Living donor kidney transplantation is considered an established treatment for end-stage renal failure and is accepted in different transplant forums, nationally and internationally, while ensuring the safety of the donation, the information, the motivation and caring, the free consent and the absence profit. Safety: the living donor nephrectomy is not extent of risks so a good assessment of the donor's health status and psychosocial situation must be performed to evaluate if the benefits to donor and recipient outweigh the risks assumed. Information and Consent: to be considered ethically acceptable, the donor must be able to give his free consent to the donation after understanding the information provided, accepting the risks and benefits of organ donation, knowing the treatment alternatives for the recipient and the long-term consequences of his decision The absence of profit: offering or receiving money for an organ or other human tissue violates the principles of justice and equity and it is considered ethically and legally unacceptable Motivation: it is important to make a good psychosocial assessment to identify whether the motivation is altruistic or not and, in other terms, to detect any kind of coercion (ex, in the family). Living donation must not be offered in desperate family situations so it is important to assess family relationships to rule out the absence of freedom in donor's choice.The Role of Health Care Ethics Committees: there exists a normative in our country that regulates living donation and establishes that the hospital ethics committees should participate in the process of living donation in all cases. Their job is to assess the process and develop a report on the donor free consent to donation. The responsible person of the living transplant program should provide the documentation necessary to the committee. An interview with the potential donor can be required in some cases.
El trasplante renal de donante vivo se considera un tratamiento establecido para la insuficiencia renal terminal y es aceptado por los diferentes foros de trasplante, tanto nacionales e internacionales, siempre que se garanticen la seguridad, la información, la motivación solidaria, el consentimiento libre y la ausencia de lucro. Seguridad: la nefrectomía de un donante vivo no es un tratamiento inocuo, por lo que deben tratarse de minimizar los riesgos con una buena valoración del estado de salud del donante y su situación psicosocial, de tal forma que los beneficios para donante y receptor superen a los riesgos que se asumen. Información y consentimiento: para se consideren aceptables desde el punto de vista ético, el donante debe ser capaz de firmar su consentimiento a la donación de forma libre tras entender la información suministrada, comprender los riesgos y beneficios que suponen la donación de órganos, las alternativas de tratamiento del receptor y las consecuencias reales a largo plazo. Ausencia de lucro: ofrecer o recibir dinero por un órgano o por cualquier otro tejido humano vulnera los principios de justicia e igualdad y supone un atentado a la dignidad individual, lo que es ética y legalmente inaceptable. Motivación: se trata de un aspecto ético fundamental y es importante que se realice una buena evaluación psicosocial para reconocer si la motivación es solidaria o no y, en otros términos, si existe algún tipo de coacción (p. ej., en el entorno familiar). No se debe plantear la donación en situaciones desesperadas y es importante conocer bien las relaciones intrafamiliares para descartar la ausencia de libertad de elección del donante. En estos casos se debe ofrecer una salida airosa al donante, sin faltar a la verdad, si éste quiere revocar su consentimiento. El papel de los Comités de ética asistencial (CEAS): en nuestro país, en virtud de la normativa que regula el trasplante renal de vivo, los CEAS deben participar en el proceso en todos los casos. Su labor consiste en elaborar un informe sobre la libre elección del donante. Para ello, el comité evaluará la documentación aportada por el responsable del programa de trasplante de vivo (o la persona que designe) y se entrevistará con el donante en caso de considerarlo necesario.
INTRODUCTION
Legal and ethical aspects associated with living kidney donation have been much debated throughout the already lengthy transplant history. Although there is consensus that living transplantation is the treatment of choice for end-stage chronic kidney failure,1 aspects such as donor safety, the donor’s motive, suspected abuse or rampant commercialism, have caused its development to vary between countries, and is even directly rejected by the transplant teams. In spite of this, we have witnessed an astonishing increase in living donation, and at present it represents almost half of all kidney transplants that are performed throughout the world. There are two types of living transplantation: one which is based on goodwill and the other which is based on business. The two coexist side by side, and both involve risk for the donor, however, the first is built upon noble values which society should foster and the second inevitably harms these values.
ETHICAL PRINCIPLES AND VALUES INVOLVED IN LIVING KIDNEY DONATION
Numerous public entities and organisations have discussed which conditions should be part of a fair living-donor organ and tissue donation and transplant system.2,3 More specifically, at the end of the nineties the Convention on Human Rights and Biomedicine established the following regulations:
- Removal of organs or tissue from a living person for transplantation purposes may be carried out solely for the therapeutic benefit of the recipient and where there is no suitable organ or tissue available from a deceased person and no other alternative therapeutic method of comparable effectiveness.
