OSERCE es un estudio multicéntrico y transversal cuyo objetivo es conocer las características bioquímicas, clínicas y el manejo de las alteraciones del metabolismo óseo-mineral, así como el grado de cumplimiento de las guías K/DOQI en pacientes con enfermedad renal crónica (ERC) no en diálisis. Se incluyeron 634 pacientes procedentes de 32 Unidades de Nefrología de España con ERC no en diálisis y filtrado glomerular < 60 ml/min/1,73 m2 (estadios K/DOQI: 33 % estadio 3 , 46 % estadio 4 y 21 % estadio 5 no en diálisis). En 409 pacientes se realizó además analítica en un laboratorio centralizado determinando creatinina, calcio, fósforo, hormona paratiroidea (PTH) intacta, 25 OH vitamina D y 1,25 OH2 vitamina D). El porcentaje de incumplimiento en los objetivos establecidos por las guías K/DOQI para niveles de calcio, fósforo, PTH intacta y producto calcio x fósforo fue 45 %, 22 %, 70 % y 4 % respectivamente. El 70% de los pacientes presentaban los niveles de PTH intacta fuera del rango establecido por las guías K/DOQI (55,5 % con valores por encima del límite superior y 14,5 % por debajo del límite inferior). El 45 % de los pacientes presentaban niveles de calcio fuera de rango (40 % por encima y 5 % por debajo del rango), mientras que el 22 % presentaron niveles inadecuados de fósforo (3 % por debajo del objetivo y 19 % por encima). El 4 % de los pacientes presentaron niveles de producto calcio x fósforo fuera de rango. Solo el 1,8 % de los pacientes cumplieron los cuatro objetivos K/DOQI. Los valores detectados en la analítica centralizada no mostraron diferencia respecto a los de la analítica general. El 81,5 % de los pacientes presentaba deficiencia de calcidiol (25 OH D3) (< 30 ng/ml). El 35 % presentaron insuficiencia moderada-grave (< 15 ng/ml), y el 47 % insuficiencia leve (15-30 ng/ml). El 64,7 % presentaron insuficiencia de calcitriol (1,25 OH2 D3 < 22 pg/ml). Mientras que el déficit de calcidiol no se relacionó con los estadios de ERC, la deficiencia de calcitriol fue más pronunciada conforme avanzó el estadio de ERC. Los resultados del estudio OSERCE confirman la dificultad de conseguir los niveles objetivo establecidos por las guías K/DOQI en la ERC no en diálisis, fundamentalmente en el pobre control del hiperparatiroidismo secundario y del déficit de vitamina D. Por ello es necesario revisar las estrategias de abordaje del tratamiento de las alteraciones del metabolismo óseo-mineral en estos pacientes, y quizá una revisión de los parámetros objetivo de las guías actuales.
OSERCE is a multi-centre and cross-sectional study with the aim of analysing the biochemical, clinical, and management characteristics of bone mineral metabolism alterations and the level of compliance with K/DOQI guideline recommendations in patients with chronic kidney disease (CKD) not on dialysis. The study included a total of 634 patients from 32 different Spanish nephrology units, all with CKD, estimated glomerular filtration rates <60ml/min/1.73m2, and not on dialysis (K/DOQI stage: 33% stage 3, 46% stage 4, and 21% stage 5). In 409 of these patients, laboratory parameters were also measured in a centralised laboratory, including creatinine, calcium, phosphorous, intact parathyroid hormone (PTH), 25-OH-vitamin D, and 1,25-OH2-Vitamin D levels. The rates of non-compliance with the K/DOQI objectives for calcium, phosphorous, intact PTH, and calcium x phosphate product among these patients were 45%, 22%, 70%, and 4%, respectively. Of the 70% of patients with intact PTH levels outside of the target range established by the K/DOQI guidelines, 55.5% had values above the upper limit and 14.5% had values below the lower limit. Of the 45% of patients with calcium levels outside of the target range, 40% had values above the upper limit and 5% had values below the lower limit. Of the 22% of patients with phosphorous levels outside of the target range, 19% had values above the upper limit, and 3% had values below the lower limit. Finally, 4% of patients also had values for the calcium x phosphate product that were outside of the recommended range. Only 1.8% of patients complied with all four K/DOQI objectives. The values detected in centralised laboratory analyses were not significantly different from those measured in the laboratories at each institution. In addition, 81.5% of patients had a deficiency of calcidiol (25-OH-D3) (<30ng/ml); of these, 35% had moderate-severe deficiency (<15ng/ml) and 47% had mild deficiency (15-30ng/ml). Calcitriol (1,25-OH2-D3) levels were deficient in 64.7% of patients. Whereas the calcidiol deficiency was not correlated with the CKD stage, calcitriol deficit were more pronounced at more advanced stages of CKD. The results of the OSERCE study confirm the difficulty in reaching the target values recommended by the K/DOQI guidelines in patients with CKD not on dialysis, in particular in the form of poor control of secondary hyperparathyroidism and vitamin D deficiency. With this in mind, we must review strategies for treating bone mineral metabolism alterations in these patients, and perhaps revise the target parameters set by current guidelines.
INTRODUCTION
Chronic kidney disease (CKD) constitutes a public health issue that is estimated to affect more than 10% of the global population, and the prevalence of which has increased in recent years.1,2 The most important complication of CKD is cardiovascular disease, which is the primary cause of death in these patients. This increase in cardiovascular morbidity/mortality associated with CKD has been described even in patients with no evidence of ischaemic heart disease,3 which helps explain the elevated mortality rate among patients in initial stages of CKD (20%, 24%, and 46% after 5 years for stages 2, 3, and 4, respectively), which far surpasses the rate for patients who finally require dialysis.4
Bone-mineral metabolism abnormalities, which the KDIGO5 guidelines recently defined as chronic kidney disease-mineral and bone disorder (CKD-MBD), have been clearly implicated not only in the development of secondary hyperparathyroidism (SHPT) and renal osteodystrophy, but have also been associated with the progression of CKD and its complications, including cardiovascular complications,6 and they ultimately contribute significantly to an increase in morbidity and mortality rates among patients with CKD.7,8
These alterations constitute a systemic disorder that is characterised by abnormal calcium, phosphorous, parathyroid hormone (PTH), and vitamin D metabolism, which, in addition to affecting the skeletal system, is related to the appearance of cardiovascular and soft tissue calcifications5 that in turn are associated with cardiovascular pathologies in patients with CKD.9-12
With the objective of improving the management of CKD and controlling abnormal bone mineral metabolism and its complications, the National Kidney Foundation published the K/DOQI guidelines for clinical practice in 2003,13 which for many years have been used as a universal reference, even though they were fundamentally based on expert opinion. Since then, several studies have been carried out to evaluate the level of implementation of the K/DOQI guidelines in clinical practice, which have revealed the difficulties in reaching the primary objectives for biochemical parameters (calcium, phosphorous, calcium x phosphorous product, and intact PTH [iPTH]), and the recommended frequency with which these should be determined. The vast majority of these studies have been carried out among patients on dialysis,14-20 and very few reports have been published regarding patients with CKD before starting dialysis.21-24 Although these guidelines were published 10 years ago, and new international guidelines such as the KDIGO guidelines of 200925 and national guidelines such as that produced by the Spanish Society of Nephrology (S.E.N.)26,27 have since been published, the K/DOQI guidelines still appear to be relevant, since many of the recommendations made therein are practically identical to the current Spanish guidelines.27 Even more importantly, in a recent European publication, the best survival values reported for patients on dialysis were associated with treatment based on the values recommended by the K/DOQI guidelines.28
Obviously, bone mineral metabolism abnormalities start during the first stages of CKD, long before the need for renal replacement therapy; these processes develop as renal function decreases, and can be positively or negatively influenced by the treatment strategy employed. As such, it is recommended that attending physicians monitor and control biochemical parameters early in the development of CKD,3-5 before the need for dialysis.25,27,29
Given the necessity of an early diagnosis and intervention upon CKD in order to prevent cardiovascular complications and mortality associated with bone mineral metabolism, taking into account the lack of data regarding the implementation of the K/DOQI guidelines in CKD stages prior to renal failure,21-24 we embarked on the OSERCE study (acronym for epidemiology of bone disease in ambulatory chronic kidney disease in Spanish). The objective of this study was to evaluate the level of compliance with the K/DOQI guidelines and the clinical and metabolic situation of patients with CKD who have not started renal replacement therapy, using for the first time a centralised analysis system in order to avoid the well-known variability in measurements of mineral metabolism parameters.30,31
The OSERCE study was carried out in two simultaneous phases. In the first phase (OSERCE questionnaire), a survey was administered among researchers to evaluate the level of awareness of the K/DOQI guidelines for the diagnosis and followup of patients with CKD in stages prior to dialysis. The results of this study have already been published.32 In the second phase of the study (OSERCE I study), data were compiled on patients with CKD who had not started dialysis in order to evaluate the level of compliance with the guideline recommendations. Here, we present the results from this second phase, the OSERCE I study, which will also serve as a precursor to the study currently being carried out to evaluate disease evolution and late complications in these patients (OSERCE II).
