The activated partial thromboplastin time (aPTT) is the time required for fibrin to appear after mixing plasma with substitutes for platelet phospholipids. It is used as screening for alteration in the intrinsic and common coagulation pathways, to monitor treatment with heparin sodium and to determine the presence of lupus anticoagulant.1,2
Coagulation abnormalities are often due to pre-analysis anomalies3 or incorrect sample collection. Moreover, aPTT is prolonged with deficiency of the coagulation factors involved, with lupus anticoagulant or in heparin therapy.
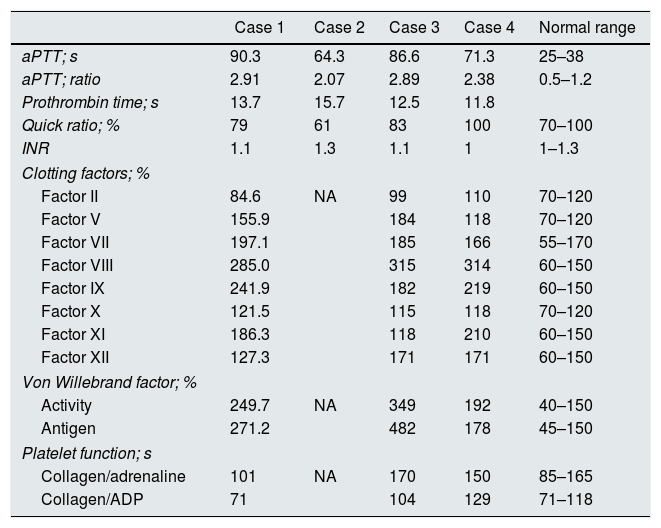
We present the cases of two patients on peritoneal dialysis (PD) (cases 1 and 2) who, on the day they were called to receive a kidney transplant, were found to have prolongation of aPTT. In both patients, four repeat coagulation tests were similar (Table 1). Possible interference was ruled out, as well as poor sample extraction. After assessment by haematology, two vials of prothrombin complex and one bag of fresh frozen plasma were administered, with no change in the intrinsic pathway values, so they were rejected for kidney transplantation. Reviewing the patients from our unit, we found that cases 3 and 4 had the same problem. All the cases were male, aged from 53 to 69, on continuous ambulatory PD with a nocturnal exchange of Extraneal. Patient 2 was being treated with acenocoumarol and received vitamin K to reverse its effect; the rest of the patients had no previous history or medication that interfered with coagulation.
Coagulation study performed with Sysmex CS-5100 automated analyser, with Pathromtin® reagent.
| Case 1 | Case 2 | Case 3 | Case 4 | Normal range | |
|---|---|---|---|---|---|
| aPTT; s | 90.3 | 64.3 | 86.6 | 71.3 | 25–38 |
| aPTT; ratio | 2.91 | 2.07 | 2.89 | 2.38 | 0.5–1.2 |
| Prothrombin time; s | 13.7 | 15.7 | 12.5 | 11.8 | |
| Quick ratio; % | 79 | 61 | 83 | 100 | 70–100 |
| INR | 1.1 | 1.3 | 1.1 | 1 | 1–1.3 |
| Clotting factors; % | |||||
| Factor II | 84.6 | NA | 99 | 110 | 70–120 |
| Factor V | 155.9 | 184 | 118 | 70–120 | |
| Factor VII | 197.1 | 185 | 166 | 55–170 | |
| Factor VIII | 285.0 | 315 | 314 | 60–150 | |
| Factor IX | 241.9 | 182 | 219 | 60–150 | |
| Factor X | 121.5 | 115 | 118 | 70–120 | |
| Factor XI | 186.3 | 118 | 210 | 60–150 | |
| Factor XII | 127.3 | 171 | 171 | 60–150 | |
| Von Willebrand factor; % | |||||
| Activity | 249.7 | NA | 349 | 192 | 40–150 |
| Antigen | 271.2 | 482 | 178 | 45–150 | |
| Platelet function; s | |||||
| Collagen/adrenaline | 101 | NA | 170 | 150 | 85–165 |
| Collagen/ADP | 71 | 104 | 129 | 71–118 | |
ADP: adenosine diphosphate; aPTT: activated partial thromboplastin time; INR: international normalised ratio; NA: not available.
After exhaustive study, coagulation abnormalities inherent to each patient were ruled out (Table 1). The coagulation study was performed with a Sysmex CS-5100 automated coagulation analyser, with Pathromtin® SL as reagent. When the reagent was changed to Actin® FS (Siemens, Marburg, Germany), the aPTT returned to normal. Samples from the affected patients had been saved, verifying the normality of the intrinsic pathway with the new reagent.
In the cases presented, there was interference from the laboratory reagent causing the false lengthening of the aPTT. It is interesting the fact that we have found several cases, all of them in PD patients. After studying 70 haemodialysis patients and 80 patients with chronic kidney disease (CKD) stages G3b, 4 and 5, we found no patients on haemodialysis or with CKD with the same interference.
Patients on PD with icodextrin are known to be subject to laboratory interferences: there may be falsely elevated blood glucose readings4 and measured plasma amylase levels decrease.5 However, to date, no interference with coagulation tests has been reported.
The only common factor found in our series is continuous ambulatory PD and treatment with icodextrin, although it is true that not all patients with icodextrin have had the same interference.
Reagents for measuring aPTT are composed of an activator and phospholipids (of synthetic or animal origin). The sensitivity and specificity for aPTT reagents to heparin and coagulation factors vary depending on their origin, their properties and the total concentration of phospholipids, even if the same activator is used.6,7 The reagents used by our laboratory are differentiated according to the surface activator they use: silicon dioxide (Pathromtin® SL) or ellagic acid (Dade Actin® FS). They have a strong correlation coefficient, but comparability is low.8 Our hypothesis is that the metabolites of icodextrin can competitively interfere in the binding of silicon dioxide surface activator, preventing the correct reading of the aPTT.
We would like to inform all nephrologists of this situation, as the laboratory interference had significant repercussions: cases 1 and 2 could not have a kidney transplant until the complete coagulation study was performed.
Nephrologists therefore need to be aware that a prolonged aPTT may indicate the existence of a bleeding disorder, be associated with an increased risk of thrombosis (due to lupus anticoagulant) or lack any thrombotic or haemorrhagic implications (in patients with PD it may be the result of interference with the laboratory reagent).
Please cite this article as: Santos García A, Millán del Valle I, Cruzado Vega L, Ruiz Ferrús R, Tordera Fuentes D, Sabater Belmar A, et al. Tiempo parcial de tromboplastina activado prolongado sin coagulopatía subyacente en diálisis peritoneal. Nefrologia. 2019;39:210–211.