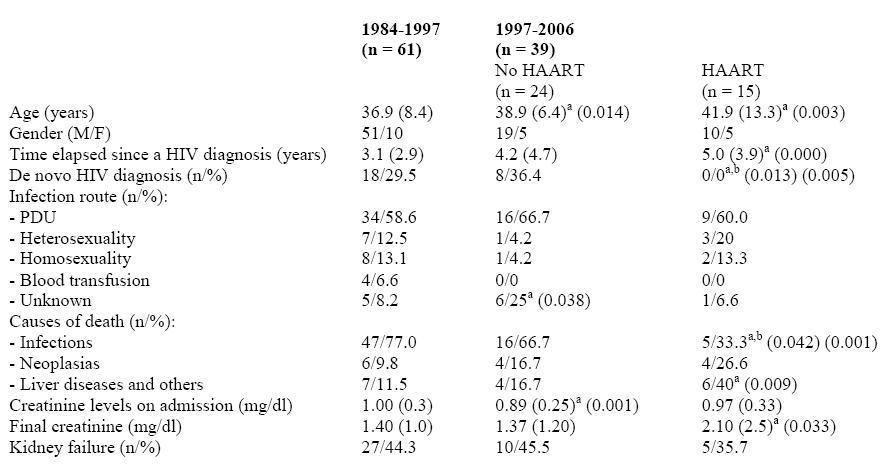Fundamento: El objetivo del estudio es analizar la evolución de las lesiones anatomopatológicas renales en VIH que hubiesen recibido o no medicación antirretroviral de alta actividad (TARGA). Sujetos y métodos: Se revisaron las historias clínicas de 100 pacientes fallecidos entre 1984 y 2006, con registros clínico-analíticos y muestras anatomopatológicas. Sesenta y uno habían fallecido antes de 1997 (grupo I) y 39 pacientes después, de los cuales 24 no habían recibido TARGA (grupo II) y 15 sí (grupo III). Las muestras renales se tiñeron con hematoxilina-eosina, PAS, tricrómico de Masson y plata-meteramina. Se registraron para todos los pacientes los diagnósticos anatomopatológicos finales, así como las lesiones a cada uno de los tres niveles: glomérulo, túbulo e intersticio. Se definió NAVIH como la presencia de glomerulosclerosis segmentaria y focal, con colapso glomerular y lesiones microquísticas túbulo-intersticiales. Resultados: Las principales causas de muerte fueron infecciones (68%) o tumores (14%), y el resto (18%) fueron otras causas, especialmente hepatopatías. Un 42% de los individuos presentaban insuficiencia renal en el momento del fallecimiento. En los tres grupos de estudio predominaban las lesiones tubulares, seguidas de las lesiones intersticiales y de las glomerulares. Cuando se compararon aquellos sujetos en tratamiento con TARGA con aquellos sin tratamiento, encontramos un porcentaje significativamente mayor de lesiones en el intersticio en el grupo con TARGA. En este grupo hubo también más casos de necrosis tubular aguda NTA, si bien estas diferencias no fueron estadísticamente significativas. Conclusiones: Las lesiones renales son frecuentes en pacientes afectos de VIH en el momento de la muerte, independientemente del período de estudio considerado y del tratamiento recibido.
Background:The aim of the present study is to analyze the impact of high activity antiretroviral therapy (HAART) on renal lesions observed in autopsies of HIV patients. Subjects and methods: Clinical records and renal pathologic samples from 100 HIV patients, who had died between 1984 and 2006, were reviewed, 61 before 1997 (group I) and 39 after. 24 of them had not received HAART (group II) and 15 had (group III). Premortem clinical and analytical data were obtained. Renal samples were stained with hematoxilin-eosin, PAS, Masson trichrome and silver-methenamine. The final pathologic diagnosis was recorded along with the findings at glomerular, tubular and interstitial levels. HIVAN was defined as the presence of focal or segmental glomerulosclerosis with glomerular collapse and microcystic tubulo-interstitial lesions. Results: The main causes of death were infections 68%, tumours 14%, and others 18%, especially liver diseases. Renal failure was present in 42% at the time of death. A predominance of tubular lesions exists in the three study groups, followed by interstitial lesions and glomerular lesions. The main diagnoses were acute tubular necrosis (ATN) and septic nephritis. Four cases of HIVAN were found. When the subjects who received HAART treatment were compared with those who did not, a significantly higher percentage of interstitial lesions in the group with HAART was observed. There were also more cases of acute tubular necrosis but these differences were not statistically significant. Conclusions: Renal lesions were frequent in HIV patients at the time of death independent of the study period and the treatment received
INTRODUCTION
Renal disease in patients with human immunodeficiency virus (HIV) includes not only HIV-related kidney disease but also immune complex disease, nephrotoxicity associated with medicines and nephrotoxicity linked with comorbidities, such as diabetes, hypertension and coinfection with hepatitis B and C.1-5 While its relevance is not indicated in clinical studies, given that it is presented together with other processes, its rate of recurrence is high in autopsy findings, ranging between 30% and 80%, with a predominance of tubulointerstitial lesions.6-16 In Spain, the two studies conducted so far included only patients in the period prior to receiving a highly active antiretroviral therapy (pre-HAART). Although these studies displayed different rates of disease (53.8 and 93.4%, respectively), they both showed a predominance of tubulointerstitial lesions.12,17
The introduction of the highly active antiretroviral therapy (HAART) has succeeded in increasing the survival rate of patients with HIV. This has led to the emergence of new diseases and complications, related to both HIV infection and treatment,18-21 in addition to a change in the causes of death. The aim of this study is to make a temporal evaluation of renal disease in the autopsies of patients with HIV, including patients in pre- and post-HAART.
