AA (secondary) amyloidosis is a severe complication of chronic inflammatory disorders. It is characterized by the systemic deposition of an abnormal protein called amyloid, affecting mainly renal function. IL-6 is a cytokine with a relevant role in this disease development. Interleukin-receptor antagonists, like Tocilizumab (TCZ), have become possible treatment choice for AA amyloidosis. In published reports, TCZ has shown good efficacy for AA amyloidosis, being associated with regression of renal amyloid deposits.
MethodsRetrospective review that included patients with histological diagnosis of AA renal amyloidosis under treatment with TCZ during the years 2018–2019 in our center. We have registered clinical and demographic variables. Renal function was measured by means of CKD-EPI equation to estimate the glomerular filtration rate (FG) and protein/creatinine ratio (IPC) at 3, 6 and 12 months. We define renal response as a decrease by at least 30% of proteinuria and/or stabilization or improvement of FG. We consider that an anti-inflammatory response is a decrease of more than 50% in serum amyloid protein (PSA) and/or C-reactive protein (CRP).
ResultsWe collected 3 cases of patients with histologically proven AA amyloidosis treated with TCZ (2 men; 1 woman; aged 55, 74 and 75 years). The follow-up was 13, 14 and 75 months. FG was stabilized in two patients. The third patient remained on hemodialysis during follow-up, although with excellent control of her underlying inflammatory disease. In all three cases, reduced PSA and CRP were observed. There have been no adverse events.
ConclusionsThe TCZ may be an effective and safe option in treatment of AA amyloidosis with renal involvement. Our results position it as an interesting therapeutic option to consider in these cases, although prospective studies would be necessary to evaluate the global role of TCZ in AA amyloidosis.
La amiloidosis secundaria (AA) es una complicación grave asociada a enfermedades inflamatorias. Se caracteriza por el depósito sistémico de proteína fibrilar AA, con especial repercusión renal. La participación de la interleucina 6 en su mecanismo patogénico ha supuesto que tocilizumab (TCZ) sea considerado una opción terapéutica en estos pacientes. Varias series publicadas muestran su eficacia en el tratamiento de la amiloidosis AA, permitiendo incluso la regresión de depósitos renales ya presentes.
Material y métodoRevisión retrospectiva que incluyó pacientes con diagnóstico histológico de amiloidosis renal AA en tratamiento con TCZ durante los años 2018–2019 en nuestro centro. Registramos variables clínicas y demográficas; evaluamos la función renal mediante filtrado glomerular (FG) calculado por CKD-EPI e índice proteína/creatinina a los 3, 6 y 12 meses de seguimiento. Definimos «respuesta renal» como la disminución > 30% de la proteinuria y/o estabilización o mejoría del FG. Consideramos «respuesta antiinflamatoria» la disminución > 50% de las cifras de proteína sérica amiloide (PSA) y/o proteína C reactiva (PCR).
ResultadosPresentamos una serie de 3 pacientes (2 varones y una mujer; 55, 74 y 75 años, respectivamente), con un tiempo de seguimiento de 13, 14 y 75 meses, respectivamente. Con la terapia con TCZ, el FG se estabilizó en 2 pacientes; el tercero permaneció en hemodiálisis durante el seguimiento, aunque con excelente control de su enfermedad inflamatoria de base; a los 12 meses recibió un trasplante renal. En los 3 casos se objetivó reducción de proteína PSA y PCR. No se han producido eventos adversos.
ConclusionesEl TCZ es una opción terapéutica eficaz y segura para los pacientes con amiloidosis AA y afectación renal. Nuestros resultados lo posicionan como una opción terapéutica a considerar en estos casos, aunque sería necesario la realización de estudios prospectivos en los que se evalúe el papel global de TCZ en la amiloidosis AA.
Secondary amyloidosis (AA) is a serious complication associated with chronic inflammatory diseases, highlighting among the most common: rheumatoid arthritis (RA), familial Mediterranean fever and chronic infections. It is characterized by the systemic deposit of fibrillar protein AA, with special repercussions at the renal level.1 This protein derives from a plasmatic precursor produced by hepatocytes as an acute phase reactant against persistent inflammatory stimuli or tissue necrosis.2 Its hepatic synthesis is regulated by various proinflammatory cytokines such as TNF-α, IL-1 and IL-6, the latter being related to the activity and severity of the disease.3
The basis of treatment lies in suppressing inflammation, thus inhibiting fibrillar protein synthesis. In recent years, the appearance of biological agents that specifically block cytokines has revolutionized the treatment of some chronic inflammatory diseases. In this line, the use of tocilizumab (TCZ) stands out, a humanized antibody that blocks the IL-6 receptor, widely used in RA and that could be particularly useful in AA amyloidosis.
