To the Editor,
Pregnancy among patients in periodical haemodialysis programmes is still unusual, conception rates ranging between 0.5% and 1.4%, depending on the series.1,2 This is due to a number of factors that decrease fertility and the difficult management of pregnancy during dialysis, owing to scarce experience. However, the most recent studies published by different work groups report encouraging pregnancy success rates of nearly 70%, thanks to improved dialysis techniques and better maternal and foetal care.3-5
We present the case of a 35 year old woman with chronic kidney disease secondary to type 1 membranoproliferative glomerulonephritis, diagnosed by renal biopsy in 1989. She started haemodialysis treatment in June 1994 and underwent cadaver-donor kidney transplantation in December 1995.
On 11 July 2008, she started periodical haemodialysis treatment again due to chronic allograft nephropathy. Co-morbidities included secondary hyperparathyroidism treated with calcitriol, hyperuricaemia treated with alopurinol, anaemia treated with subcutaneous epoetin alfa and oral iron supplements, and hypertension treated with calcium channel blockers, beta blockers and diuretics.
When the patient returned to dialysis, immunosuppressant drugs were gradually discontinued (steroids, calcineurin inhibitors and mycophenolate mofetil) and the patient was transitioned to darbepoetin alfa and intravenous iron supplements Transfusion of 2 units of red blood cells was necessary during the first session due to anaemia with a haemoglobin level of 6.9g.
On 13 August 2008 the patient reported amenorrhea lasting 2 months. Human chorionic gonadotropin-beta levels and a vaginal ultrasound revealed that she was approximately 4 weeks pregnant. In light of these results, the patient started a daily haemodialysis programme.
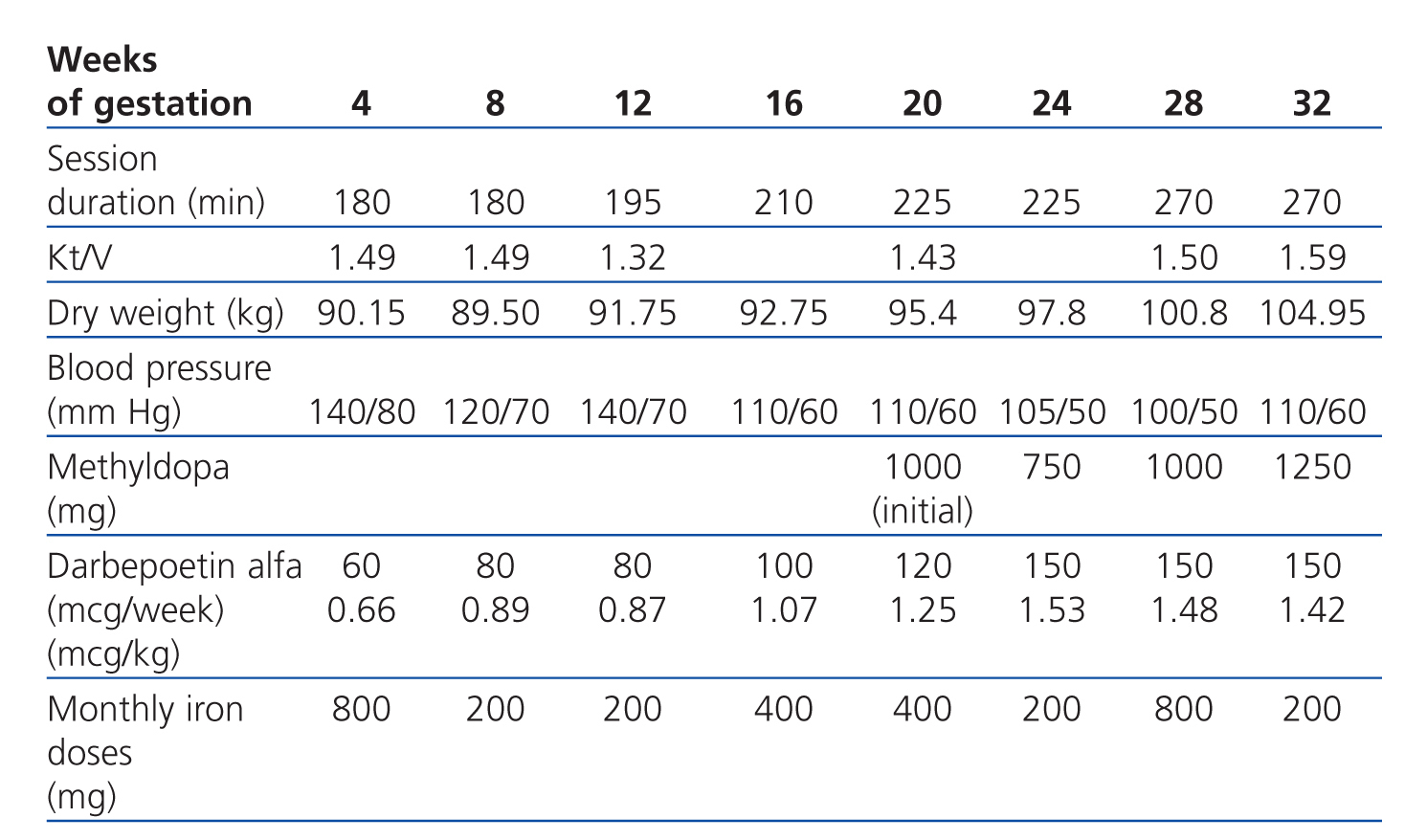
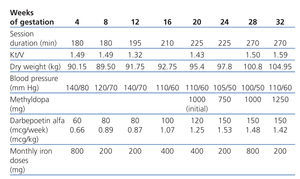