MATERIALS AND METHODS
This is a retrospective study of 100 autopsies of patients with HIV, who died between 1984 and 2006 in a tertiary hospital in Valencia.
The deceased were temporarily grouped according to the year of death: 61 (group I), who died before 1997 (pre-HAART period), and 39 (group II), who died after 1997. In the latter group, the patients were further classified according to whether or not they had received HAART: 24 had not been treated with HAART (group II) and 15 had (group III), depending on the availability of such treatment at that time.
The patients of group I, that is, those who passed away prior to 1997 (pre-HAART period), correspond to the study of an autopsy cohort published by our team, which had been previously carried out in our hospital.17 We have therefore used the same methodology for the analysis of the patients in groups II and III (HAART period) in order to make comparisons and to be able to get an overview of the progression of renal disease in patients with HIV in both pre- and HAART periods.
Therefore, following the same methodology as in the previous study,17 we collected retrospectively from patient records the following data: age at the time of diagnosis of HIV infection, age at the time of death, causes of death, route of infection, antiretroviral therapy and renal function assessed by the levels of creatinine on admission and at the time of death. We considered as kidney failure serum creatinine levels greater than 1.3 or 1.2 mg/dl for men and women respectively, and as HAART therapy the treatment that combined at least three or more drugs. We identified three main causes of death: infection, neoplasia and others, including liver disease.
For the study of autopsy kidney specimens, we followed the same methodology described in the previous study.17 Kidney samples were placed in 4% neutral formaldehyde, embedded in paraffin and stained with haematoxylin-eosin, PAS, Masson’s trichromic staining and silver-methenamine. We considered separately the lesions to different kidney structures: glomerular, tubular and interstitial. We later made a renal anatomopathological diagnosis based on the findings at various levels and the clinical information. We considered as septic nephritis the acute suppurative interstitial inflammation caused by infection. In these cases, there is a limited leukocyte infiltration to the interstitial tissue. This corresponds to the initial stages of acute pyelonephritis, where scattered foci of interstitial suppurative inflammation can be found, either as independent focal abscesses or as wedge-shaped suppurative areas that tend to coalesce. HIV-associated nephropathy (HIVAN) was defined as the presence of collapsing focal segmental glomerulosclerosis, associated with the presence of tubular microcystic dilatations.22
Statistical analysis
The numeric variables are expressed as the mean and the standard deviation. The prevalence of each of the anatomopathological lesions is expressed in both absolute numbers and percentages. To compare the variables between groups, we applied the ANOVA test for quantitative variables and the chi-square test for qualitative variables, using the statistical program SPSS version 12 for Windows (SPSS Inc. Headquarters, 233 S. Wacker Drive, 11th floor, Chicago, Illinois).
RESULTS
General characteristics
Table 1 shows the clinical characteristics of the sample. Our population consists of only white HIV patients in stages C2 and C3 (95%). There is a predominance of male patients in the three groups (78%), with an average age of 36.5 years. In this regard, it should be noted that the patients in group III, who had received HAART, were older at the time of death (an average of 41.9 years compared with 36.9 years, p = 0.003) and that the illness had evolved over a longer period of time (an average of 5 years compared with 3.1, p < 0.001). The most common route of infection (59%) in the three groups was PDU (Parenteral Drug User). Moreover, the most common cause of death in patients without HAART was infection (groups I and II). On the other hand, the most common causes of death in those who received HAART (group III) were liver diseases and others (40% compared with 11.5%, p = 0.009), although there was a higher rate of neoplastic disease as a cause of death (26.6% compared with 9.8%). Finally, the mean values of creatinine at the time of death were significantly higher in group III (2.10 compared to 1.40, p = 0.033), although there was no difference in the percentage of patients with kidney failure in any of the three groups.