MATERIAL AND METHOD
Study design and population
The OSERCE study is an observational, cross-sectional study involving the participation of 32 hospitals in Spain. The patients were included on a consecutive basis from nephrological consultations between April and May 2006. The study population included patients older than 18 years of age, with CKD and a glomerular filtration rate <60ml/min/1.73m2, who had not started renal replacement therapy. Thus, we included patients in stage 3 (30-59ml/min/1.73m2), stage 4 (15-29ml/min/1.73m2), and stage 5 (<15ml/min/1.73m2) CKD based on the K-DOQI classification system (NKF 2003), who were not on dialysis. We excluded all patients with acute renal failure, those who had previously received a kidney transplant, those with severe disease unlikely to be followed-up for at least 12 months, and those who had been hospitalised within the previous month. In accordance with the Helsinki Declaration, patients were required to provide informed consent. We also received the approval of the hospital research ethics committee.
The primary objective of our study was to determine the level of compliance with K/DOQI guidelines for controlling PTH levels. As stated, these guidelines are essentially similar to the 2011 S.E.N. guidelines.27 Secondary objectives included assessing other biochemical markers for CKD mineral and bone metabolism disorder (CKD-MBD) (calcium, phosphorous, calcium x phosphorous product, 25-OH vitamin D, and 1,25 [OH]2 vitamin D), analysing the level of compliance with K-DOQI guidelines, describing treatments administered to the study patients (vitamin D, phosphate binders, bicarbonate), and evaluating whether comorbidities (aetiology of CKD, diabetes, hypertension, anaemia, nutritional state, renal function, and DOQI state) are associated with the results obtained in CKD-MBD.
Evaluations
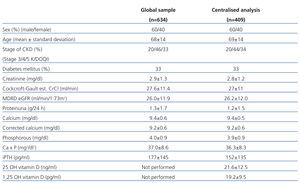
Data were compiled using a specific online folder for this express purpose. We registered data regarding patient demographic and clinical variables (sex, age, aetiology of CKD, comorbidity, and treatment received during the last six months for CKD). Laboratory parameters were evaluated at each centre and again at a centralised location, the Dr Echevarne Analytical Laboratory in Barcelona. Results are presented in two tables, such that one column reflects the results from laboratory analyses at each centre (n=634) and the other column reflects the results from the centralised laboratory analysis (n=409). Of the 634 patients included from 32 different hospitals, 409 samples were sent from 21 centres for the centralised analysis (64.5%).
The central laboratory utilised the standard methodology for determining plasma levels of total calcium, phosphorous, total proteins, albumin, and creatinine. Creatinine was measured under the reference method of isotopic dilution mass spectrometry. Given that the active form of calcium is ionised, total calcium measured in plasma samples was corrected using the Orrell formula: Corrected calcium (mg/dl) = Measured calcium (mg/dl) + 0.0176 (34 – serum albumin [g/l]).33 iPTH was measured using chemiluminescence (Immulite® 2000). Calcitriol (1,25 [OH]2 vitamin D) was measured by radioimmunoassay (Diasorin®) and calcidiol (25 OH vitamin D) was measured by chemiluminescence (Diasorin®).
In order to analyse the prevalence of calcidiol deficits, we excluded 22 patients with nephrotic-range proteinuria (>3.5g/24h). These patients have reduced calcidiol concentrations due to an increased loss in the urine of vitamin-D-binding protein.34,35 For this analysis, we also excluded 51 patients due to the lack of a 24-hour urine sample, thus making it impossible to rule out nephrotic proteinuria. As such, a total of 336 patients were evaluated for the analysis of calcidiol deficits. We included all patients in the analysis of correlations between vitamin D and the various study parameters.
Statistical analysis
The primary study variable was the percentage of patients on CKD and with a glomerular filtration rate <60ml/min/1.73m2 not on dialysis that had iPTH levels outside of the range established by the K/DOQI guidelines (iPTH levels outside of the ranges of 3570pg/ml, 70-110pg/ml, and 150-300pg/ml for stages 3, 4, and 5, respectively).13 In addition, we calculated the percentage of patients who were tested for iPTH levels at the frequency recommended by the guidelines (every year for stage 3 patients, and every 3 months for stage 4 and stage 5 patients).13
In a similar manner, we described adherence to the cut-off values established by the K/DOQI guidelines for calcium, phosphorous, and calcium x phosphorous product. Target serum phosphorous levels were 2.7-4.6mg/dl for stage 3 and 4, and 3.5-5.5mg/dl for stage 5. For calcium levels, the K/DOQI guidelines only recommend a range of corrected calcium values of 8.4-9.5mg/dl for patients with stage 5 CKD, suggesting that calcaemia be maintained within the normal range for the reference values established by each individual laboratory for other stages. The calcium x phosphorous product at stages 3, 4, and 5 should be maintained below 55mg2/dl2.
Cut-off values for vitamin D were: <30ng/ml for calcidiol insufficiency and <15ng/ml for moderate calcidiol deficiency, as established by the K/DOQI guidelines, and <7ng/ml for severe deficiency. Since no recommendations exist for calcitriol deficiency, this was defined as values <22pg/ml, based on previous publications.22,36
Keeping in mind the results published from a cohort of patients with stage 3 or 4 CKD,37 we estimated that the percentage of patients who comply with K/DOQI guidelines for iPTH levels would be approximately 25%. As such, in order to detect a 25% proportion, with a precision of 0.0375 and an alpha error of 0.05, and taking into account a 15% rate of patient loss, we estimated that our study sample would need to include 600 patients.
We described our sample using percentages for categorical variables and mean (standard deviation) for continuous variables. The primary variable was presented as the prevalence of patients with values that fell outside of the range recommended by the K/DOQI guidelines, for the overall analysis and by CKD stage, with a 95% confidence interval. We compared between subgroups using chi-square tests for categorical variables and Student’s t-tests or Wilcoxon tests, as appropriate, for continuous variables. We also determined the coefficient of determination between the different biochemical parameters. We considered a Pvalue <.05 to be statistically significant in all cases. We also applied a Pearson’s correlation analysis and multivariate logistic regression.
All analyses were carried out using SPSS statistical software for Windows version 13.0 and “R” statistical software version 2 (Language and Environment for Statistical Computing and AlcEst).
RESULTS
Patient characteristics
Our study included a total of 634 patients with CKD and glomerular filtration rates <60ml/min/1.73m2 and who had not started treatment with dialysis. Samples were sent for the centralised analysis for 409 patients (64.5%).
