The existence and type of renal involvement influences the prognosis of systemic lupus erythematosus and this information may be critical when it comes to taking appropriate therapeutic decisions.
ObjectiveTo evaluate statistical correlations between clinical and histological data in patients with biopsied lupus nephropathy.
MethodsReview of clinical information in adult kidney biopsy requests reported between 2002 and 2014 with a definitive clinical and histopathological diagnosis of renal involvement in systemic lupus erythematosus.
Results134 cases (86% women), aged 15–59 years. Indication for renal biopsy: asymptomatic urinary abnormalities (30%), nephrotic proteinuria without hypoalbuminaemia (9%), nephrotic syndrome (19%), renal failure (40%) and two cases without clinical renal manifestations. The most common lesions were purely proliferative (68%). In patients with asymptomatic urinary abnormalities, 35% were class IV, 30% class III, 23% mixed, 10% class V and 2% class II. In subjects with nephrotic proteinuria, 75% were class IV, 17% mixed and 8% class III. In nephrotic syndrome patients, 46% were class IV, 27% class V, 19% mixed and 8% class III. In renal failure subjects, 67% were class IV, 22% mixed, 7% class III and 4% class V. These proportions were not statistically different. Although class IV showed the worst renal function, almost half (44%) of those without renal failure belonged to this class.
ConclusionWe could not demonstrate a consistent clinical–pathological relationship that predicts patterns or severity of histological findings based on the clinical profile in patients with systemic lupus erythematosus and renal manifestations. These results highlight the importance of biopsy as a key diagnostic tool in this disease.
La presencia y patrón morfológico del compromiso renal afecta el pronóstico del lupus eritematoso sistémico, información que puede ser indispensable para tomar decisiones terapéuticas apropiadas.
ObjetivoEvaluar relación estadística entre datos clínicos e histológicos en pacientes con nefropatía lúpica biopsiada.
MétodosRevisión de información clínica en solicitudes de biopsias renales de adultos, informadas entre 2002 y 2014, con diagnóstico clínico e histopatológico inequívoco de compromiso renal por lupus eritematoso sistémico.
ResultadosCiento treinta y cuatro casos (86% mujeres), edad 15-59 años. Cuadro clínico: 30% alteraciones urinarias asintomáticas, 9% proteinuria nefrótica sin hipoalbuminemia, 19% síndrome nefrótico y 40% por insuficiencia renal, existiendo 2 casos sin manifestaciones clínicas renales. Las lesiones más frecuentes fueron proliferativas puras (68%). De los que tenían alteraciones urinarias asintomáticas, 35% eran clase IV, 30% clase III, 23% mixtas, 10% clase V y 2% clase II. Entre los de proteinuria nefrótica, 75% clase IV, 17% mixtas y 8% III. De los de síndrome nefrótico, 46% clase IV, 27% V, 19% mixtas y 8% clase III. Entre los de insuficiencia renal, el 67% eran IV, 22% mixtas, 7% III y 4% V. Estas proporciones no fueron estadísticamente diferentes. Aunque la peor función renal fue observada en la clase IV, casi la mitad (44%) de aquellos sin insuficiencia renal eran de esta misma clase.
ConclusiónNo se demuestra una relación clínico-histológica consistente que permita predecir los patrones ni la gravedad de los hallazgos histológicos a partir del cuadro clínico en el lupus eritematoso sistémico con manifestaciones renales. Esos resultados refuerzan la importancia de la biopsia como herramienta diagnóstica fundamental en esta enfermedad.
Systemic lupus erythematosus (SLE) is a multi-systemic autoimmune disease that may involve almost all body organs. Renal involvement may be present in more than half of the patients at the first year of diagnosis,1,2 with a significant impact on morbidity and mortality,3 and the inherent risk of progression to advanced chronic renal failure requiring dialysis or transplantation.4–6 A 10% of SLE patients with renal involvement will reach terminal chronic kidney disease, which can be up to 40% at 15 years in cases of diffuse proliferative lesions.7 For this reason, it is important to identify and categorize individuals showing clinical renal involvement so an appropriate treatment is initiated to stop the progression of the disease.8 Although some researchers have suggested that the experience in the management of lupus nephropathy, together with clinical information, are sufficient to indicate adequate treatment,9–11 adherents to this attitude are being progressively reduced.
