To the Editor:
Pneumocystis jiroveci pneumonia is a serious opportunistic infection that affects immunocompromised patients. It has been extensively described in the context of acquired immunodeficiency syndrome (AIDS), long-term steroid treatment and solid organ transplant recipients, mainly in the period covering the first six months after transplantation.
We report the case of a patient who, having recommenced haemodialysis after eight years with a functioning renal graft, developed progressive respiratory symptoms with bilateral pulmonary infiltrates, showing Pneumocistis jiroveci pneumonitis after recent hospitalisation due to graft pyelonephritis. The occurrence of this fungal infection is exceptional outside the first months following transplantation, but it may occur at any time during progression, and as such we must bear it in mind for its early diagnosis and treatment.
Our patient is a 66-year-old male, a former smoker, with long-term high blood pressure, systemic arteriosclerosis and intermittent claudication and he had a transient ischaemic episode in 1994. He has chronic kidney disease secondary to nephroangiosclerosis and has been on haemodialysis since August 2002 through an arteriovenous fistula in his left forearm.
He received a deceased donor renal transplant in November 2004, and achieved baseline creatinine of between 1.2 and 1.4 mg/dl. In August 2005, following a progressive decline in renal function, a renal biopsy was performed that showed an acute rejection grade IB with a good response to steroid therapy. He subsequently developed type 2 diabetes mellitus, requiring oral anti-diabetic medication (September 2008). In January 2012, he was diagnosed with a moderately differentiated adenocarcinoma of the lung in the upper right lobe; he underwent 3 chemotherapy and 33 radiotherapy sessions with a good response to treatment.
In September of the same year, he started to experience a deterioration in renal function and uraemia, accompanied by fever and pain in the area of the graft (left iliac fossa). He was admitted to the Nephrology department, where he was diagnosed with pyelonephritis due to E. coli sensitive to ciprofloxacin, for which he was treated for seven days. After recommencing haemodialysis, he was discharged on the third day to complete treatment at his home as an outpatient.
One week later, the patient came to the Emergency Department due to recurrence of symptoms. He was eupnoeic at rest, with normal colour and was well perfused and had a fever up to 38°C. Blood pressure was 150/80mmHg and was rhythmic at 108bpm. Cardiac auscultation revealed the presence of a systolic murmur. Pulmonary examination was unremarkable, without unusual sounds. Abdominal examination showed a soft and palpable abdomen, and there was pain in the left iliac fossa on palpation, without masses, organomegalies or peritonitis. There was no oedema in the lower extremities. The blood test showed the following results: haemoglobin 10.8g/dl, haematocrit 34.6%, mean corpuscular volume 90fl. Platelets: 160,000. Leukocytes: 5160/mmc. Neutrophils: 78.3%. Prothrombin time: 72.5%. The biochemistry results were as follows: glucose 109mg/dl, creatinine 5.48mg/dl, sodium 140mEq/l, potassium 3.78mEq/l, chloride 102mEq/l, CRP 42.6, lactate dehydrogenase (LDH) 422U/l, albumin 2.09g/dl, CD3 count: 422 cells/Ul (87.2%) and CD4 count: 106 cells/Ul (21.2%). The chest X-ray on admission showed a calcified aortic arch with a mass effect in the right upper lobe, present in previous studies, with a marked improvement following radiotherapy.
The abdominal X-ray showed an enlarged liver without space-occupying lesions, gallbladder and suprahepatic veins of normal appearance, splenomegaly and both kidneys were atrophic and displayed bilateral cortical cysts. The renal graft showed a slight increase in cortical echogenicity with vascularisation in its interior and a 6 x 3cm subcapsular collection reported in previous studies. Serial blood cultures were obtained with negative results. The urine culture obtained was positive for E. coli sensitive to ciprofloxacin and resistant to cephalosporins, and as such, we started a new cycle of intravenous antibiotics.
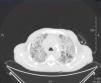
After 24 hours, the patient developed symptoms of a cold, consisting of a dry cough and progressive dyspnoea that did not respond to an increased antibiotic dose. Another chest X-ray was requested, which showed a bilateral interstitial pattern with a ground glass appearance (Figure 1).
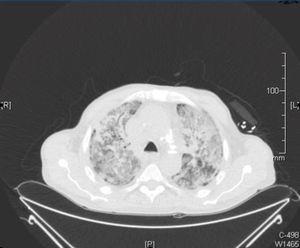
A computerised tomography (CT) scan was requested, along with a bronchoscopy and bronchoalveolar lavage and a PCR for cytomegalovirus (CMV) and sputum cultures, expanding the antimicrobial spectrum with sulfamethoxazole trimethoprim, liposomal amphotericin and ganciclovir. In the chest CT scan, we observed a localised mass in the right upper lobe with pleural contact and a marked decrease with respect to the previous CT scan, with a consolidative pattern and ground glass areas accompanied by a cobblestone pattern (Figure 2).
In the sputum culture, we isolated the normal oropharyngeal microbiota, with PCR being negative for CMV, while the bronchoalveolar lavage showed Pneumocystis jiroveci and Candida parapsilosis DNA. Given these findings, we continued treatment with therapeutic doses of 800/160mg trimethoprim-sulfamethoxazole for three weeks and 500mg/day ciprofloxacin, adding 100mg/day fluconazole. Clinical progress was excellent, with the progressive disappearance of symptoms, as well as gradual normalisation of radiological test data.
Pneumocystis pneumonia continues to be the most common opportunistic disease in immunocompromised patients. Currently, the terminology that should be used is Pneumocystis jiroveci pneumonia, although, the acronym “PCP” continues to be employed, since it stands for “Pneumocystis pneumonia”. It is necessary to keep it in mind in differential diagnoses at all times in post renal transplantation progression, as well as in patients who recommence dialysis and who may be immunosuppressed due to another cause, such as oncological treatment, as in the case we describe.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Figure 1. Chest X-ray.
Figure 2. Chest computerised tomography scan.