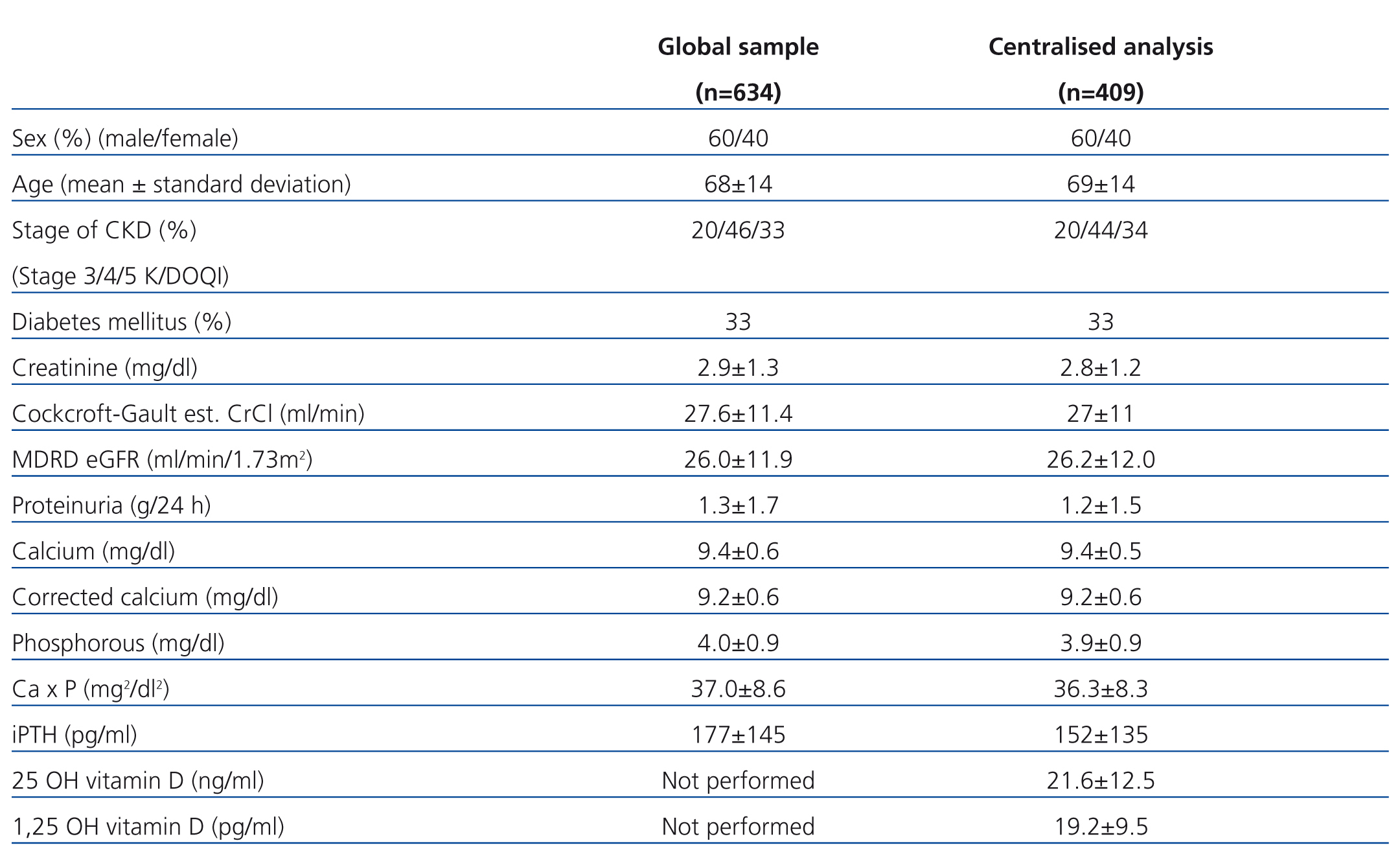
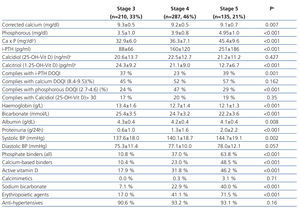
Table 1 displays the demographic, clinical, and biochemical characteristics for the patients in the global sample (n=634) and in the centralised laboratory analysis (n=409) (n=336 for the analysis of calcidiol deficits). Mean patient age in the overall sample was similar in both groups. Approximately half of all patients had stage 4 CKD, and 33% of all patients were diabetic. Biochemical parameters were similar between groups, with no statistically significant difference between parameters in the global sample and in the centralised laboratory analysis. The correlation between the two measurements was significant, with a Pearson’s correlation coefficient of 0.59 for calcium, 0.71 for phosphorous, and 0.74 for iPTH.
Biochemical parameters according to stage of chronic kidney disease
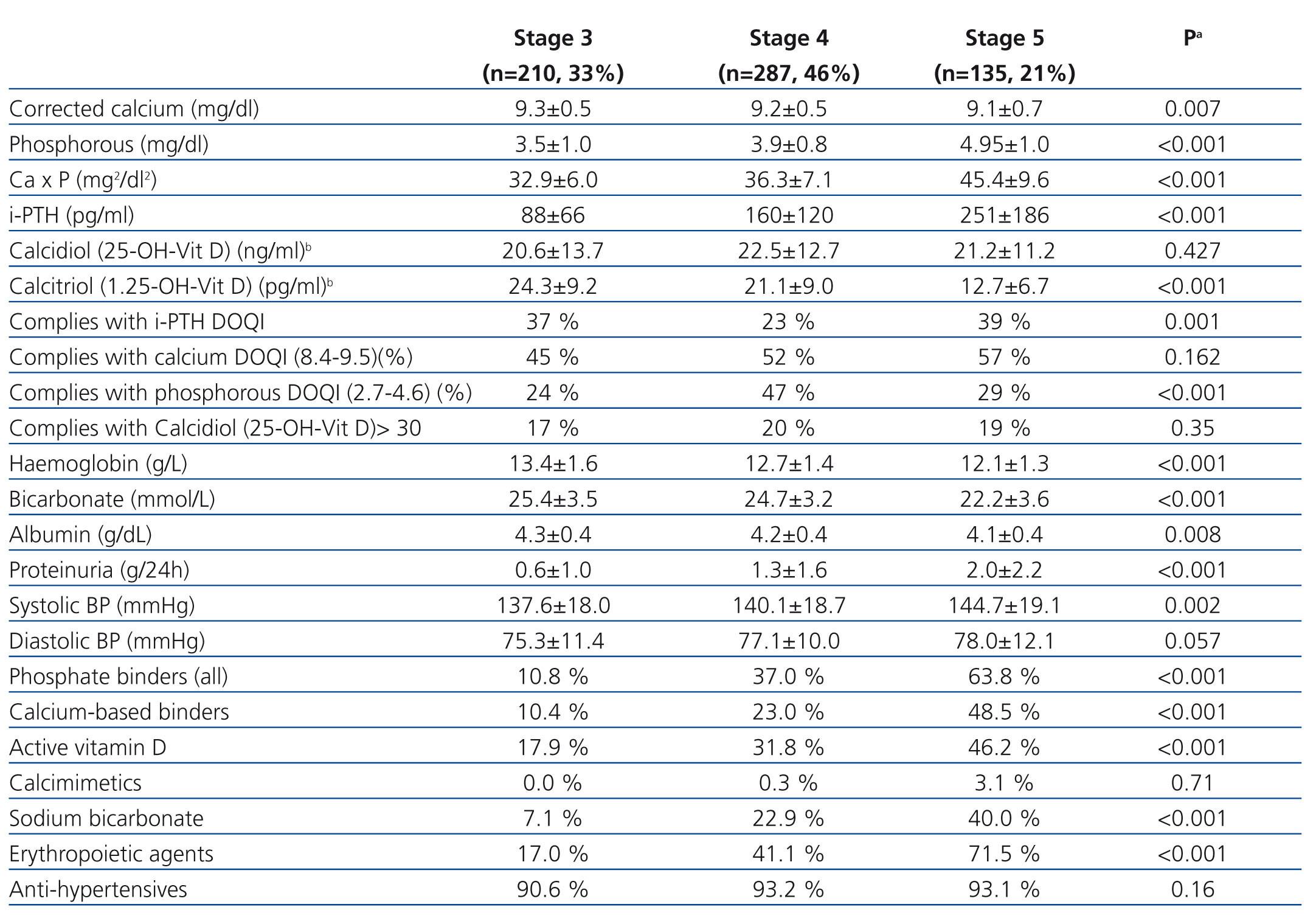
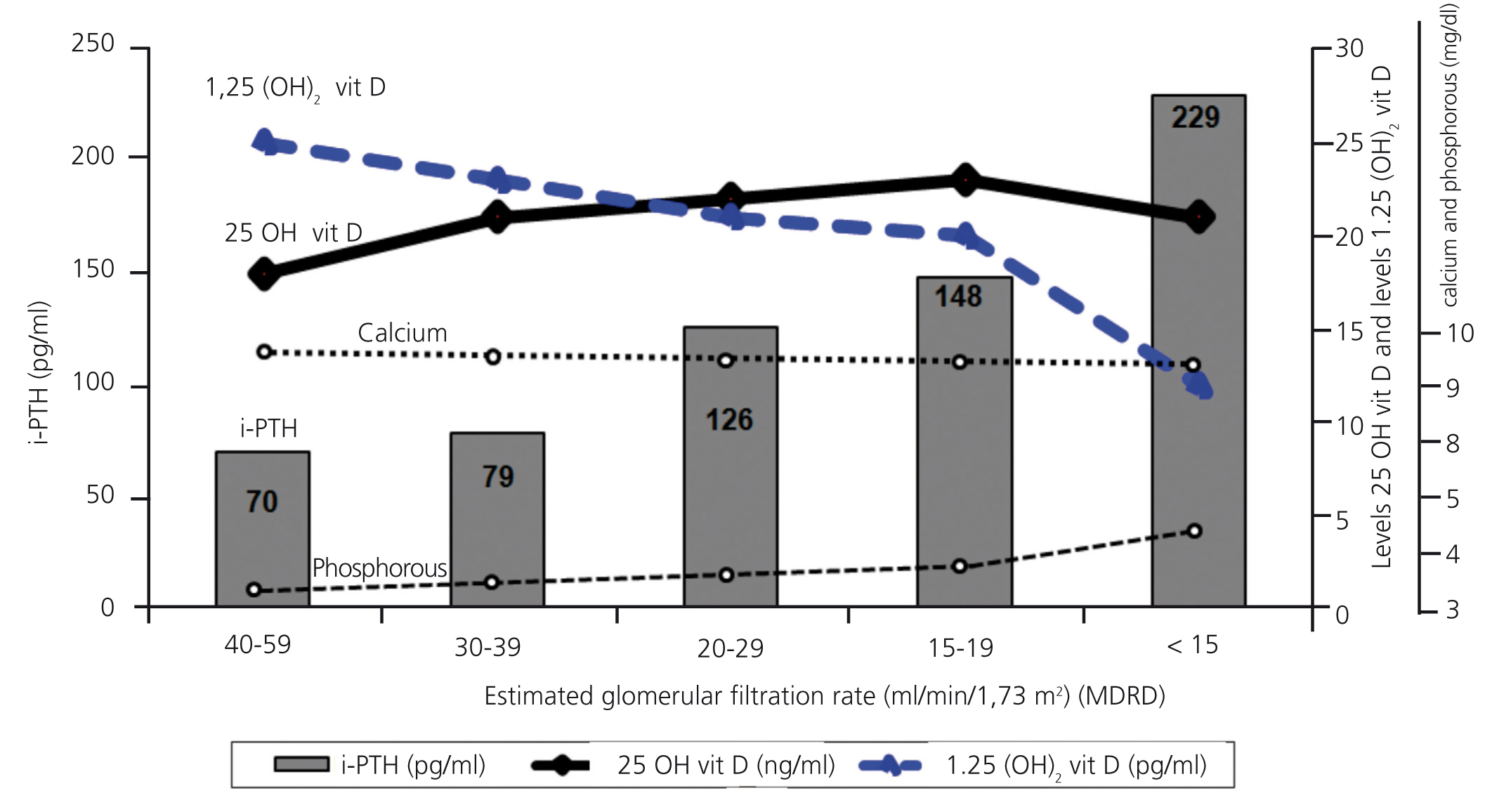
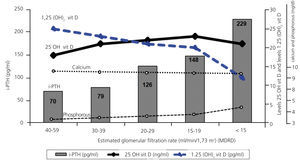
Table 2 displays mean levels for the different laboratory and clinical parameters measured, as well as treatments administered based on CKD stage. As the severity of renal failure increased, we observed significantly lower calcium, calcitriol, haemoglobin, albumin, and bicarbonate levels, and greater hyperparathyroidism, arterial hypertension, proteinuria, phosphataemia, and