- The necessary consent must have been given expressly and specifically either in written form or before an official body. The human body and its parts shall not, as such, give rise to financial gain.
In 1991, a set of Guiding Principles on Human Organ transplantation was approved during the 44th World Health Assembly. This had an important impact on developing legislations, practices and professional codes within Member States. Almost 20 years later the WHO updated them in the 124th session due to the new challenges brought about by a shortage of organs and due to related ethical issues. The following has been established with regards living donation:
Adult living persons may donate organs as permitted by domestic regulations. In general, living donors should be genetically, legally or emotionally related to their recipients.
Live donations are acceptable when the donor's informed and voluntary consent is obtained, when professional care of donors is ensured and follow-up is well organized, and when selection criteria for donors are scrupulously applied and monitored. Live donors should be informed of the probable risks, benefits and consequences of donation in a complete and understandable fashion; they should be legally competent and capable of weighing the information; and they should be acting willingly, free of any undue influence or coercion.
More recently, given the excellent results from living-donor transplantation between two non-genetically related people and the increasing need for organs, the Committee of Ministers of the Council of Europe (CMCE) allowed kidney transplantation from living donors who are not genetically related to the recipient in their Resolution CM/Res(2008).5 This is permitted provided that the listed conditions are respected for the given transplant, regulations have put in place in view of prohibiting and preventing organ trafficking, and clearly defined rules have been established for non-residents.
All of the values that must be guaranteed in any type of living-organ and tissue donation programme are given implicitly within these recommendations, i.e. donor’s safety, availability of information, donor’s decision making ability, altruistic motivation, non-coercion or financial gain, and ensuring free, voluntary and expressed consent.
SAFETY
Living donation is a complex therapeutic procedure, the responsibility for which is not only held by the patient requesting the transplantation, but is shared with the professionals that perform the intervention.6 The intervening professionals must assess whether the necessary technical and ethical requirements are met, and whether their participation in the intervention can be morally justified. The main ethical problem since the first successful living kidney transplant in 1954 between identical twins has been to establish whether it is morally justifiable to subject a healthy patient to a high-risk process so as to save a life or improve another patient's health. The main opposing argument is that the ‘Nonmaleficence’ principle is violated even with the donor's consent (Primum non nocere: first, do no harm). It is clear that it is almost impossible to point out a risk-free medical intervention, meaning that donor risks should be minimised so that the donor-receiver benefits overcome the risks. The donor’s health status, the receiver’s clinical situation, the surgical technique employed and the surgical team's experience have an influence on transplant success probability and are factors for assessing the procedure’s risk-benefit ratio and whether it can be ethically justified.7
Nowadays, open nephrectomy donor mortality rate is less than 0.03%, and the outcome and expected quality of life are similar to those of the general public. Furthermore, the introduction of minimally invasive surgical techniques (laparoscopic nephrectomy) has reduced preoperative morbidity, improving aesthetic results and shortening the time it takes for the donor to resume everyday activities.8 For ethical reasons, it would therefore be desirable for all accredited living transplant programmes to use this technology. Life and health insurance contracted by donors can be used as a sensitive gauge of the risk involved in the intervention as it is clear that there are no significant risks when the premiums do not increase.9 Nevertheless, the risks that the donor is subject to should be accepted by the scientific community, independently of the donor-recipient relationship.
For several years, and in line with the Oviedo Agreement, it is still believed that living donation should be considered as a last resort, i.e. once any possibility of a deceased donor is exhausted or when there are no other comparably efficient treatments available. This strategy would justify why little development has been made regarding living donation in Spain, which has the highest level of deceased transplants in the world. At present, we know that prolonged dialysis reduces the possibility of accessing a transplant and it has a negative effect on graft and recipient survival, while transplant before dialysis entails an improved graft survival.10 Furthermore, living transplantation, even with a non-related donor, gives the recipient a better average life-span (16 years compared with 10 from deceased donors).11
The international transplant community met at the Amsterdam Forum and accepted living kidney transplant as an adequate therapeutic procedure, provided that the intervention is in line with its ethical recommendations, i.e. that physical, psychological and social consequences are minimised for the donor, that he or she is able to make an independent decision, and that the clinical outcome is rigorously monitored.12
INFORMATION AND CONSENT
As indicated in the Additional Protocol to the Convention on Human Rights and Biomedicine,13 a organ or tissue may be removed from a living donor only after the person concerned has given free, informed, and specific consent to it in written form before an official body. Furthermore, the donor has the freedom to withdraw consent at any time.