For the dialyser, we used a high-flux polyethersulfone membrane with a surface area of 2.1m2 and a dialysate bath consisting of 3mEq/l calcium and 1.5mEq/l K+, although this was adjusted depending on laboratory results. Dalteparin was administered as an anticoagulant agent at an initial dose of 2500IU per session. Dry weight was adjusted by applying increases of 0.3-0.5kg per week from the second trimester.
Regarding drug treatment, tacrolimus was suspended and steroid and calcitriol treatments were maintained. We did not continue to decrease steroids in order to prevent graft rejection, using a dose of 5mg daily. The diuretic agents and the calcium channel blocker were suspended and folic acid and Vitamin B complex were increased. No oral iodine supplement was prescribed due to lack of experience and because it is excreted in urine. The patient received no zinc supplements because plasma levels were normal (Table 1).
The patient was placed on an unrestricted diet.
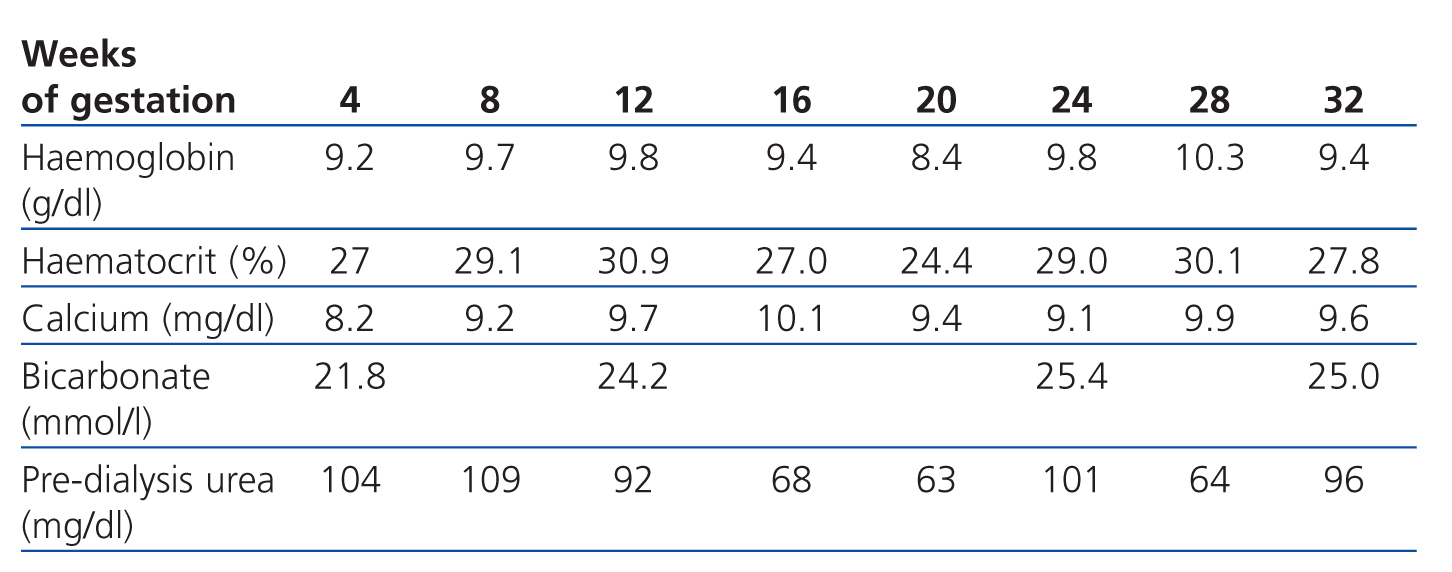
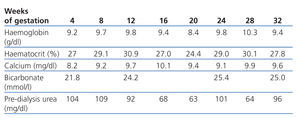
Our goal was to maintain pre-dialysis urea levels below 100mg/dl and normal calcium levels, prevent acid-base balance disorders and maintain haemoglobin levels at approximately 11g/dl. The dialysis dose that we established as our target was an equivalent renal urea clearance of 30ml/min according to the Casino and López method.
In this case, evolution of anaemia during the pregnancy was slow, and maintaining haemoglobin levels above 9g/dl was difficult. The patient required 2 units of red blood cells in week 21 (Table 2).
Session duration was gradually increased from an initial length of 3 hours. After gestation, the patient’s programme consisted of 6 weekly sessions of 4.5 hours (27 hours weekly). No relevant episodes of hypotension were recorded and the maximum ultrafiltration rate was 500ml/h.
It was necessary to increase the anticoagulant dose to 5000IU dalteparin per session. The only incident was graft thrombosis in week 24 and doctors placed a jugular tunnelled catheter so as not to expose the patient to thrombolytic drugs or excessive radioscopy.
