Extended hemodialysis (HDx), with medium cut-off membranes, could favor the effective elimination of medium-sized molecules such as IL-6 and other inflammatory mediators.1 In the current literature, there are few reports on the use of these membranes in hemodialysis patients with SARS-CoV-2 infection, but their potential therapeutic effect is not described.2
We present the case of a 68-year-old man with a history of hypertension type 2 diabetes mellitus, congestive heart failure, sleep apnea-hypopnea syndrome, ischemic heart disease and chronic kidney disease stage G3a A3 according to 2012 KDIGO of probable diabetic etiology with serum basal creatinine values of 1.3–1.5 mg/dl.
He referred a recent trip to the SARS-CoV-2 risk area and came to the Hospital Emergency Department for a 24-h course of illness characterized by fever and associated dry cough. Nasopharyngeal exudate was performed, which was positive for SARS-CoV-2. On the day of admission to the hospital's Emergency Department (day 1), dyspnea to moderate efforts was added to the symptoms, so the patient was admitted to the hospital with subsequent, almost immediate, transfer to the Intensive Care Unit (ICU) due to torpid evolution. His respiratory symptoms are compatible with bilateral pneumonia complicated by acute respiratory distress syndrome and multi-organ failure: hypotension (mean arterial pressure of 60 mmHg), tachypnea, use of accessory musculature, and O2 saturation of 89% despite reservoir mask (FiO2 99%). The patients requires mechanical ventilation and inotropic drugs (norepinephrine and dopamine). In addition, it presents a situation of oliguria (10 mL/h) that initially responds to intravenous diuretics, presenting, in parallel, deterioration of renal function up to serum creatinine of 3.4 mg/dl, so on day 4 it is decided to start continuous venovenous hemofiltration (CVVH) that is maintained until day 26. He received treatment with hydroxychloroquine, azithromycin, lopinavir/ritonavir, interferon beta, ceftriaxone, levofloxacin, methylpredinisolone and heparin anticoagulation intermittently due to bleeding tendency. From day 21 to day 36, he received treatment with cloxacillin and cefepime, successively, due to the presence of methicillin- sensitive Staphylococcus aureus in the bronchial aspirate and worsening of the inflammatory parameters.
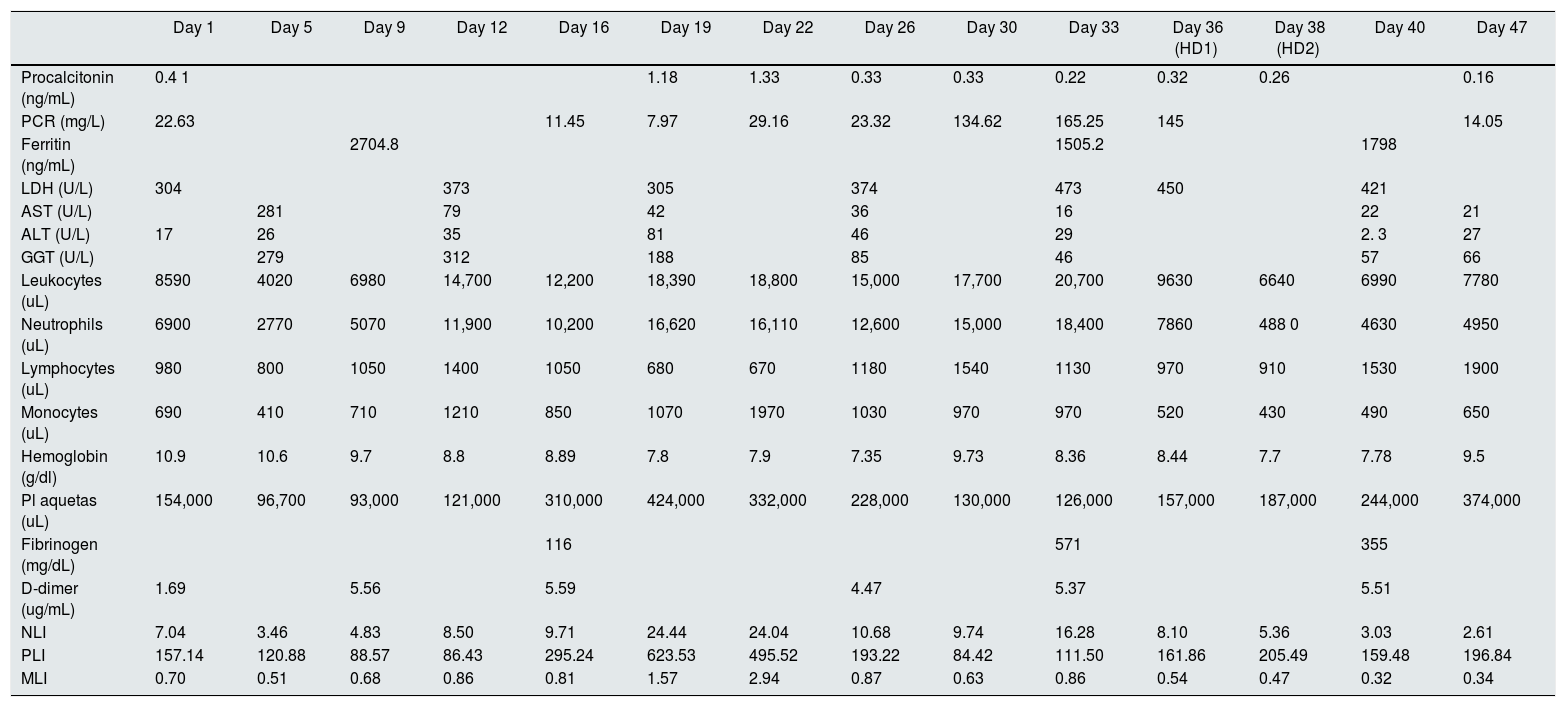