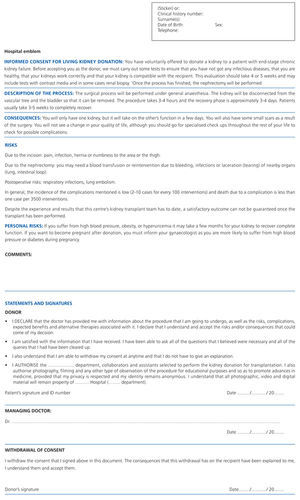
Informed consent is not a one-off and isolated event on which the donor prints his or her signature onto a document. It is, however, a gradual process, shared with all of the healthcare professionals who provide information to the donor and answer his or her questions. Finally, the managing doctor will evaluate whether the donor fully understands the information provided, will assess his or her decision making ability, and answer any question that the donor may have. The doctor will then give the donor a printed form (Attachment 1) so that he or she can take time to read it and reflect upon this decision before signing.
This consent form accompanies the information that the doctor has discussed with the patient, and serves as a record that the patient has been correctly informed and consents to the donation. It also has legal importance and indicates the donor's preferences.14 The document should be written with simple words and short phrases, should avoid numerical expressions indicating probability, and should be no longer than two pages. It should at least include a brief description of the procedure, the intervention’s risks and those related to the donor’s personal circumstances, the important health consequences and a section for statements and signatures where any specific conditions and revocation clauses may be included.15
Other requirements must also be met so that the consent process is ethically acceptable: all potential donors should be able to show that they understand the information provided, the risks and benefits that are involved with the donation, the benefits and alternative treatments available to the recipient and the actual medium- and long-term consequences. Medical staff must also evaluate the donor’s social, employment and psychological situation, consider the donor’s relationship with the recipient, his or her motive for donating, and ensure that the donor has not been coerced. This psychosocial evaluation16 determines the acceptance of the donor (especially for unrelated donors) and should be performed before the clinical trials using invasive techniques, so as to ensure that the donor is not submitted to unnecessary risks.
It is necessary to prove that there is no coercion involved and that the donor has made his or her decision freely and voluntarily, but this is not always easy. Often for related donation cases, when a loved one is suffering, the potential donor may feel like he or she owes it to the recipient or that there is a certain amount of pressure from other family members. This could prevent the potential donor from making a completely free and voluntary decision. It is for this reason that donation should not be offered in desperate situations. In any case, family relationships must be studied, time must be given for reflection and the potential donor must be supported if he or she decides to change his or her mind (but always giving true version of events).17
Another important ethical aspect is the motivation for the donation. Not all donations are for pure altruist reasons such as: satisfaction in helping the person who is suffering, feeling that it is a moral obligation, searching for improving self-esteem by doing a good deed or thinking that the recipient would have done the same in his or her position (reciprocity), all of which are perfectly acceptable motives. Motives are debatable when they are religiously influenced, when the donor wants to cleanse feelings of blame, or there is family interference and it is like an obligation. They must be rejected when the donor has financial motives, is looking for social recognition, wishes to solve personal problems or if there is a hidden pressure from the recipient or the healthcare professionals that are caring for him or her.18 The psychosocial evaluation should therefore be performed by professionals with extensive experience with these issues and that are not part of the teams that participate directly in the donation and transplant process.
Organ donation is decided upon freely and voluntarily. No one should ever be demanded or obliged to donate an organ. It is a heroic act that ennobles the person that donates, highlights healthy and supportive society values and should make us all feel proud. Non-directed donation aimed at the recipient at the top of the waiting list (Good Samaritan) is ethically acceptable and should be encouraged and socially recognised, provided that the donor’s motive is altruistic.
The Healthcare Ethics Committees’ (HECs) mission is to establish that living-donor consent has been given freely, voluntarily and altruistically.
HECs AND LIVING DONATION
In Spain, if a hospital is able to provide a living transplant, its HEC must participate in the process (Article 9.2 of the Royal Decree 2070/1999, of 30 December). Despite the Committee’s role being particularly ambiguous within this law, it specifies that it must be consulted in all cases and that the Committee must issue a non-binding preceptive report in which the donor’s free consent is declared (Article 9.2, section 1.c). Some consider that the regulation should also discuss the likelihood of transplant success, but given that HEC19 members are of various disciplines, it is difficult for them to be able to answer such a technical question, which should be the responsibility of a transplant committee or the healthcare professionals that have indicated the intervention.
Another issue that has generated much debate has been how the Committee gathers the information used for the report. As it can not be carried out in any other way, each Committee can freely and independently design its own strategy. Some Committees obtain information from the professionals that intervene in the different evaluations or from the documents that they have provided, others ask the donor or the donor and recipient a series of questions, and some use a combination of all of these strategies. No studies have been conducted to show which the best strategy is, but the most important aspect is that the procedure complies with a protocol and that the transplant team agrees with it, to ensure that a fast response can be given and to guarantee that the donor’s consent meets all of the ethical and legal requirements.