Several case series have shown good efficacy for the treatment of this disease, even allowing the regression of renal deposits already present. Our objective is to analyze the efficacy and safety of TCZ in AA amyloidosis with renal involvement in our center.
Material and methodPatient selectionRetrospective monocentric study that included patients with a histological diagnosis of AA amyloidosis who received treatment with TCZ during the years 2018–2019. Clinical and demographic variables were recorded, including previous immunosuppressive treatment, tolerance, and adverse effects. Clinical follow-up was carried out from the diagnosis of amyloidosis until the end of the study (January 2020).
Clinical evaluationWe evaluated renal function using serum creatinine values (mg/dl), glomerular filtration rate calculated by the CKD-EPI formula (GFR; ml/min/1.73 m2) and protein/creatinine ratio (CPI; mg/g) at the 3, 6 and 12 months of follow-up. We define renal response as a >30% reduction in proteinuria and/or stabilization or improvement in glomerular filtration rate. The activity of amyloidosis and its response to treatment have been evaluated by serum amyloid protein (PSA; mg/l) and C-reactive protein (CRP; mg/l). We consider an anti-inflammatory response to be a decrease >50% in SAA protein and/or CRP levels.
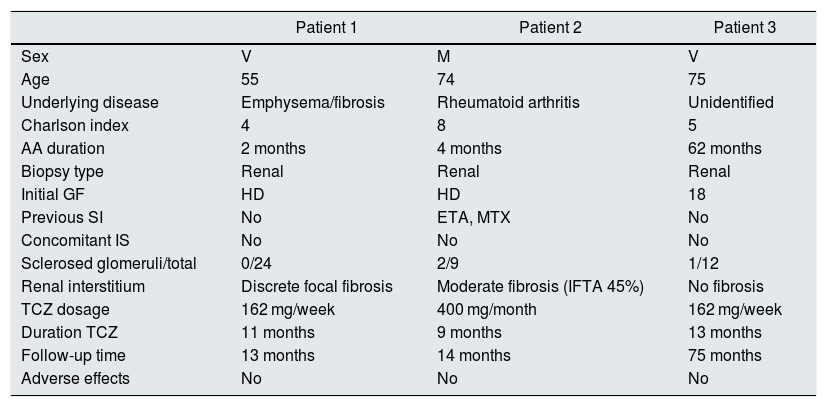
ResultsCharacteristics of the patientsWe identified 3 patients receiving treatment with TCZ after diagnosis of AA renal amyloidosis by renal biopsy. The baseline clinical characteristics of the subjects are shown in Table 1. The 3 cases presented renal involvement at the diagnosis of amyloidosis, with the renal function parameters being collected in Table 2. There have been no adverse events attributable to the use from TCZ.
Clinical and demographic characteristics of the patients included in the study.
| Patient 1 | Patient 2 | Patient 3 | |
|---|---|---|---|
| Sex | V | M | V |
| Age | 55 | 74 | 75 |
| Underlying disease | Emphysema/fibrosis | Rheumatoid arthritis | Unidentified |
| Charlson index | 4 | 8 | 5 |
| AA duration | 2 months | 4 months | 62 months |
| Biopsy type | Renal | Renal | Renal |
| Initial GF | HD | HD | 18 |
| Previous SI | No | ETA, MTX | No |
| Concomitant IS | No | No | No |
| Sclerosed glomeruli/total | 0/24 | 2/9 | 1/12 |
| Renal interstitium | Discrete focal fibrosis | Moderate fibrosis (IFTA 45%) | No fibrosis |
| TCZ dosage | 162 mg/week | 400 mg/month | 162 mg/week |
| Duration TCZ | 11 months | 9 months | 13 months |
| Follow-up time | 13 months | 14 months | 75 months |
| Adverse effects | No | No | No |
AA duration: time from diagnosis of amyloidosis to initiation of TCZ; ETA: etanercept; HD: hemodialysis; IS: immunosuppressive treatment; M: woman; MTX: methotrexate; TCZ: tocilizumab; V: male.
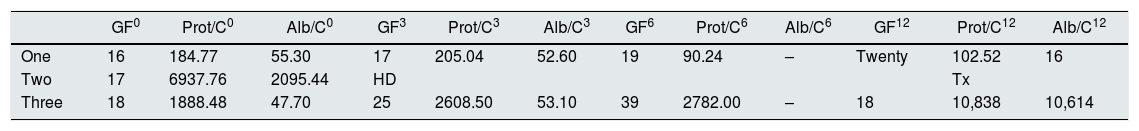
Initial and final evaluation of renal function.
| GF0 | Prot/C0 | Alb/C0 | GF3 | Prot/C3 | Alb/C3 | GF6 | Prot/C6 | Alb/C6 | GF12 | Prot/C12 | Alb/C12 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| One | 16 | 184.77 | 55.30 | 17 | 205.04 | 52.60 | 19 | 90.24 | – | Twenty | 102.52 | 16 |
| Two | 17 | 6937.76 | 2095.44 | HD | Tx | |||||||
| Three | 18 | 1888.48 | 47.70 | 25 | 2608.50 | 53.10 | 39 | 2782.00 | – | 18 | 10,838 | 10,614 |
Alb/C: albumin/creatinine ratio (mg/g); GFR: glomerular filtration rate (CKD-EPI; ml/min); HD: hemodialysis; Prot/C: protein/creatinine ratio (mg/g); Tx: kidney transplant.