calcium x phosphorous product values. The decreases in calcidiol levels were constant, regardless of the stage of CKD, whereas calcitriol deficit increased with decreasing glomerular filtration rates. Figure 1 displays the progression of vitamin D, iPTH, calcium, and phosphorous levels based on renal function in the centralised analysis (n=405). The decrease in glomerular filtration rates was associated with a decrease in calcitriol levels (correlation coefficient: r=0.456; P<.001) and an increase in PTH levels (r=-0.429; P<.001), whereas there was no such a correlation between calcidiol levels and renal function (r=-0.018; P=.778).
Patient treatment
Table 2 also shows the treatments provided to study patients. As renal function decreased, the percentage of patients receiving phosphate binders, vitamin D, bicarbonate, and erythropoiesis-stimulating agents increased (P<.001). A total of 34% of patients received treatment with phosphate binders (57.5% calcium carbonate, 27.6% calcium acetate, 14% sevelamer hydrochloride, and 4.7% aluminium hydroxide). Approximately 29% received active vitamin D (calcitriol in 98%, alfacalcidol in 2%, and no cases of paricalcitol), and 0.8% were treated with calcimimetics. Calcium was administered as the calcium-based binder in 37% of patients, and 10.6% of patients simultaneously received calcium-based binders and calcitriol.