HEC Evaluation Process
The Transplant Coordinator contacts the Committee and informs of the cases that need to be evaluated and their level of urgency. The secretary, who summons the Committee, should receive the documents from the transplant team within the time agreed. They should normally be available 48 hours before the Committee meeting is to take place so that the Committee members are able to consult them. Given that this documentation is confidential, it should not be circulated and should only be consulted by HEC members in the Committee’s office.
The documentation needed by the HEC may vary depending on the health centre, but it should at least include the following:
- Evaluation application sent to the HEC Chairman.
- Donor’s clinical report, conducted by a doctor who is not involved in the removal and transplant process. This report also confirms that the patient has been informed of the risks, consequences, repercussions on his or her life and the predicted outcomes for the recipient.
- Report/psychological evaluation by an experienced professional.
- Social-employment evaluation by a social worker.
- Donor’s informed consent.
- Recipient’s clinical report.
- Recipient’s informed consent.
- Transplant Coordination report which states whether he or she agrees with the intervention.
To ensure that the consent is valid, the HEC is recommended to perform the evaluation once all of the clinical trials have been completed, all the other potential donors have been ruled out and the donor is aware that he or she is the only candidate. In exceptional cases, when there is only one donor and it is a clinical emergency, the HEC can bring their evaluation forward even if there are still analyses to conduct, so as to facilitate the process and act on the recipient’s best interest (Figure 1).
Evaluation
We are going to make reference to an evaluation that combines data from the documentation with information obtained from a semi-structured interview, although there are other, equally useful methods.
Although each Committee is able to design its own strategy, the Committee can conduct the interview with the donor during an ordinary session or using a subcommittee created especially for this purpose, which convenes an extraordinary session. When a subcommittee is necessary it should have at least four members and be representative of the HEC.
Once the donor has been introduced to the Committee members and the meeting’s objectives explained, the group will have a relaxed conversation with the donor about the different donation-related aspects. Using open questions (Attachment 220) the Committee will go on to review aspects related to the information that he or she has been provided, the consent process, motive for donating, decision making process, social, work and financial aspects associated with donation. The Committee will also inform the donor that he or she is able to withdraw consent without any consequences.
Lastly, the HEC must issue a report within the agreed period of time. The report will include the information regarding the affiliation between the donor and the recipient, documentation that has been used, donor’s decision making ability, and degree of satisfaction with the information received. It will also state that there is no coercion, that the donor has made his or her decision freely and voluntarily and will include a conclusion which justifies whether the Committee agrees with the donation.
FINANCIAL GAIN
Living kidney donation from related or unrelated donors has doubled during the past decade. In 2006 alone, 27 000 kidney transplants were performed (data from 69 of the 90 countries that provide living kidney transplants), which represents 39% of all kidney transplants.21 According to the World Health Organization, more than 63 000 kidney transplants are performed annually throughout the world, and that payment is made to approximately 10% of unrelated living donors.
There are many positive arguments for creating a regulated non-vital organ market, such as: controlling organ trafficking, establishing a fair distribution system, ensuring donor security, and reducing the shortage of organs. However, the reality is that unrelated commercial living donation exploits poverty and has fateful outcomes. In countries where the legislation does not prohibit organ trading, this type of transplantation is the only one which has developed, showing that it is not for health reasons but financial gain. The most vulnerable populations, such as impoverished and illiterate individuals, undocumented immigrants, refugees, and prisoners are the source of organs for rich tourists. Health care centres, surgeons and financial intermediaries that have no qualms justify paid donation saying that it is a way of reducing waiting lists, but really only the rich are able to gain access to transplants, not the country’s citizens that need them.22 These procedures unforgivably damage some of our society’s most solid values such as life, health and freedom, which cannot be priced. Offering and receiving money for an organ or any other type of tissue from the human body further violates the principles of justice and equity and represents an attack towards an individual’s dignity (people have dignity, not a price tag). It is therefore ethically unacceptable.23
Due to the increasing problems associated with organ trade, transplant tourism and organ donor trafficking, The Transplantation Society (TTS) and the International Society of Nephrology (ISN) recently issued the Istanbul Declaration.24 The Declaration urges countries to legislate and prohibit unethical practices, provide healthcare to citizens that have been victims of organ trafficking, improve controls to protect their people from exploitation and to develop procedures that allow national self-sufficiency in organ donation. Furthermore, they recommend dissolving scientific societies that accept members who do not adhere to the declaration. They also recommend that industry should not collaborate with these professionals or finance their initiatives, and that scientific journals should not accept their publications.
ATTACHMENT 1. Informed consent model
ATTACHMENT 2. Checklist for interviews with living kidney donors (ref. 20)