Renal disease
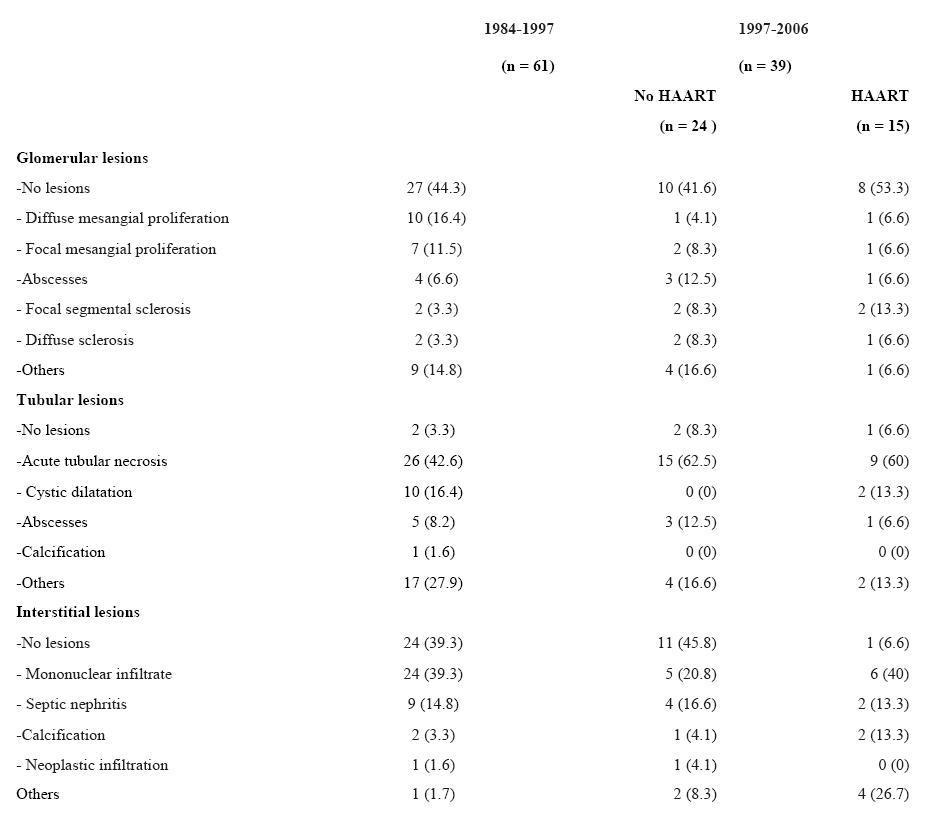
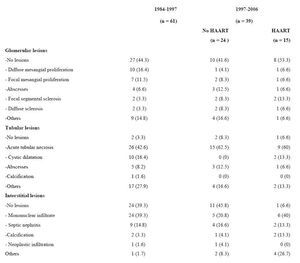
Table 2 shows the morphological alterations found in the histopathological examination of the kidney during autopsy. In all three groups, there is a predominance of tubular lesions, followed by interstitial and glomerular lesions.
The percentage of tubular lesions was very high in all three groups (96.7%, 91.6% and 93.2% respectively), while the most common lesion in all the groups was acute tubular necrosis (42.6%, 62.5% and 60%, respectively), followed by abscesses (8.2%, 12.5% and 6.6%, respectively).
The main interstitial lesion in the three groups was mononuclear infiltrate, followed by septic nephritis and the presence of calcifications.
Concerning glomerular lesions, the results were very different. In the first group, the findings indicate mesangial disease (defined as mesangial cell proliferation or matrix increase, without alterations in the capillary wall, or epithelial cell proliferation or subendothelial or extramembranous deposits). It was present in almost one third of the samples. However, in the other groups there was a clear predominance of one glomerular lesion over the others.