Values: 0: prior to TCZ; 3: third month after TCZ; 6: sixth month after TCZ; 12: twelfth month after TCZ.
AA: secondary amyloidosis; RA: rheumatoid arthritis; TCZ: tocilizumab; GFR: glomerular filtration calculated by CKD-EPI; CPI: urine protein/creatinine ratio; IAC: urine albumin/creatinine ratio; PSA: amyloid serum protein; CRP: C-reactive protein; HD: hemodialysis.
A 55-year-old man who presented with postcontrast acute kidney injury requiring hemodialysis (HD) in the context of massive pulmonary thromboembolism. Corticosteroid boluses were administered without renal recovery, so a renal biopsy was finally performed. This biopsy showed glomeruli with minimal widening due to increased cellularity, without identifying inflammatory infiltrate, mesangial deposits or fibrinoid necrosis. The tubules presented focal edema; the interstitium, discrete focal fibrosis with mild nonspecific inflammatory infiltrate. A slight increase in the size of the vascular wall was observed at the expense of the deposit of eosinophilic material with birefringence, compatible with amyloid. The immunofluorescence study was negative.
His personal history included a smoking habit of 20 cigarettes a day and a combined emphysema/pulmonary fibrosis syndrome, diagnosed 2 months before the biopsy and considered an underlying cause of AA amyloidosis.
In this case, the subcutaneous presentation of TCZ was chosen, starting at a dose of 162 mg per week as described in the drug’s data sheet. Proteinuria levels decreased by 44.5% at 12 months, with a slight increase in GFR from 16 to 20 ml/min; at the inflammatory level, a reduction in PSA and CRP was observed, without evidence of systemic clinical manifestations at any time. Given these results, and until the writing of the article, the same dose of TCZ was maintained (11 months of treatment).
Case 2A 74-year-old woman diagnosed with arterial hypertension treated with losartan and diabetes mellitus treated with linagliptin, both of which were well controlled. As background, rheumatoid arthritis of 4 years’ evolution stands out, for which he had undergone treatment with etanercept and methotrexate with adequate symptomatic control. The subsequent diagnosis of Binet stage A chronic lymphatic leukemia forced the suspension of etanercept, maintaining methotrexate as monotherapy; after this, he presented several episodes of arthritis requiring corticotherapy. Ten months after the diagnosis of leukemia, given the progressive deterioration of renal function, a renal biopsy was performed. Histological findings showed a membranoproliferative pattern with increased mesangial substance in glomeruli, images of basement membrane duplication and nodularity with a tendency to sclerosis. There was tubular atrophy, interstitial fibrosis (35%–45%), discrete nonspecific inflammatory infiltrate, and intimal hyperplasia with arteriolohyalinosis and reduplication of elastic bands. Amyloid material was identified in vessels and interstitium. The immunofluorescence study showed C3++ and IgM+ deposits.
Being on HD, intravenous TCZ was administered. The dose, adjusted for body weight (8 mg/kg/every 4 weeks), was maintained until renal transplantation (9 months of treatment). The patient remained on HD, performing only urine sediments at 5 and 7 months (proteins 200 mg/dl). Although treatment with TCZ did not achieve recovery of kidney function, it allowed an adequate anti-inflammatory response with a reduction in PSA and CRP. In addition, during the follow-up period, the patient did not present inflammatory data suggestive of activation of her rheumatological disease.
Case 3A 75-year-old man with arterial hypertension treated with amlodipine and subclinical hypothyroidism. He was diagnosed with AA amyloidosis after an episode of nephrotic syndrome, with no underlying disease recorded. Renal biopsy showed glomeruli with slight acellular mesangial enlargements at the expense of amorphous, eosinophilic material with birefringence, compatible with amyloid. Isolated hyaline cylinders were found in tubular lumens. In the interstitium and arterial walls, there were interstitial acellular widenings by material similar to that previously described.
