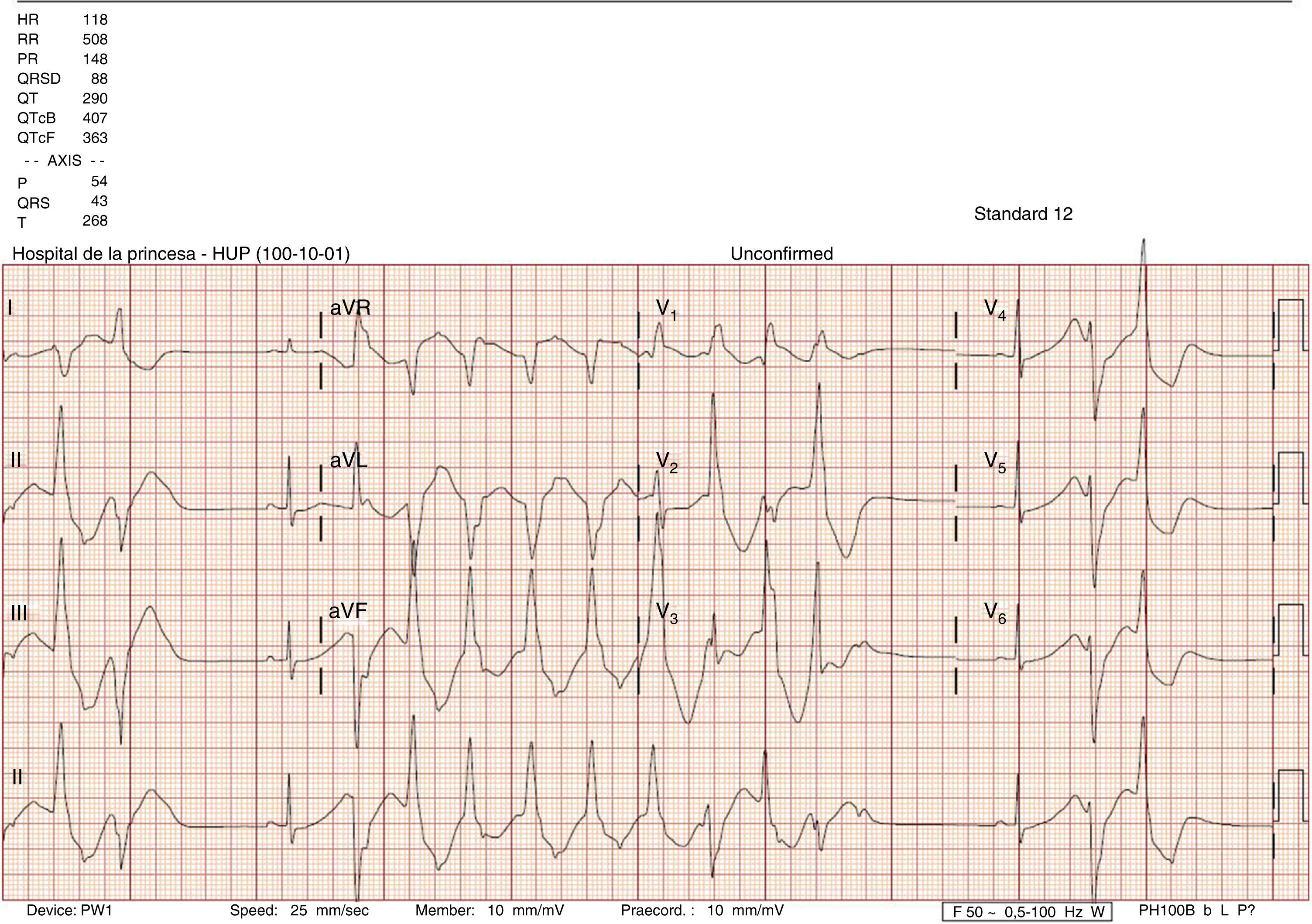
This was a case of a 25-year-old woman with no known allergies or relevant medical history and no toxic habits. She is an attorney and drinks 500–750ml of beverages containing taurine and 1l of caffeinated soda per day due to stress. The following details were observed: height 170cm, weight 58kg and BMI 20. She was admitted to the hospital for headache and tachycardia during the last two days after she did some sports and coinciding with an increase in the consumption of a beverages containing taurine. She denied chest pain or dyspnoea. Had no vomiting or diarrhoea and had no change in diuresis. She did not consume herbal products, drugs, teas, diuretics, liquorice or alcohol. Physical examine: Conscious, oriented, blood pressure 108/86, heart rate 110 beats per minute. Afebrile. Anodyne cardiopulmonary auscultation. Rest of the examination was normal. Blood test: normal red cell count, no elevation of cardiac or hepatic enzymes and coagulation test without alterations; creatinine 1.04mg/dL, urea 31mg/dL, potassium 1.73mEq/L, sodium 134mEq/L, magnesium 2.2mg/dL, chloride 85mEq/L, Albumin 4g/dL. Arterial blood gas: Ph 7.580, PCO2 46mmHg, PO2 86mmHg, bicarbonate 43.1mmol/L. Plasma anion gap (AG): 5.9mEq/L. Urine: chloride 22.2mEq/L, potassium 68.28mEq/L, sodium 210mmol/L, urea 920mg/dL, creatinine 192.72mg/dL, glucose 15mg/dL. Urine anion gap: 256mEq/L. Plasma osmolality: 278.2mOsm/L. Urine osmolality: 573.3mOsm/l. Transtubular potassium gradient: 15. Cortisol at 8am and aldosterone in supine position were within the normal range. No alterations in urinary sediment. ECG: sinus rhythm, markedly enlarged QT (580ms; corrected 700ms); with frequent polymorphic ventricular tachycardia (Fig. 1). An infusion of CLK was initiated via central line: 80mEq within two hours and maintained with an infusion of 120mEq/day. After 18h, urine test was: sodium 25.3mEq/L, potassium 6.2mEq/L; in serum: sodium 142mEq/L, potassium 2.8mEq/L transtubular potassium gradient: 4. Venous blood gas: Ph 7.380, PCO2 52mmHg, HCO3 30.8. Blood test at discharge: sodium 143mEq/L, potassium 4.84mEq/L, chloride 105m Eq/L, pH 7.380, pCO2 49mmHg, bicarbonate 29mmol/L. Urine: potassium 11.59mmol/L, sodium 89mmol/L, creatinine 266.27mg/dL, urea 642mg/dL. The ECG was normal. The evolution of ions in the urine suggested the presence of a diuretic substance that was suspended at admission. Diagnoses: hypokalaemia due to diuretic