Since the patient has anasarca and persistent serum creatinine levels of 2 mg/dl and urea 162 mg/dl, intermittent hemodialysis with Theranova 400® 1.7 m2 filter was started (Baxter International Inc., Deerfield, IL, USA.) with a QB of 270−300 ml/min and a QD 500 mL/min, requiring only two sessions (days 36 and 38). Given the clinical improvement, her discharge from the ICU was decided on day 47 with serum creatinine levels of 1.3 mg/dl. The analytical parameters evolution is described in Table 1.
Evolution of inflammatory parameters of the patient presented.
| Day 1 | Day 5 | Day 9 | Day 12 | Day 16 | Day 19 | Day 22 | Day 26 | Day 30 | Day 33 | Day 36 (HD1) | Day 38 (HD2) | Day 40 | Day 47 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Procalcitonin (ng/mL) | 0.4 1 | 1.18 | 1.33 | 0.33 | 0.33 | 0.22 | 0.32 | 0.26 | 0.16 | |||||
| PCR (mg/L) | 22.63 | 11.45 | 7.97 | 29.16 | 23.32 | 134.62 | 165.25 | 145 | 14.05 | |||||
| Ferritin (ng/mL) | 2704.8 | 1505.2 | 1798 | |||||||||||
| LDH (U/L) | 304 | 373 | 305 | 374 | 473 | 450 | 421 | |||||||
| AST (U/L) | 281 | 79 | 42 | 36 | 16 | 22 | 21 | |||||||
| ALT (U/L) | 17 | 26 | 35 | 81 | 46 | 29 | 2. 3 | 27 | ||||||
| GGT (U/L) | 279 | 312 | 188 | 85 | 46 | 57 | 66 | |||||||
| Leukocytes (uL) | 8590 | 4020 | 6980 | 14,700 | 12,200 | 18,390 | 18,800 | 15,000 | 17,700 | 20,700 | 9630 | 6640 | 6990 | 7780 |
| Neutrophils (uL) | 6900 | 2770 | 5070 | 11,900 | 10,200 | 16,620 | 16,110 | 12,600 | 15,000 | 18,400 | 7860 | 488 0 | 4630 | 4950 |
| Lymphocytes (uL) | 980 | 800 | 1050 | 1400 | 1050 | 680 | 670 | 1180 | 1540 | 1130 | 970 | 910 | 1530 | 1900 |
| Monocytes (uL) | 690 | 410 | 710 | 1210 | 850 | 1070 | 1970 | 1030 | 970 | 970 | 520 | 430 | 490 | 650 |
| Hemoglobin (g/dl) | 10.9 | 10.6 | 9.7 | 8.8 | 8.89 | 7.8 | 7.9 | 7.35 | 9.73 | 8.36 | 8.44 | 7.7 | 7.78 | 9.5 |
| Pl aquetas (uL) | 154,000 | 96,700 | 93,000 | 121,000 | 310,000 | 424,000 | 332,000 | 228,000 | 130,000 | 126,000 | 157,000 | 187,000 | 244,000 | 374,000 |
| Fibrinogen (mg/dL) | 116 | 571 | 355 | |||||||||||
| D-dimer (ug/mL) | 1.69 | 5.56 | 5.59 | 4.47 | 5.37 | 5.51 | ||||||||
| NLI | 7.04 | 3.46 | 4.83 | 8.50 | 9.71 | 24.44 | 24.04 | 10.68 | 9.74 | 16.28 | 8.10 | 5.36 | 3.03 | 2.61 |
| PLI | 157.14 | 120.88 | 88.57 | 86.43 | 295.24 | 623.53 | 495.52 | 193.22 | 84.42 | 111.50 | 161.86 | 205.49 | 159.48 | 196.84 |
| MLI | 0.70 | 0.51 | 0.68 | 0.86 | 0.81 | 1.57 | 2.94 | 0.87 | 0.63 | 0.86 | 0.54 | 0.47 | 0.32 | 0.34 |
HD1: first hemodialysis session; HD2: second hemodialysis session; MLR: monocyte-to-lymphocyte ratio; NLR: neutrophil-to- lymphocyte ratio; PLR: platelet-to-lymphocyte ratio