The introduction of routine renal biopsy in the 1950s, advances in immunofluorescence techniques and electron microscopy in the 1960s, together with knowledge of the immunopathogenesis of glomerular damage, allowed us to know the different histopathological patterns associated with SLE.12 There has been progressive improvement in the histopathological classification criteria recommended by the World Health Organization in the 70s, which has been reviewed several times to reach the current classification ISN-RPS 2003.13 This has allowed to conform the basis for prospective therapeutic and prognostic studies14 in an effort to identify those patients with a higher risk of progression that justifies the use of more aggressive immunosuppressive therapies.
Knowing the importance of histopathological findings in therapeutic decisions in SLE patients with renal involvement, we wanted to investigate if patterns of clinical presentation can reliably predict the findings obtained from a renal biopsy.
Material and methodsThe cases were selected from the kidney biopsy file of the Nephrology Unit of the Universidad Austral de Chile, Regional Hospital of Valdivia. The study included the material reported between 2002 and 2014 corresponding to subjects older than 13 years whose clinical–histopathological diagnosis corresponded unequivocally to the renal involvement of the LES and with sufficient clinical information justifying the indication of the histological study and a complete anatomopathological evaluation including optical microscopy, immunofluorescence and electron microscopy. In the case of the samples obtained prior to the clinical use of the ISN-RPS 2003 classification, they were re-analyzed and classified according to the new criteria.
This research was approved by the Scientific Ethics Committee of the Valdivia Health Service and the Regional Hospital of Valdivia.
DefinitionsClinical picture that justified the histological study- •
SLE without renal manifestations: patients diagnosed with SLE who were indicated for a biopsy without having clinical manifestations of renal involvement (hematuria, proteinuria or deterioration of renal function).
- •
Asymptomatic urinary abnormalities (AUA): patients with clinical-serologic findings compatible with SLE and alterations such as glomerular hematuria and/or proteinuria in the non-nephrotic range (<3.5g/24h) and with estimated glomerular filtration rate (eGFR) >60ml/min/1.73m2.
- •
Proteinuria in the nephrotic range without hypoalbuminemia (NP): patients with clinical–serological symptoms compatible with SLE and proteinuria> 3.5g/24h, without hypoalbuminemia, and with eGFR> 60ml/min/1.73m2.
- •
Nephrotic syndrome (NS): patients with clinical-serologic symptoms compatible with SLE and proteinuria >3.5g/24h, hypoalbuminemia <3g/dl and FGe >60ml/min/1.73m2.
- •
Renal insufficiency (RI): patients with a clinical-serological picture compatible with SLE and eGFR ≤60ml/min/1.73m2, with or without other renal manifestation. If the eGFR was between 59 and 30ml/min/1.73m2 it was considered moderate and if it was <30ml/min/1.73m2 as severe.
It was used the ISN-RPS-2003 classification, including the criteria necessary to consider mixed lesions (coexistence of membranous nephropathy and proliferative lesions) and those that consider segmental/global lesions for the case of type IV nephropathy.13 Additionally, for the cases of proliferative lesions III, IV or mixed, there was information available on NIH indices of activity/chronicity published by Austin et al.15
Renal functionThe abbreviated four variables-MDRD formula based on values of serum creatinine was used for the estimation of the glomerular filtration rate.16
Statistical analysis: The continuous variables were expressed as mean±standard error and the comparisons between groups performed by the Kruskal–Wallis-ANOVA test followed by a nonparametric Mann–Whitney T-test when applicable. The comparison of frequencies of qualitative variables was obtained through the Fischer contingency test. The Spearman test was used to analyze correlations. A p value <0.05 was accepted as significant. All calculations were made using the GraphPad Prism version 6.00 for Windows, GraphPad Software, La Jolla California USA, www.graphpad.com.
ResultsWe analyzed the clinical and histopathological data of 134 patients corresponding to 116 women and 18 men, all of them more than 13 years of age. The average age was 29 years (range 15–59), without statistical differences between genders (27±2.9 for men vs. 29±0.9 for women).
The clinical manifestation that justified the histological study had the following distribution: AUA (30%), NP (9%), NS (19%), RI (40%) and two cases without renal clinical manifestations; the general histological classification showed the following distribution: class II 1%, class III: 14%, class IV: 53%, class V: 10% and mixed lesions: 22%. Of type IV lesions, 58 (82%) were global (IV-G) and 13 (18%) segmental (IV-S). Of the 29 mixed lesions, 11 corresponded to classes III+V and 18 cases to IV+V.
