Autosomal dominant polycystic kidney disease (ADPKD, Q61.2) is a multisystemic genetic pathology with renal impact; half of the affected patients require renal replacement therapy (RRT) and almost all die with some degree of renal failure. It is the sixth leading cause of renal failure in Spain and it causes personal and family suffering, decreases life expectancy and increases healthcare costs.1 His approach has focused on the treatment of complications and the need for RRT. For a few years we have had tolvaptan,2 a drug capable of slowing the progression of renal cystogenesis.
Since 2009, our group has studied its impact3 and recommends an approach based on prevention: to encourage couples with affected members to have healthy children who do not transmit the disease. This simple concept was presented in the Parliament of Andalusia in 2016 by the Granada parliamentarian Mrs. Olga Manzano Pérez through Proposition Not of Law:10-16 / PNLC-000248 for the creation of the Primary Prevention Plan of the ADPKD, following the proposition of our Study Group of Autosomal Dominant Polycystic Disease (SGADPD)and the Friends of the Kidney Association, which was unanimously approved.4 Numerous drawbacks make this initiative difficult: members within the affected families that have not been studied (5 on average), late diagnosis of the disease (34 years old on average) and in 56% of non-diagnosed parents cases have already had a first child, patients who abandon medical follow-up, ignorance on the part of the medical staff who care for these patients of assisted human reproduction techniques (HRT) to prevent the transmission of the disease such as gamete donation and the preimplantation genetic testing (PGT) and a remiss Public Health to make accessible this last reproductive option.3
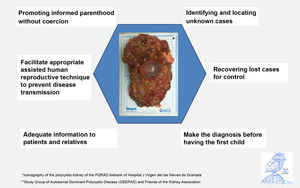
Our proposal, summarized in Fig. 1 using the Preventive Hexagon, is based on: a) locating unknown cases within affected families, b) recovering lost cases for follow up and control, c) diagnosing the disease before having the first child d) adequate information to patients and relatives, e) facilitate the most appropriate reproductive options for each couple aiming to have healthy children and f) promote informed parenthood without coercion.3 To carry out these measures, four basic tools are needed: a) involve doctors in prevention, especially those in Primary Health Care, b) have a registry of patients and family trees, c) favor the performance of genetic studies, and the pathogenic variant should be identified in each family, and d) facilitate the availability of human reproductive techniques to prevent its transmission.
The doctor taking care of these patients, in addition to solving the specific questions of each individual, must address how to reduce the health impact of the disease; this should be done by providing information to individual and family, recommending to study those members of affected families not yet evaluated and persuading those who left our consultations to come back to the clinics.
Patient registration and pedigree trees are necessary and the analysis of this information will allow us to: a) know the general magnitude of the disease within a given healthcare setting and in each family, b) its geographical distribution5, c) the evolution of incidence and prevalenced) identify and locate non-studied cases, which in our province, Granada, with a population of 914,678 inhabitants, should range between 198–240 people. That is, between 0.021−0.026% of the Granada population has the disease and it is not aware. By doing genogram analysis we have identified in Granada four “hot” spot of ADPKD: Vega Granada with 345 identified cases, Costa Tropical with 160 cases, Loja with 158 cases and Alpujarra with 78 cases. In Granada the prevalence ranges between 1/1003-1/1527. Between 2010–2019 we have diagnosed 16–29 cases/year, with an average of 23 cases/year, which yields an incidence of 2.35–3.05 cases/100,000 inhabitants. If on average six patients die/year, there is an average annual increase of 17 patients. Our informative intervention has managed to reduce the mean age of diagnosis, and in 2019 it was 24 years.3
The study of the disease is based on the knowledge of its genes, PKD1, PKD2, GANAB, DNAJB11, among others. In our serie, the renal survival of patients who carry pathogenic variants in the PKD1 gene is 56 years as average, compared to 70 years for those who carry pathogenic variants in the PKD2 gene. Women need RRT later than men: 59 vs. 53 years respectively on average, p < 0.01.2 The genetic study helps to establish the diagnosis when the imaging techniques are not yet conclusive and makes the diagnoses both affected people who are in the initial stages of the disease and those in RRT. Not all the variants associated with ADPKD have the same prognosis, and we refer to truncating and non-truncating, although in this sense there is still much to clarify. Many of these pathogenic variants, not yet described (58% in PKD1 and 60% in PKD2 in our registry), must be reported, especially if PGT6 is to be used.
Couples on reproductive age who wish to have offspring must know the risk of having children in a natural way: autosomal dominant inheritance conditions a 50% probability of conceiving a healthy or affectionate child, and this rule of chance applies on each occasion. It is convenient to inform them about the HRT techniques that offer the advantage of conceiving healthy children, who will no longer transmit the disease. Public Health Systems (SSP) must be sensitive and understand the needs of these patients and families and, just as dialysis and transplantation are facilitated, it must also provide primary prevention measures, which will contribute to a decrease in prevalence and incidence of this disease in two or three generations. It is not about eradicating the disease, since there will always be de novo variants on which we can hardly influence, but we can reduce its frequency. Lastly, reorienting the focus towards primary prevention also has its economic advantages since it is a cost-efficient model and its implementation will make the health system more sustainable.7
FinancingRafael J. Esteban de la Rosa and Antonio M. Poyatos Andújar have participated in training talks on PQRAD financed by Otsuka.
Conflict of interestsThe authors declare that they have no conflict of interest.
Please cite this article as: Esteban de la Rosa RJ, Poyatos Andújar AM, Morales García AI, Martínez Navarro L, García Valverde M, Bravo Soto JA. Proyecto preventivo de la enfermedad poliquistosis renal autosómica dominante (PQRAD). Nefrologia. 2022;41:704–706.