Level of compliance with K/DOQI guidelines
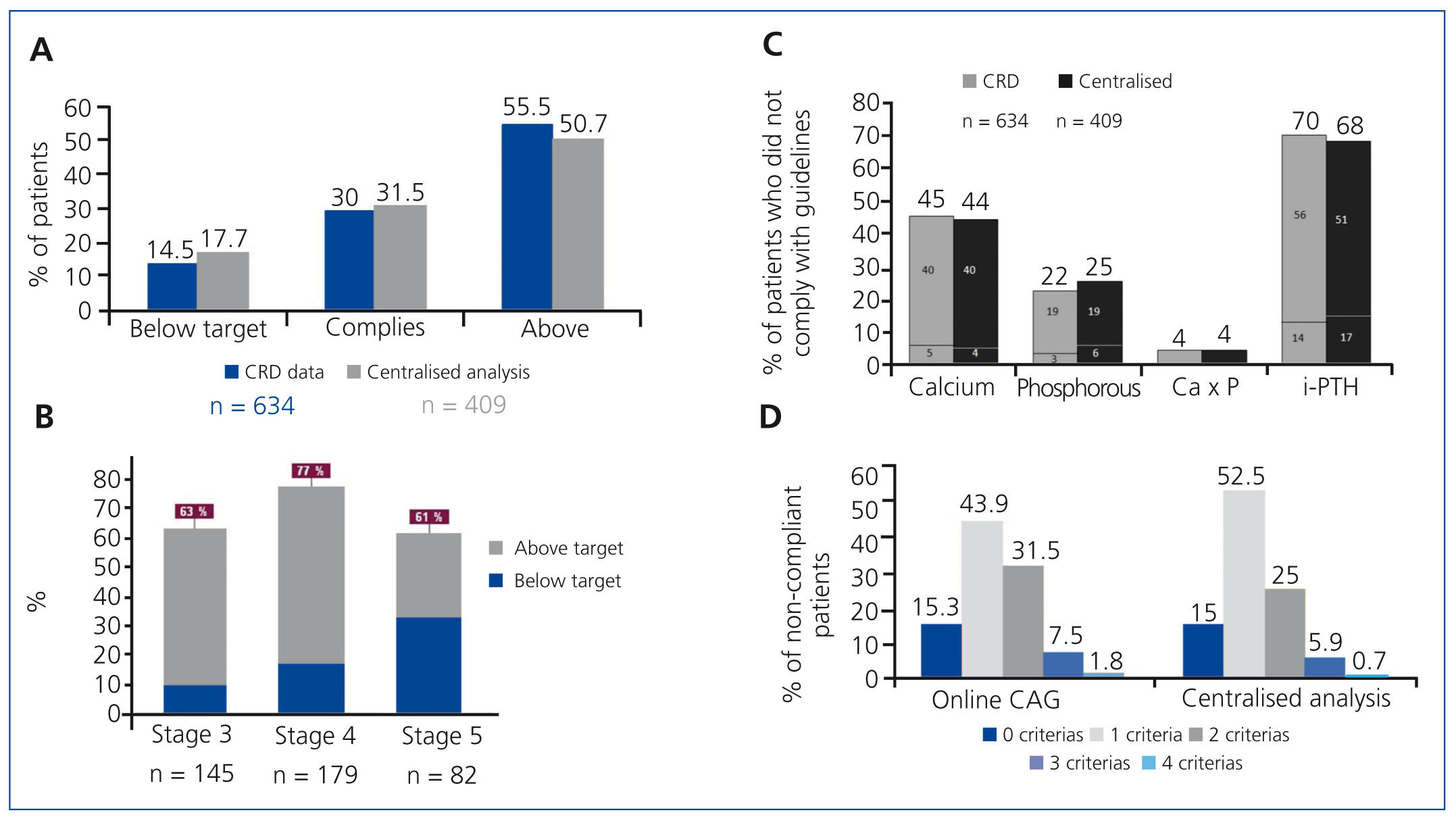
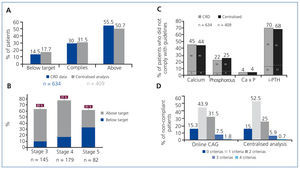
In both the overall sample and in the centralised analysis, approximately 70% of patients had iPTH values outside of the range established by K/DOQI guidelines, more than half of which had values above the upper limit (Figure 2A). A significant, although lower, percentage of patients had PTH values below the lower limit. The level of non-compliance with iPTH levels was similar between different stages of CKD (63%, 77%, and 61% for stages 3, 4, and 5, respectively; Figure 2B), and it was more common for values to above the recommended range than to fall below recommended levels. The percentages of patients with calcium, phosphorous, and calcium x phosphorous levels outside of the range established by the K/DOQI guidelines are shown in Figure 2C, both for the overall sample and for the sample analysed at the centralised laboratory. Values did not comply with guideline recommendations in more than one-third of all patients for calcium, and in less than one-fifth of all patients for phosphorous. The calcium x phosphorous product did not present a problem for compliance in these patients with CKD. Globally, the majority of patients (75%-77%) did not comply with 2 or 3 K/DOQI criteria, whereas only 15% of patients were in compliance with all K/DOQI target levels (Figure 2D).
Factors associated with non-compliance with K/DOQI guidelines
None of the variables analysed (age, sex, diabetes, time of follow-up, aetiology, treatment, associated comorbidity, or values for laboratory parameters) were significantly associated with non-compliance with K/DOQI target values. As in other studies, the multivariate logistic regression analysis demonstrated that treatment with binders (Odds Ratio [OR]: 2.39; 95% confidence interval [95% CI]: 1.15-4.97; P<.05), phosphorous levels (OR: 1.77; 95% CI: 1.22-1.55; P<.01), calcidiol levels (OR: 0.35; 95% CI: 0.170.71; P<.01), and calcium levels (OR: 0.41; 95% CI: 0.21-0.83; P<.05) were the only factors independently associated with iPTH levels above 300pg/ml. In this model, in addition to the aforementioned variables, we took into account age, sex, diabetes and other associated diseases, aetiology of CKD, time of follow-up, treatment, and calcitriol levels. Applying the same model, phosphorous levels (OR: 0.70; 95% CI: 0.51-0.95; P=.023) and PTH (OR: 0.50; 95% CI: 0.35-0.72; P<.001) were associated with calcium levels greater than 9.5mg/dl, whereas PTH levels (OR: 1.9; 95% CI: 1.22-3.05; P=.005), calcitriol (OR: 0.13; 95% CI: 0.07-0.24; P<.001), and the use of binders (OR: 0.44; 95% CI: 0.22-0.87; P=.018) were associated with phosphorous levels above 4.6mg/dl.
Percentage of patients in which intact parathyroid hormone was measured as a standard practice
Upon evaluating the last laboratory analyses provided for each patient, we observed that in 36% of cases, PTH values were not measured at this last follow-up session, having passed a mean 4.5±3.6 months since the second-to-last visit. An analysis of the data from the previous year revealed that 18% of patients (all of which had been monitored for at least 6 months) had not undergone an analysis of PTH in this time period.