Taking into account the analysis before and after therapy, there is a tendency to a decrease in procalcitonin, C-reactive protein (PCR), the neutrophil-to-lymphocyte ratio (NLR) and the monocyte-to-lymphocyte ratio (MLR) (). In addition, there is an increase in the levels of platelets, lymphocytes and the platelet-lymphocyte ratio (PLR) (PLI), the antiphospholipid antibodies and lupus anticoagulant were negative. Neither IL-6 nor IL-1 levels were measured because no specific treatment to control these cytokines was started.
Currently, many aspects of the pathogenesis of SARS-CoV-2 infection are unknown. A “cytokine storm” has been proposed as one of the key aspects in the torpid course of some patients.3 Therefore, it has been considered, among others, the use of antagonists of IL-6 and IL-1 such as tocilizumab and anakinra respectively.3 Similarly, in sepsis, the effect that the use of high-permeability membranes could have on cytokine removal has been recognized.4
The HDx is a renal replacement therapy modality recognized for its high convection capacity and removal of medium-sized molecules (eg, cytokines) without further removal of albumin; but, unlike online hemodiafiltration, it maintains its effectiveness despite not having high QB, as is often the case in ICU patients with temporary catheters.1 Therefore, with a view to a possible SARS-CoV-2 outbreak in our hospital, we established a protocol in which, in cases such as the one described, we would use HDx using mediun cut-off membranes (Theranova®).
The PCR, neutrophil-to-lymphocyte, platelet-to-lymphocyte, and monocyte-to-lymphocyte ratios are parameters that have been shown to be useful as inflammation and prognostic markers in different entities, including in SARS-CoV-2 infection.5–7 In our patient, a trend towards improvement of these parameters can be seen parallel to the initiation of therapy, especially NLR and MLRI (Table 1). However, given that this is a single case and there are concomitants factors, such as the evolution of infection process itself, we cannot affirm that the effects obtained are attributable to the use of the aforementioned filter.
In conclusion, we consider that these findings should be taken into account for the design of larger studies and the use of these membranes could be considered, if they are available, due to their potential immunomodulatory effect, in patients requiring hemodialysis and affected by infection by SARS-CoV-2; especially at the beginning of stage III proposed by Siddiqi et al.8 when the inflammatory component is predominant.
Please cite this article as: Valga F, Vega-Diaz N, Monzon T, Gonzalez-Cabrera F, Santana A, Baamonde E, et al. Papel de la hemodiálisis extendida en COVID-19: a propósito de un caso. Nefrologia. 2020;40:487–489.