Proton pump inhibitors (PPIs) are used in the treatment of dyspepsia, peptic ulcer, gastroesophageal reflux, as well as in the prevention of gastropathy derived from the use of NSAIDs. They are generally well tolerated and with few side effects which makes them among the most prescribed medications. We present the case of a patient who developed acute interstitial nephritis (AIN) after the use of PPI.
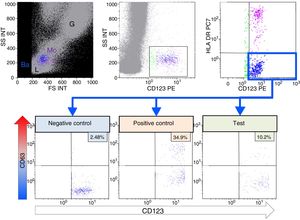
A 31-year-old woman with no previous medical history of interest who started treatment with omeprazole for epigastric pain. Two days later, the lab work revealed a deterioration of renal function (Cr: 2.28mg/dl and eGFR of 27ml/min). After one week of treatment, there was a worsening of renal function (Cr.2.75mg/dl and eGFR 22ml/min), and omeprazole was discontinued. Complementary tests showed proteinuria (+) and leukocyturia ++ (10–15/field). Given the progressive worsening of renal function (up to Cr of 3mg/dl), it was decided to perform a renal biopsy. The sample contained 21 glomeruli without sclerosis and the interstitium had edema and massive infiltration of lymphocytes and numerous eosinophils, and absence of granulomas or vasculitis. Extensive C4d staining in peritubular capillaries. With these findings, prednisone was initiated orally at a dose of 1mg/kg for 10 days, followed by a descendingcorticoid regimen until its complete suspension 6 months later. In the presence of suspected interstitial nephropathy secondary to omeprazole, a basophil activation test (BAT) was performed, showing a 10.2% activation rate and a stimulation index of 4.1, which confirmed the diagnosis.
Drug-induced AIN is a frequent cause of acute renal failure (ARF), characterized by the presence of inflammatory infiltrates and interstitial edema, tubulitis, and in some cases, interstitial fibrosis. It has been confirmed that after the withdrawal of the inducer drug between 30 and 70% of patients do not fully recover their baseline renal function, remaining with some degree of renal failure, and may progress to chronic kidney disease (CKD) and death.1,2 The AIN is present in 1–3% of the biopsies performed for the evaluation of hematuria or proteinuria, and approximately 15% of those performed in patients with ARF, although its actual incidence may be higher, especially among elderly patients >65 years old, in many cases polymedicated with combinations of antibiotics and NSAIDs.3–5 The incidence due to PPIs is relatively low, although its massive use and sometimes without justification, motivates the increase observed in recent years. The fact that the AIN occurs only in a small percentage of patients exposed to a given drug, the absence of a dose/dependent relationship, the association with extrarenal manifestations of hypersensitivity and the recurrence of new doses of medication suggest an immunological basis as a cause of the AIN,4 which has led to the use of corticosteroids for its treatment.
Basophils are cells used for the in vitro study of allergic reactions. They constitute a small fraction of total leukocytes (<0.3%) and just as mast cells express on their surface high affinity IgE receptors to which the specific IgE binds that in contact with the allergen causes the activation of basophil and the subsequent release of various cytokines and mediators of inflammatory reactions that are responsible for the symptomatology of allergic reactions.
The basophil activation test (BAT), with the support of flow cytometry for the quantitative determination of the degranulation of basophils, constitutes a complementary method for the diagnosis of drug allergies, especially for new or non-detectable drugs by means of serological techniques, such as the determination of IgE. Specific, allowing to avoid in many cases, the performance of the allergen exposure test and unlike other procedures, allows the simultaneous evaluation of several drugs.6,7
In our case, it was highlighted the early onset of manifestations of kidney damage and the rapid aggravation of ARF. The keys to the diagnosis were based on a high level of clinical suspicion for an ARF of unknown origin, associated to leukocyturia and exposure to a known drug inducing allergic hypersensitivity reactions. The rapid discontinuation of drug exposure and the favorable response to corticosteroids were the key to the recovery of the patient's renal function. The results of the BAT were conclusive. With a percentage of activation greater than 5% and a stimulation index higher than 2, the suspicion based on the inducing agent was confirmed (Fig. 1).
Basophil activation test. By means of flow cytometry, different leukocyte subpopulations can be differentiated (lymphocytes (L), monocytes (Mo), granulocytes (G)), by size or forward scatter (FS) and complexity or side scatter (SS). Basophils (Ba) in blue are identified by expression of the IL-3 receptor (CD123) and absence of HLA-DR expression. Degranulation of basophils is evaluated by the % expression of CD63 in the absence of stimulus (Negative Control), after activation with fMLP and when incubated with omeprazole. The stimulation index is calculated with the quotient between the %CD63 of the test and the %CD63 of the negative control.
We conclude that in the presence of an unexplained ARF and exposure to PPIs, even without the presence of eosinophiluria and pyuria, physicians should not exclude the risk of AIN, as well as discourage the indiscriminate use of these drugs. The BATs are useful tools for the identification of AIN-inducing drugs.
Please cite this article as: Belmar Vega L, López Hoyos M, San Segundo Arribas D, Irure Ventura J, Fernández Fresnedo G, Ruiz San Millán JC, et al. Test de activación de basófilos. Herramienta para el diagnóstico de nefritis intersticial. Nefrologia. 2019;39:436–438.