Treatment with TCZ was started subcutaneously at 162 mg/week. The GFR progressively improved until reaching 39 ml/min at 6 months, so it was decided to reduce the dose of TCZ to 162 mg subcutaneously fortnightly. Seven months later, he presented a sudden drop in GFR in the context of nephrotic syndrome, and a new dose increase to 162 mg/week was agreed upon. Until the writing of the article, he maintained stable renal function parameters (13 months of treatment). At the inflammatory level, a decrease in PSA and CRP values was observed, although with a slight rebound in PSA, coinciding with the aforementioned deterioration in renal function. He has not presented systemic clinical manifestations prior to the start of treatment with TCZ or during follow-up.
DiscussionIL-6 is a proinflammatory cytokine with an important role in the regulation of immune activity, participating in most inflammatory responses.5 One of the mechanisms through which this cytokine acts is the stimulation of hepatic synthesis of SAA protein; hence, it is considered a key element in the development of amyloidosis.
TCZ is a humanized antibody that blocks the IL-6 receptor, thus inhibiting this mechanism of action. It is considered an effective specific treatment in the treatment of chronic inflammatory diseases such as RA, although few data have been published on its use in AA amyloidosis.
We report a series of 3 cases of patients with AA renal amyloidosis treated with TCZ. In patients 1 and 3, a slight increase in GFR was observed after the start of TCZ, although they maintained proteinuria values similar to those detected prior to treatment. Patient 2 did not show improvement in renal function parameters, although she preserved residual diuresis until the time of transplantation and maintained adequate control of RA with the use of TCZ.
The renal biopsy of the latter patient showed other renal lesions in addition to the amyloid deposit, such as a pattern of membranoproliferative damage, a poor prognostic finding in histology. This finding, added to the degree of chronicity, could justify the lack of improvement in GFR. Probably, earlier initiation of TCZ would have allowed better results at the renal level.
We have defined renal response as a >30% reduction in proteinuria and/or stabilization or improvement in GFR.4 In our series, the GFR remained stable in patients 1 and 3, obtaining a renal response to the effects of proteinuria in the first. However, proteinuria in this patient was not too high at the start of treatment, so the results could be limited.
The variability of the baseline characteristics of the patients, in terms of inflammatory disease, renal stage and histological data, could influence the response to treatment with TCZ. It would be interesting to carry out new studies that analyze the possible prognostic factors of renal evolution.
We define anti-inflammatory response as a >50% decrease in SAA protein and/or CRP levels. All patients experience a rapid decrease in acute phase reactants after the start of TCZ, which would translate into a reduction in the inflammatory state of these subjects. In this sense, we could conclude that the anti-inflammatory response to TCZ treatment in AA amyloidosis is 100% in our series. Lane et al. demonstrated that TCZ can be effective in reducing inflammation by reducing SAA protein levels in patients with chronic inflammatory diseases and amyloidosis.5 Uda and Saiki suggest that the use of this drug not only lowers CRP and SAA levels, but also delays the start of HD in patients with amyloidosis and advanced chronic kidney disease.6
Both the efficacy and safety of TCZ in patients with kidney disease have been reported in different publications, including even HD patients.7,8 In the study by Mori et al.9 no differences were found in terms of safety in the group of patients with chronic kidney disease compared to those without this complication, showing significant improvements in clinical activity indices in all groups. In our series, TCZ has been administered with good tolerance and no adverse effects have been recorded during follow-up.
Therefore, the use of TCZ in patients with AA renal amyloidosis seems to be effective and safe, even in situations of progressive kidney damage. Its administration has been shown to be useful in stabilizing or improving GFR and reducing inflammatory markers, so its indication seems reasonable in patients who do not respond to other treatments or even be considered as the first therapeutic option, in the absence of randomized prospective studies.
Our study contains some limitations. This is a monocentric review, with a small number of subjects, which makes it difficult to obtain significant results, as well as their extrapolation to the population. The retrospective nature of the study, although practically unavoidable as it is a rare disease, limits the proper recording of the initially proposed variables. New, prospective, randomized studies with long-term clinical assessment would be necessary.
ConclusionsIL-6 plays an important role in the pathophysiology of AA amyloidosis, so TCZ could represent a therapeutic option to consider in these patients. Our results show its efficacy both at the renal level and in the control of the underlying disease, especially in patients with a high inflammatory component in whom it starts early. The absence of adverse effects associated with TCZ in our series, as in previous studies, supports the safety of this drug in patients with kidney disease. TCZ should be considered within the therapeutic arsenal for the treatment of AA renal amyloidosis, as it may improve the long-term prognosis of these patients.
FinancingThis research has not received specific aid from public sector agencies, the commercial sector or non-profit entities.
Conflict of interestsThe authors declare that they have no conflict of interest.
Please cite this article as: Almenara-Tejederas M, Alonso-García F, Aguillera-Morales WA, de la Prada-Álvares F, Salgueira-Lazo M. La inhibición de la interleucina-6 como posible diana terapéutica en la amiloidosis AA. Nefrologia. 2022;42:28–32.