Regarding obstetrics, hypertension was recorded in week 16, and treatment with methyldopa was initiated. Polyhydramnios and foetal macrosomia were detected in week 20. In week 32, the patient was referred to a tertiary hospital for assessment and to end the pregnancy due to the foetal monitor showing poor responsiveness. A planned caesarean section was performed 2 weeks later, delivering a live newborn weighing 2200g.
Available information on managing pregnant patients in haemodialysis programmes is scarce; it is mainly based on small cohorts and a handful of meta-analyses published by different hospitals around the world. This highlights the need for creating national and international registers.6 In these series, we observed a proportional relationship between the time the patient remained on dialysis and the prognosis for the pregnancy. In general, programmes of more than 20 hours weekly are related to longer gestational periods and decreased risk of preterm birth.6 It also seems that increasing dialysis frequency to a programme of 6 weekly sessions is better than scheduling longer, less frequent sessions.
One hypothesis is that prolonging dialysis sessions may decrease risk of polyhydramnios and foetal distress and improve the patient’s nutritional state, thereby decreasing the risk of preterm birth.7
Anaemia, another chronic complication of patients on dialysis, must be monitored and treated with iron and folic acid supplements, and erythropoietin treatment must be increased in order to maintain haemoglobin levels at 10g/dl and transferrin saturation above 30%.8
Although there is still very little available information on the management of these patients, the most recent published results give us a more optimistic view.
Conflicts of interest
The authors affirm that they have no conflicts of interest related to the content of this article.
Table 1. Haemodialysis and treatment parameters
Table 2. Laboratory data