substances: taurine and caffeine, but not being able to rule out the presence of other diuretics, aggravated by the increase of insensitive losses and alkalemic state. A Bartter vs Gitelman-type tubulopathy was ruled out given the evolution of the ions in urine and the hormonal axis normality. Alteration in heart conduction due to hypokalaemia. Mixed alkalaemia: Chloride-resistant metabolic alkalosis due to diuretic substances and reactive respiratory alkalosis.
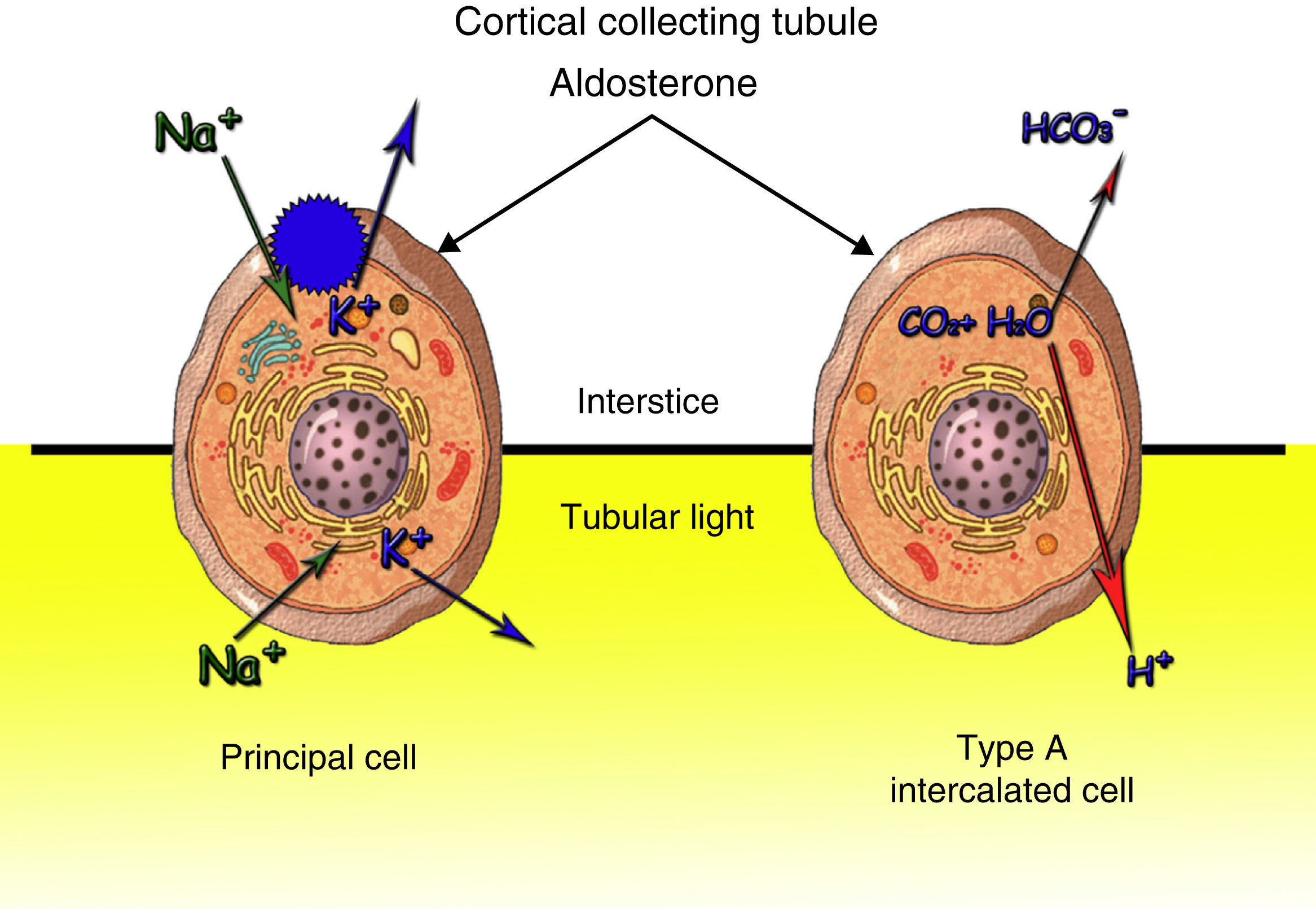
Ninety percent of the potassium filtered at glomerular level is reabsorbed in the proximal tube. The distal tubule, by effect of the aldosterone, regulates the urinary output according to the body needs (Fig. 2). Distal excretion of Kis modified by the amount of fluid, distal contribution of sodium, mineral corticoids and excretion of non-reabsorbable anions. The most common cause of hypokalaemia due to renal losses are non-potassium-sparing and similar diuretics. Hereditary tubulopathies (Bartter and Gitelman) may not be distinguishable from the intake of diuretics. Hyperaldosteronism and hypermineralcorticoidism cause hypokalaemia due to their action on the distal nephron. Potassium is a predominant intracellular cation. The best marker to assess the renal management of potassium is TTKG in euvolemia that assesses the mineralocorticoid action on the distal nephron: values <4 indicates absence and >7 presence of activity. Blood pressure, extrarenal losses, acid-base state, urinary ions and urine and plasma AG have to be assessed. In the presence of metabolic alkalosis, as in our case, chloride concentration decreases in order to compensate for the elevation of bicarbonate and the AG increases in proportion to the alkalosis severity, due to the lactate and the concentration of more anionic serum proteins. In turn, the kidney tends to increase the excretion of bicarbonate at proximal and distal tube level where there is a Cl−/HCO3 exchange in the beta-intercalated cells of the collecting tubule. Serious chloride, potassium or extracellular space depletion inhibits this exchange. Urine AG is an indicator of urinary acidification. Positive values indicate that renal acidification is intact. Treatment must be oral. The intravenous line is reserved for serious hypokalaemia (K<2.5mEq/l), arrhythmia, acute myocardial infarction or digitalisation.1
Some characteristics of the energy drinks diuretic components:
CaffeineNatural xanthine. Energy drinks have levels between 75 and 174mg per serving, others exceed 500mg.2 This stimulates the central nervous system, cardiovascular system, and central respiratory system; it relaxes the smooth bronchial muscle and striated muscle, increases acid gastric secretion and renal blood fluid and has diuretic properties. Many of these effects are caused by antagonic action on adenosine receptors.3 It is rapidly distributed through the organism and crosses the placental and blood–brain barrier. It has hepatic metabolism (cytochrome P-450). It is clinically used as a respiratory stimulant in newborns with apnoea of prematurity. The adverse effects include insomnia, agitation, headache and tachycardia at elevated doses.4 The changes in blood pressure response are not conclusive.5 It can produce dependence syndrome.6
TaurineConditionally essential amino acid. Its deficit is associated with cardiomyopathy, retinal degeneration and failure to thrive.7 Metabolic actions include: bile acids conjugation, osmolar regulation, detoxification, membranes stabilisation and modification of cellular sodium and calcium levels. It has positive inotropic action and protects the cardiac membrane from the adverse effects of hyperglycaemia.7,8 Its renalprotective effect is caused by its antioxidant action by controlling the effects generated by TGF-B1 and type I and IV collagen.8 It increases the glomerular filtration rate, reduces sodium tubular reabsorption, reduces urine protein and inhibits antidiuretic hormone production.8,9 Clinically, it has been used in hypercholesterolaemia, epilepsy, cardiopathy, retinal macular degeneration, Alzheimer's disease, cystic fibrosis and hepatic diseases.10
Both of them increase natriuresis increasing the arrival of sodium at the distal tubule: activating aldosterone and producing the entry of cellular sodium and the exit of potassium to the tubular light causing hypokalaemia.
Most of the supplements contained in the energy drinks have concentrations below the amounts associated with adverse effects.4 The association of heart conduction alterations is not clear with studies for and against them.11,12 The combination of these drinks with alcohol may cause arrhythmias in subjects prone to them.13
Please cite this article as: Salanova-Villanueva L, Bernis-Carro C, Alberto-Blazquez L, Sanchez-Tomero JA. Arritmia cardiaca grave por hipopotasemia. Influencia de las sustancias diuréticas. Nefrologia. 2015;35:334–336.