Vitamin D deficit
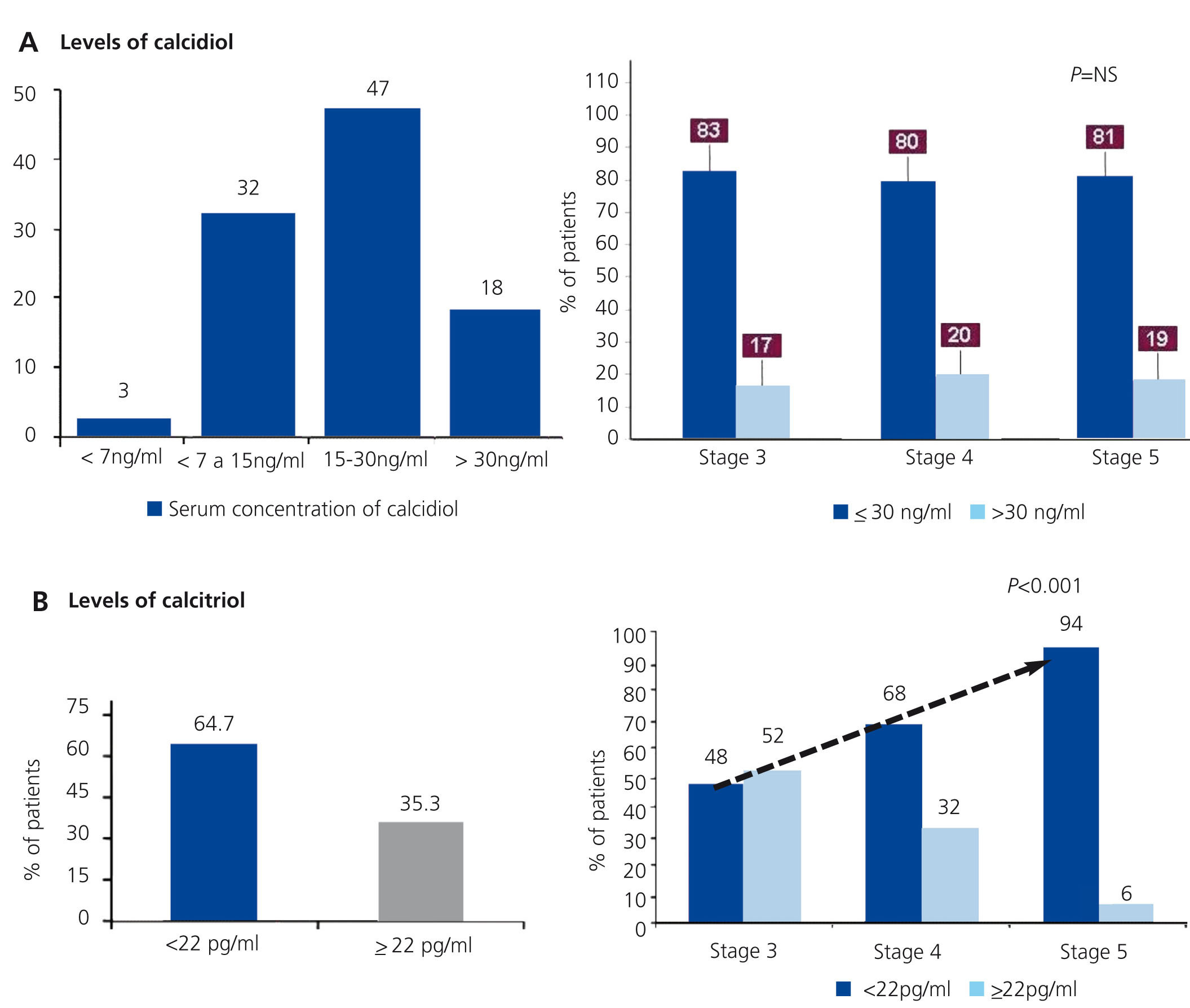
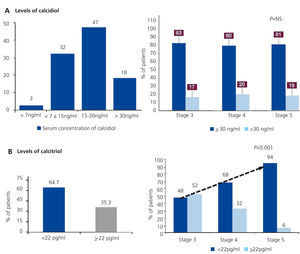
Of the 336 samples analysed in the central laboratory, 82% had inadequate calcidiol levels (Figure 3A). These patients were further sub-divided into 47% with insufficiency (15-30ng/ml), 32% with moderate deficiency (7-15ng/ml), and 3% with severe deficiency (<7ng/ml). The proportion of patients with inadequate values of calcidiol did not differ significantly between patients treated with active vitamin D and those who did not receive this treatment, nor did it vary between the different stages of CKD (Figure 3A). Inadequate calcitriol levels (<22pg/ml) were observed in 64.7% of patients (Figure 3A and 3B). Curiously, we did not observe a significant relationship between latitude and geographical distribution among autonomous communities and vitamin D levels (data not shown). Mean calcidiol and calcitriol levels were highest in the Canary Islands, Murcia, Valencia, and Galicia, in decreasing order. The lowest values were detected in Extremadura, followed by Castilla-La Mancha, Cantabria, and Basque Country.
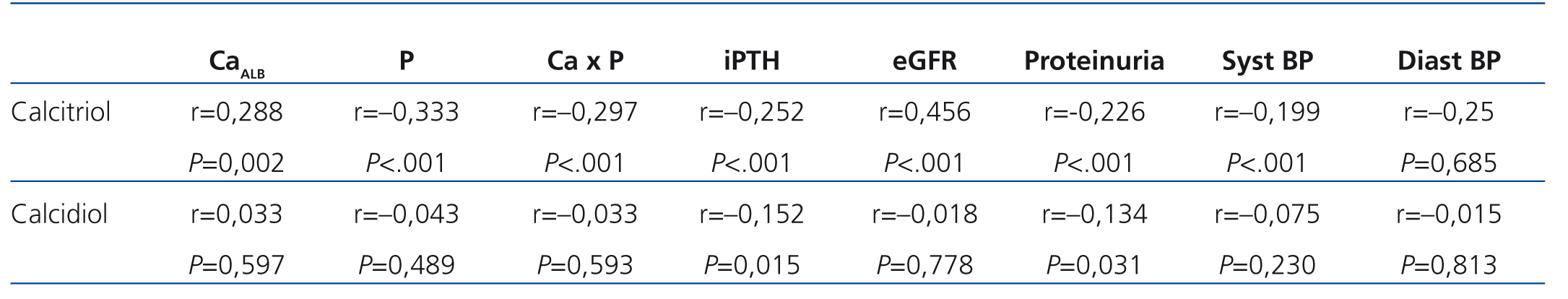
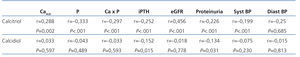
As was expected, as glomerular filtration rates decreased, the percentage of patients with calcitriol deficit increased significantly (Figure 3B). In addition to the association between calcitriol levels and glomerular filtration rate, we observed a weaker but still significant relationship with calcium, phosphorous, iPTH, proteinuria, and blood pressure (Table 3). These associations were not observed for calcidiol, except for in the case of iPTH values (r=-.0152; P=.015) and proteinuria (r=-0.134; P=.031). An analysis of these associations by linear regression confirmed that both correlations with calcidiol were weak but significant (R2=0.024, and R2=0.018 for iPTH and proteinuria levels, respectively).
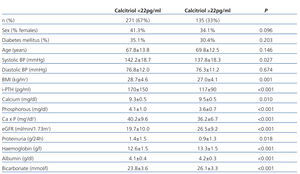
Differences between patients according to calcitriol levels
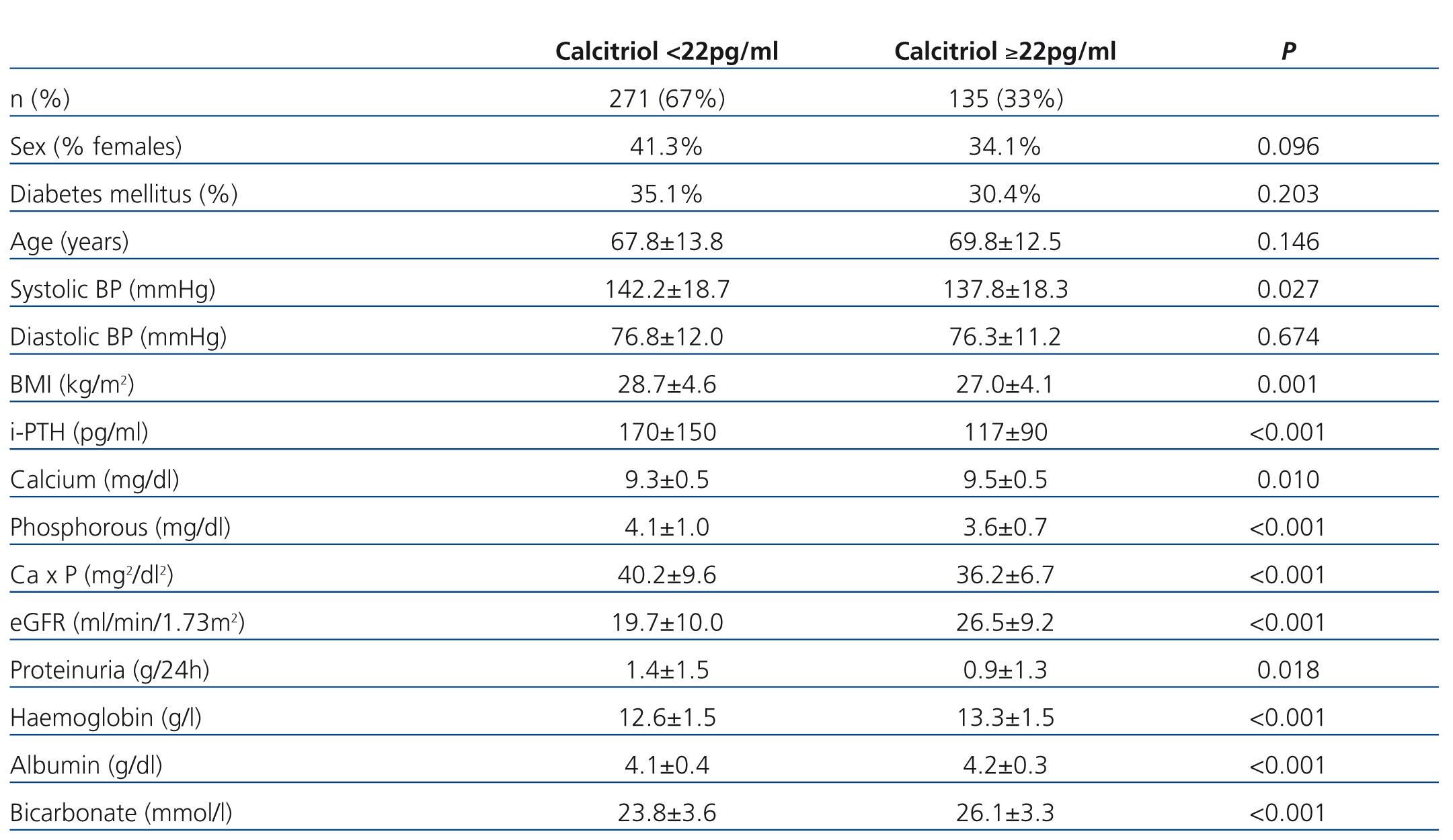
A univariate analysis among patients with normal and low levels of calcitriol demonstrated significant differences in all parameters of bone mineral metabolism, including calcidiol, glomerular filtration rate, proteinuria, systolic blood pressure, and nutritional parameters, including albumin and body mass index (Table 4). The multivariate logistic regression analysis demonstrated that only the decrease in calcidiol levels (OR=0.946; 95% CI: 0.922-0.971; P<.001) and the decrease in estimated glomerular filtration rate (OR=0.916; 95% CI: 0.879-0.954; P<.001) predicted low levels of calcitriol. The variables included in this model were sex, age, diabetes mellitus, calcium, phosphorous, PTH, calcidiol, glomerular filtration rate, and proteinuria. All laboratory parameters (including calcidiol levels) were introduced into the model as independent quantitative variables after testing them for linearity.