Clinical–histopathological relationshipsAnalysis of the clinical manifestations that motivated the histological study compared with the histology findings (Fig. 1) revealed that in 40 cases biopsied because of AUA, 14 corresponded to class IV, 12 to class III, 9 to mixed injuries, 4 to class V and 1 to class II. Of the 12 with N P, 9 corresponded to class IV, 2 to mixed injuries and 1 to class III.
Distribution of histological classes (ISN-RPS 2003 classification) found in the biopsy according to renal clinical manifestation. Classification ISN-RPS 2003. Source: Weening et al.13
AUA: asymptomatic urinary abnormalities; RI: renal insufficiency; NP: proteinuria in the nephrotic range without hypoalbuminemia; NS: complete nephrotic syndrome.
Among the 26 with NS, 12 were of class IV, 7 class V, 5 mixed lesions and 2 of class III.
In the 54 cases with renal insufficiency, the most frequent lesion was class IV (36 cases), followed by mixed (12), class III (4) and class V (2), without differences between those with moderate or severe renal failure. Of the 2 patients who underwent a biopsy without clinical evidence of renal involvement, one corresponded to a class II nephropathy and the other to a mixed proliferative lesion (IV+V).
Renal functionNo significant difference in eGFR was observed between those with NP as compared with those with NS or AUA.
Renal function was significantly lower in class IV lesions compared to class III or V. There were no differences in eGFR between global class IV lesions and the segmental forms. Mixed lesions showed no differences in renal function with respect to pure proliferative lesions, whether there were class III or IV (Table 1).
Clinical and histopathological variables in patients with lupus nephropathy according to histological class.
| Class III | Class IV | Class V | Mixed | |
|---|---|---|---|---|
| eGFR | 85±8.6 (n=19) | 60±4.2**** (n=71) | 87±8.9 (n=13) | 77±7.7 (n=29) |
| Proteinuria (g/24h) | 3.7±1.0 (n=19) | 4.3±0.3 (n=71) | 4.6±0.8 (n=13) | 4.9±0.6 (n=29) |
| Serum albumin (g/dl) | 3.1±0.1* (n=19) | 2.5±0.1 (n=71) | 2.4±0.2 (n=13) | 2.4±0.1 (n=29) |
| C3 (mg/dl) | 55±6.5 (n=16) | 48±2.9 (n=51) | 79±6.2** (n=12) | 50±5.2 (n=23) |
| C4 (mg/dl) | 7.4±1.3 (n=17) | 7.0±0.7 (n=49) | 9.5±1.4 (n=12) | 8.1±1.4 (n=23) |
| ANA (% positive) | 100% | 93% | 82% | 87% |
| Anti-DNA (% positive) | 88% | 79% | 80% | 76% |
| % Crescent in the biopsy | 10±2.0 (n=19) | 32±2.8**** (n=70) | 0.7±0.5 (n=13) | 19±3.3 (n=29) |
| Activity index | 6.7±0.5 (n=19) | 12.6±0.3*** (n=70) | nc | 8.9±0.7 (n=29) |
| Chronicity index | 3.0±0.3 (n=19) | 3.1±0.2 (n=70) | nc | 3.3±0.3 (n=29) |
ANA: antinuclear antibodies; Anti-DNA: anti-DNA antibodies; C3: complement factor C3; C4: complement factor C4; FGe: glomerular filtration rate (ml/min/1.73m2); nc: it is not appropriate to evaluate; indices of activity and chronicity in proliferative lesions according to NIH. Source: Austin et al.15
Regarding the magnitude of the proteinuria, it was significantly higher in the NS group as compared to both the NP and RI group and also when compared to the group with IR. There were no differences in values of proteinuria between moderate and severe RI.
The magnitude of proteinuria was not different between the various histological classes (Table 1).
Significant higher levels of albuminemia were found in class III, with no differences between the other histological groups (Table 1).
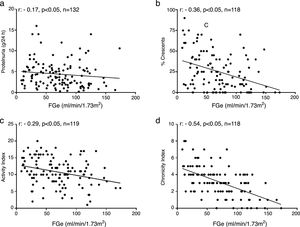
Additionally, a negative statistically significant correlation was found between the magnitude of proteinuria and renal function estimated by MDRD (Fig. 2a).
Correlations in lupus nephropathy. (a) Between proteinuria and renal function. (b) Crescents and renal function. (c) Index of activity and renal function in proliferative classes. (d) chronicity index and renal function in proliferative classes.
