type I, dense deposit disease) with a good response to eculizumab.22-26
5. In view of the foregoing, extensive C3G case series are needed in order to create a more definitive clinical and histological profile to identify prognosis factors and to analyse the response to assayed treatments. Furthermore, the diseases identified can be studied in more depth in order to describe the complement abnormalities involved. We believe The Spanish Nephrology Society's Glomerular Diseases Study Group (GLOSEN) would provide the ideal framework in which to develop work of this nature, since the Group's potential has already been demonstrated by the widespread impact of its collaborative work.27,28 While a work proposal and data-collection protocol is to be submitted to members shortly, we take the opportunity here to send a message to arouse the interest of all nephrologists and pathologists interested in glomerular diseases and request their collaboration in the study.
KEY CONCEPTS
1. C3G is characterised by isolated deposition of C3 in IF, without Ig and classical complement pathway factors.
2. The majority of cases describe involve MPGN types I, II (dense deposit disease) and III, although there are also descriptions of patients with other glomerular lesions.
3. Its pathogenesis is due to dysregulation of the alternative complement pathway, either by mutations in the genes controlling the regulatory factors of this system (principally factor H) or by the formation of autoantibodies aimed at said factors.
4. Dysregulation of the alternative pathway gives rise to anomalous activity of terminal complement attack factor (C5b-9) causing cell lysis and tissue damage in the glomerular capillaries.
5. Although experience is still very limited, there are description of positive responses by C3G patients to the administration of eculizumab, an antibody that selectively inhibits the complex C5b-9.
6. Further studies - preferably multicentre, in view of the apparently low prevalence of the disease - are needed in order to define more precisely its clinical, morphological and pathological characteristics. Likewise, the data available on prognosis and response to different assayed treatments is very scarce.
7. Progressive knowledge of the molecular and genetic bases of these selective anomalies (congenital or acquired) of complement which cause glomerular damage may open the way for new therapeutic alternatives for glomerular diseases, in which the role of complement has historically been recognized. The connection with other clinical entities associated with complement dysregulation (haemolytic-uraemic syndrome, macular degeneration) would also surely open up some extremely important lines of research.
Acknowledgements
Cristina Rabasco and Manuel Praga are members of the "Grupo de Investigación Complemento-CM" (Complement-CM Research Group), supported by the Community of Madrid R+D programmes (S2010/BMD-2316). This work has received help from the Association for Research and Treatment of Renal Diseases (AITER) and the Healthcare Research Fund (FIS) (10/02668).
Conflict of Interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Figure 1. Classification of Membranoproliferative Glomerulonephritis