DISCUSSION
As yet, the OSERCE I study is the largest observational, multi-centre study with a centralised laboratory analysis and time limit for sample collections performed in Spain regarding the prevalence of bone mineral metabolism alterations in patients with various degrees of CKD not on dialysis. Our results provide a key insight for understanding and improving the management of these patients, especially since certain treatments were not available during the study, such as paricalcitol. Some of these results are in accordance with those from other studies, while others are novel. Our study alerts us to the situation of a high rate of noncompliance with K/DOQI guidelines at all stages of CKD, in which only 15% of patients complied with all four guideline criteria (PTH, calcium, phosphorous, and calcium x phosphorous product) before requiring renal replacement therapy.
In our study, 70% of patients had PTH levels that fell outside of the recommended range, and more than 50% were above the K/DOQI established upper limit. Non-compliance was greater than 60% for all three stages, with a slight tendency towards a greater degree of non-compliance in stage 4 patients (77%).
These results are in agreement with the percentages observed in previous studies including patients with CKD not on dialysis, which range from 62%-69%.21,24 In this manner, Craver et al.21 reported a heterogeneous rate of non-compliance of 57.6%, 75.4%, and 53.2% in stages 3, 4, and 5, respectively. This was the largest study performed in this context until now, referencing data from patients in all stages of CKD at two different hospitals, with no centralised data analysis, which could in part explain the difference observed between this and our study. In a small single-centre study performed in Spain with 125 patients,23 the cut-off point used was slightly more restrictive in stage 3 (PTH>65mg/dl according to the S.E.N. guidelines, as compared to PTH>70mg/dl recommended by the K/DOQI guidelines), which could also explain in part the fact that the percentage of patients with PTH levels outside of the recommended range (79%) was slightly greater than in our study, in addition to the possible differences in the clinical management of these patients and the different measurement kits used. The characteristics of the patients were similar between the different studies.21,23,24
We observed that 40% of patients had calcium levels outside of the recommended range, which was superior to the rates reported from other studies in patients with CKD not on dialysis (5%-14%),21,24 whereas our value of less than 20% of patients with phosphorous levels outside of the recommended range was similar to the values reported in other studies,21,22,24 and clearly inferior to the rates reported for patients currently on dialysis, in which this percentage surpasses 60%.14 The calcium x phosphorous product was the parameter that exhibited the best level of control.
We observed a strong correlation between the samples analysed at the centralised laboratory and the control measurements taken at each individual hospital, even though both samples were not taken on the same day. This correlation was especially high for calcium, phosphorous, and iPTH levels.
Among the different pathophysiological factors that influence the development of SHPT, calcidiol deficiency is a relatively untreated aspect in patients with CKD, and this parameter is also controversial in terms of its role in pathogenesis and in relation to target levels for CKD. However, calcidiol measurements are considered to be the best metric for evaluating the reserve of vitamin D in the body, and thus provide the optimal measure of the non-renal component of SHPT.
The majority of the patients in the OSERCE study (82%) had inadequate calcidiol levels, with an important deficiency in more than one third of the study sample and no differences between patients who had received active vitamin D supplements and those who had not. Perhaps surprisingly, we also did not observe a significant difference between patients from the different autonomous communities, which had been expected in some cases, especially given the relatively short, homogeneous, and Spring-time (April-May) sampling period. It is not well-known that vitamin D deficits can be common even in equatorial countries, in which solar exposure would be expected to be more than sufficient. Different reasons have been set forth to explain this phenomenon, including nutritional arguments and the case of solar exposure with relation to ancestral exposure, which could justify this generalised deficiency that even affects the general population.38
As in previous studies,22 we did not observe differences in calcidiol levels between the different stages of CKD, regardless of treatment with vitamin D. This appears to indicate that calcidiol levels are fundamentally determined by nutritional intake and exposure to sunlight, rather than by renal function, although recent studies have described direct effects of uraemia on photoconversion of vitamin D39 and/or hepatic hydroxylation.40 In this context, the short duration of the study period allowed for ruling out differences in exposure to sunlight that could have affected different patients disparately during the study. Reduced calcidiol levels were independently associated with calcitriol deficits and more severe SHPT, as has been reported previously.41,42 Another aspect would be to establish a standard method of measurement and cut-off levels defining “deficiency” for calcidiol, which continues to be a source of controversy. Although current guidelines25,27,43 suggest levels >30ng/ml, the Institutes of Medicine have recently recommended levels >20ng/ml for the American population.44 Other studies have shown that levels surpassing 15ng/ml do not improve mortality rates,45 and still other studies have shown low bone turnover rates at levels greater than 40ng/ml.46
The OSERCE study also shows that calcidiol levels provide an independent predictive factor for the development of calcitriol deficit and SHPT, as has been suggested previously.41,42 These results support prioritising normalisation of calcidiol levels as the first step in treating CKD-MBD, as recommended by current guidelines.13,25,27 Treatment with native vitamin D (cholecalciferol and calcifediol) has been associated with mild decreases in iPTH levels, but not sufficiently to warrant using this strategy as a monotherapy for treating SHPT.47 In these cases, active vitamin D supplements (calcitriol, alfacalcidol, or selective analogues for vitamin D receptors, such as paricalcitol in particular), using compounds that provide the greatest value in terms of effectiveness and secondary side effects, particularly hypercalcaemia, hyperphosphatemia, and the development of vascular calcifications. The OSERCE study also analyses calcitriol levels, despite the fact that monitoring these levels is not recommended by the K/DOQI guidelines, and no current guidelines establish target levels for calcitriol based on phosphorous and iPTH, known inhibitors and stimulants, respectively, of 1-α- hydroxylase. In this context, our study expands upon the previously published data regarding calcitriol deficits in patients with CKD, considering for this goal a cut-off point used previously for establishing calcitriol deficits (<22pg/ml).22,36 Based on these ranges, 64.7% of the patients in our sample had calcitriol deficits, and this deficiency was significantly accentuated as glomerular filtration rates decreased. The calcitriol deficit appears to be a triggering factor in the increase of PTH in patients with stage 3 CKD.48 Previous studies have suggested that decreases in calcitriol may appear before PTH levels increase,22 and the OSERCE study reinforces the evidence for this hypothesis. With this in mind, and given the implications of this metabolite in the development of SHPT, it would appear logical in future revisions of the clinical guidelines to include recommendations for the monitoring and early treatment of vitamin D deficits.