FGe: glomerular filtration rate estimated by MDRD formula; activity and chronicity indexes according to NIH. Source: Austin et al.15
The level of C3 complement fraction were decreased in 88% of the cases, with higher values for the class V. Regarding C4, 72% of the cases had levels below the normal range, without differences between the different histological classes. A 86% had positivity for antinuclear antibodies and 80% for anti-DNA, without differences between histological classes (Table 1).
Other findings in renal biopsyPresence of glomerular crescents: the presence of crescents was a common finding in pure or mixed proliferative lesions, however they were not observed in class II lesions and were detected only in 2 of the 13 class V lesions. Of the total number of biopsies analyzed, 73% showed crescents involving on average a 31% of the glomeruli (range: 4–90%), being significantly more extensive in class IV biopsies as compared to class III, IV or mixed. Patients with mixed lesions of class IV+V had a significantly higher percentage of crescents than those of class III+V. No differences were observed in the percentage of crescents when comparing IV-G lesions with IV-S.
There was also a negative correlation, statistically significant, between renal function and percentage of crescents in the group of proliferative lesions (Fig. 2b).
Histological activity and chronicity indexes: NIH activity and chronicity indexes were evaluated in pure or mixed proliferative lesions, excluding the two cases without clinical evidence of renal compromise and a patient with class IV because information on these indices was not available.
The comparison of the activity indexes in biopsies separated according to the clinical symptoms that motivated the biopsy showed that patients with AUA had values of activity indexes significantly (p<0.05) lower (8.6±0.5, n=35) than those with proteinuria in the nephrotic range (10.8±1.0, n=12), with NS (10.6±1.0, n=19) or with renal failure (12.27±0.5, n=51), and there was not statistical significant differences among these last three groups. There were no significant differences between those with moderate and severe RI also.
The histological class showing higher activity indices (p<0.05) was class IV as compared with class III or mixed lesions (Table 1). This was reproduced in the separate analysis of mixed lesions in which IV+V had higher activity indices than III+V (11.1±0.6 vs. 5.3±0.9, p<0.05). Additionally, the IV-G lesions (n: 57) were compared with the IV-S lesions (n: 13) and no differences in the activity indexes were observed (12.8±0.4 vs. 11.9±0.8, p: NS).
Regarding the chronicity indexes, excluding patients without renal clinical manifestations, those with renal failure showed significantly higher values (4.2±0.2, p<0.05) than those with AUA (2.4±0.2), NP (1.8±0.5) and those of NS (2.3±0.2), but no difference was observed between moderate and severe RI. Comparisons of chronicity indexes according to histological class, showed no differences between the three groups of proliferative lesions (Table 1). No statistically significant differences were found in the chronicity indexes of the global class IV lesions (2.9±0.2) compared with the segmental ones (3.6±0.6).
Additionally, a negative statistically significant correlation was observed between renal function and histological activity and chronicity indices in the group of proliferative lesions (Fig. 2c and d).
The activity indexes were similar in men and women (12.2±0.9 vs. 10.5±0.4 respectively) as were the chronicity indexes (2.4±0.4 vs. 3.3±0.2). In the subjects <30 years, the activity indexes were similar to those ≥30 years (10.6±0.4 vs. 11.1±0.6); however, the rates of chronicity were significantly higher in subjects ≥30 years (3.6±0.2) than in <30 years (2.9±0.2).
DiscussionThis series of renal biopsies performed on Chilean adults with SLE shows the weak clinical–histopathological correlation in this disease.
It is noteworthy the presence of class IV lesions in 35% of the cases that clinically manifested as AUA. This is relevant because the position of restricting renal studies to the severity of the clinical picture of SLE is strongly questioned with these results.
There is a believe that severe histological lesions are associated with high levels of proteinuria, and vice versa.17 We did not find higher levels of proteinuria in any particular histological class, and, although many of our patients with significant proteinuria (NP or NS) presented histologically more serious lesions (pure or mixed proliferation), many of the patients with AUA also presented serious lesions. These figures are in agreement with what has been described in patients with glomerular hematuria and proteinuria <500mg/day, where only 5% of patients have class II lesions being more predominant the most severe forms of lupus nephropathy.18 Furthermore, 15% of biopsies obtained from subjects without clinical manifestations have shown severe nephropathies, class III or IV19 something that we have also found in our series. This is reinforced by the similar distribution of histological classes in patients with severely compromised renal function compared with those with moderate kidney failure.
Additionally, the clinical manifestations of renal involvement of SLE are nonspecific and may be due to other etiologies. Patients with SLE frequently receive anti-inflammatory and immunosuppressant medications, so it is not easy to rule out renal damage secondary to nephrotoxicity or infections.17 It has been reported that in 5% of patients with SLE there may be glomerulopathies not mediated by immune complexes, which can be recognized only with a renal biopsy. Such is the case of minimal changes disease and focal and segmental glomerulosclerosis (currently known as lupus podocytopathies), thin basement membrane disease, amyloidosis or IgM nephropathy; all of them with a therapeutic conflict and a very different prognosis.20,21
Additionally, thrombotic microangiopathy associated with an antiphospholipid syndrome, which can reach a frequency of 30% in patients with SLE, may appear as an isolated form or accompanying the classic lupus nephropathy.22,23 These lesions, not analyzed in our series, can not be diagnosed without a histological study and are important to recognize because they imply an additional treatment to the immunosuppression that includes anticoagulation.17,22
Another interesting aspect to note is that mixed lesions did not show differences in renal function as compared with pure proliferative lesions, suggesting the importance of proliferation in a more aggressive course of the disease. In general, pure proliferative classes and mixed lesions have been recognized as characteristically progressive lesions. Fortunately, in diffuse proliferative lesions the survival of patients has improved from 30% before the 70s to more than 80% in the last two decades, probably due to a better diagnostic accuracy, better availability of dialysis, transplantation and more rational use of immunosuppression.14,24,25
The evaluation of the indexes of histological activity and chronicity of the NIH is an important contribution in the global assessment of lupus nephropathy.15,26 Although it has been questioned by someone, it is still a method used by many groups to guide immunosuppression, avoiding overtreatment of patients with predominantly chronic, non-reversible lesions.27,28 Our results showed higher activity indexes in the more severe lesions (class IV), whether they were pure or mixed, as compared with focal lesions (class III). These indices were not different in global class IV lesions vs. segmental lesions, something that could have been expected given their probable pathogenic and prognostic differences.29
Interestingly, activity rates in 14% of patients with AUA were equal or above the average shown by those with RI, and, were not different in patients with NP vs. RI. Although the highest rates of chronicity were in patients with RI, a 23% of the subjects with AUA showed absolute values equal to or greater than the average in RI patients. It is also remarkable the presence of an activity index of 12 and a chronicity index of 4 in a biopsy from a patient without renal clinical manifestations. All this combination of values illustrates how little is the possibility of establishing a right prognosis of reversibility using exclusively pure clinical expression as an element of judgment.
It must be recognized that the histopathological classification currently in use may suffer from some limitations in terms of precise therapeutic orientation and prognosis.28,30 In fact, it is focused on the glomerular compartment, although the evidence supports the fundamental role that interstitial alterations may have in the prognosis.31,32 In parallel, the recognized effectiveness of mycophenolate as a first-line therapy for all severe forms of lupus nephropathy33 has generated, in some authors, an argument against early biopsy collection. So, there are groups that perform biopsy after completion of induction therapy with this drug proposing that this deferral could better guide the functional prognosis.34
One weakness of our data is how accurate is the estimation of renal function and the possible variation over time. Considering the known limitations of the estimation of the glomerular filtration rate from a single value of serum creatinine reported at the time of requesting the biopsy, it is possible that in the RI group there were cases of acute renal injury mixed up with chronic renal damage. Taking this into account but considering the age of the population studied, changes in renal function could be attributed to the damage associated with lupus itself.
Nowadays, it is widely accepted that a timely renal biopsy in a lupus patient, with any degree of clinical involvement, may play an important role in both diagnosis and management35 and its deferral can affect the therapy, influencing negatively the prognosis.35–37 Unfortunately, we do not have reliable information regarding the evolution period of the renal manifestations prior to renal biopsy.
Our results favor the timely histological documentation of renal involvement in SLE patients. This information may objectively guide the therapy, rule out the presence of coexisting pathologies or other diagnoses and allows the physician to responsibly assume the potential adverse effects of a potent immunosuppression in an attempt to control the disease. Additionally, a full knowledge and classification of the lesions allows to establish the bases to improve the design of more complex studies in the search of better immunosuppression schemes or complementary therapies. At the same time this contributes to the study of tissue biomarkers that help to design personalized therapies and to establish long-term prognosis.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Fulgeri C, Carpio JD, Ardiles L. Lesiones renales en el lupus eritematoso diseminado: ausencia de relación entre datos clínicos e histológicos. Nefrologia. 2018;38:380–387.