Calcidiol values were significantly correlated only with proteinuria (R2=0.018; P=.031), and not with other renal factors such as blood pressure and glomerular filtration rate, which undoubtedly influence proteinuria. This finding is consistent with the results from other recent studies, which have described an inverse correlation between albuminuria and calcidiol levels.49 Despite the fact that the cross-sectional study design implemented here did not allow for demonstrating whether the relationship between vitamin D and albuminuria is causal, recent studies have shown the anti-proteinuria effect produced by normalising calcidiol levels.50,51
Calcitriol levels are also weakly but significantly correlated with proteinuria (R2=.005; P=.001). However, in contrast to calcidiol levels, calcitriol is also correlated with other parameters of renal function, such as blood pressure and glomerular filtration rate, which can act as confounding factors for proteinuria.
Globally, the results of the OSERCE study show an important lack of adherence to K/DOQI guidelines, even though these are quite similar to the current S.E.N. guidelines. The cause for this non-compliance with guidelines continues to be a subject of debate, and may be due to a combination of factors including a lack of awareness, the attitude of the attending nephrologist with regard to therapeutic inertia, infrequent monitoring of CKD-MBD parameters, especially iPTH, and even scepticism, which continues to grow, concerning the application of these guidelines in clinical practice, since the majority of the recommendations therein are based on expert opinion and often simplify what in reality is a very complex process of attempting to maintain biochemical parameters within target ranges.
The first stage of our study has demonstrated the overall non-compliance with these recommendations, especially in stages of CKD prior to dialysis.32 While this is in part explained by the extremely high prevalence of inadequate calcidiol levels, which often go un-measured. This analysis forms part of a relatively new paradigm for the diagnosis and treatment of CKD-MBD, which is still not completely solidified today.25,27 The lack of awareness of non-compliance with the target iPTH and phosphorous values in early stages of CKD, even in the absence of evidence that goes beyond expert opinion, would appear less justifiable. In this sense, the current S.E.N. guidelines (2011)27 clearly recommend preventing phosphorous overload and SHPT, due to the well-established tendency for progressing towards bone mineral metabolism alterations and associated systemic complications, as well as the possibility that delaying treatment measures will further hinder proper control of these alterations and their complications, often requiring added therapies that make the treatment of CKD-MBD much more costly.
In the OSERCE study, we have shown that, in addition to non-compliance with target levels, there is also widespread nonadherence to the recommended frequencies of measuring biochemical parameters. Despite the fact that the guidelines recommend monitoring PTH levels every year in stage 3 patients and every 3 months in stage 4 and 5 patients, PTH levels were not measured during the last year of follow-up in 18% of our study patients. If the parameters used to monitor CKDMBD, especially iPTH, are not monitored, we will be unable to correctly tackle this disease in its early stages, when the parathyroid gland can still respond to treatment. In late stages, the gland becomes hypertrophied and the number of vitamin D receptors decreases, which makes treatment increasingly more difficult, requiring in many cases additional therapies that make the cost of CKD-MBD substantially more expensive.
In addition, the therapeutic strategy chosen can also influence the difficulty in complying with K/DOQI guidelines. Treatment with traditional calcium-based phosphate binders controls phosphorous levels, but it can also alter calcium levels. Approximately one-third of our patients were on treatment with phosphate binders, the majority of which were calcium based. This could explain the good results obtained in controlling phosphorous and less positive results in terms of calcaemia, especially considering the fact that levels above the targets established in the guideline recommendations in 40% of cases. One-third of the patients in the OSERCE study were also on treatment with calcitriol, and none received paricalcitol, since this drug was not commercially available in our country at the time of this study. The use of calcitriol in addition to a calcium-based binder could explain this elevated rate of high calcium levels. The current availability of non-calcium-based binders for use prior to dialysis could contribute to improving these parameters in the future. In addition, the current use of selective activators of vitamin D receptors (paricalcitol) may improve the level of compliance with guideline recommendations, especially in terms of calcium and iPTH levels.
In our interpretation of the results of the OSERCE study, we must keep in mind its inherent limitations. The most important limitation, which we share with the majority of studies published following the release of the K/DOQI guidelines, is the crosssectional nature of our study. This allows for describing the clinical and metabolic situation of the study patients at a certain moment in time, but limits the capacity of the study to establish cause-effect relationships between metabolic alterations and complications and the evolution in time of variations in the different parameters measured. On the other hand, in contrast to the majority of studies previously published regarding compliance with K/DOQI guidelines, the OSERCE study compiled information in a very short span of time (important for avoiding the variability in exposure to sunlight and its effects on vitamin D), and utilised a centralised laboratory analysis in order to determine levels of biochemical parameters, which provides internal validity to the results and underlines the importance of using centralised laboratory analyses, especially if traceable creatinine methods or various methods for measuring iPTH are not used.27,31,52 Finally, and despite not performing a randomised sampling method and only having access to data from a small number of hospitals, the majority of autonomous communities in Spain are represented in our study sample, which contributes to the external validity of the study results, in addition to its concordance with other, smaller national patient cohorts. However, when extrapolating the results of the OSERCE study, it is important to take into account the framework in which the study was carried out (nephrological consultations), given that the monitoring and follow-up of patients with CKD is starting in primary care at an ever-increasing rate. (Sanchez-Hernandez, 2008).
CONCLUSION
The results of the OSERCE study confirm that a high percentage of patients with CKD and who are not on dialysis have levels of biochemical parameters that fall outside of the range suggested by the K/DOQI guidelines and the current 2011 S.E.N. guidelines, primarily in the form of poor control of SHPT and calcidiol deficits. This demonstrates the need for revising these recommendations.
In this context, treatments introduced in recent years, such as non-calcium based binders and active selectors of vitamin D receptors, as well as the future availability of new biochemical parameters such as phosphaturia and FGF-23, may aid in improving the early treatment recommended for CKD-MBD. Our results also highlight the importance of measuring and correcting levels of calcidiol due to its influence on the appearance and development of calcitriol deficits and SHPT. In addition, we now have novel results regarding early calcitriol deficits, the monitoring of which does not form part of the K/DOQI recommendations, despite the fact that decreases in this parameter appear to occur prior to increases in iPTH. These results must suffice as we wait expectantly for results from prospective studies that may inform us as to changes in the level of compliance with parameters for alterations in CKD-MBD, as well as the influence of control of these on cardiovascular events, hospitalisations, and survival, which will be examined in our next endeavour (the OSERCE II study).
Acknowledgements
This study was carried out under the auspices of the Spanish Society of Nephrology and with the logistic assistance of Abbott Laboratories.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Table 1. Demographic and clinical characteristics of the study patients
Table 2. Clinical, biochemical, and treatment characteristics for patients at each stage of chronic kidney disease
Table 3. Association between vitamin D levels and clinical and laboratory parameters
Table 4. Differences between patients in calcitriol levels
Figure 1. Calcidiol, calcitriol, intact parathyroid hormone, calcium, and phosphorous levels based on renal function (centralised laboratory analysis)
Figure 2. Non-compliance with K/DOQI guidelines for intact parathyroid hormone
Figure 3. Vitamin D deficiencies and inadequate levels (excluding nephrotic proteinuria and treatment with vitamin D; n=334)