When comparing the patients in group III with the rest, there were no significant differences in the prevalence of glomerular and tubular lesions. On the other hand, there were significant differences concerning interstitial lesions. There was a considerably higher percentage of lesions at this level in the patients treated with HAART (93.3% compared to 60.7%, p = 0.015), with a higher incidence rate of mononuclear infiltration, calcifications and other lesions.
Diagnoses of renal lesions
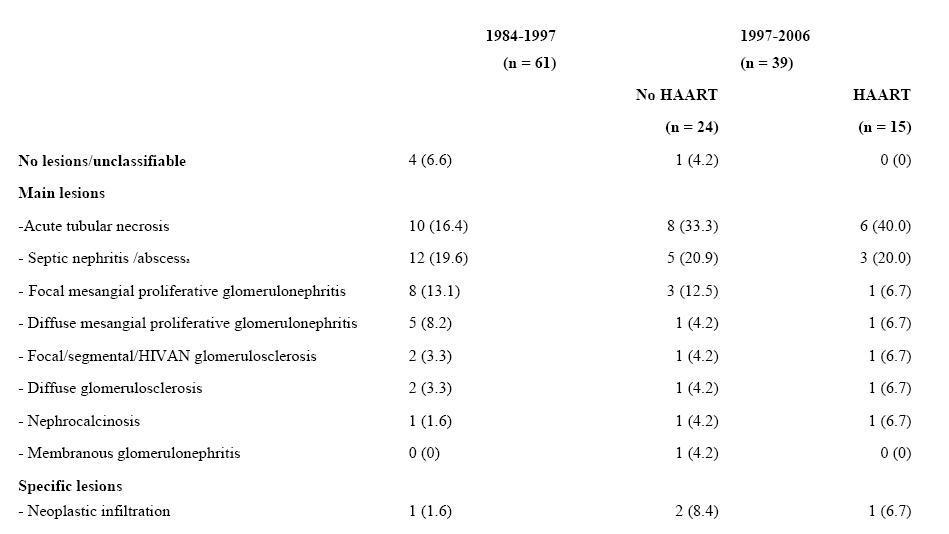
The primary final diagnoses are shown in Table 3. In group I (pre-HAART), there was a predominance of infections (septic nephritis, which also includes abscesses, miliary tuberculosis and fungal infiltration) and acute tubular necrosis was the second diagnosis in order of frequency. However, in groups II and III, the primary final diagnosis was Acute Tubular Necrosis (ATN), followed by septic nephritis. Four cases were compatible with HIV-associated nephropathy (HIVAN), two in group I, one in group II and one in group III. It should be noted that none of the four patients were black. In addition, the typical initial progression of the renal function impairment with proteinuria only took place in group III, having diagnosed HIVAN by means of a renal biopsy. Only this patient received antiretroviral treatment with zidovudine, lamivudine and saquinavir, corticosteroids and angiotensin-converting enzyme inhibitors (ACE inhibitors), which improved and stabilise the patient’s condition and prolonged their survival. In the end, the patient required peritoneal dialysis and died more than a year after the start of the dialysis due to panlobular pneumonia. In the remaining patients, the diagnosis was made following the results of the renal autopsy samples.
The patients treated with HAART had a higher prevalence of ATN, although the difference was not statistically significant (21.2% in patients without treatment [groups I and II] compared to 40% in patients treated with HAART [group III] respectively; p = 0,116).
DISCUSSION
The results of this study confirm the high prevalence of renal anatomopathological lesions in patients with HIV, regardless of the study period considered and the treatment received. Furthermore, they highlight their heterogeneity, being tubulo-interstitial lesions more frequent than the glomerular ones. The same distribution of lesions has already been described in other previously published studies, corresponding mainly to patients in pre-HAART who have not received antiretroviral treatment.6-17 The high prevalence of PDU as an HIV transmission route, the frequent coinfection with the hepatitis C virus (HCV) – present in 73.7% of the total sample – and the advanced stage of HIV infection could explain the high prevalence of renal disease found in our study, which was superior to that reported in other studies.7,13-15 However, this histopathological damage is hard to identify in the analysis, since the creatinine levels on admission had a mean of 1.00 (0.3), 0.89 (0.25) and 0.97 (0.33) in the three groups, respectively. This was already described in the Spanish study by Martinez et al.,12 in which only 10% of the patients had a clinical correlation. Similarly, in the study by Wyatt et al.,30 in which they used the glomerular filtration and proteinuria rates (more specific than creatinine) to define chronic renal failure, more than half the renal pathology observed could have been overlooked.
The creatinine values at the time of death were considerably higher than the values shown on admission: 1.40 (0.1), 1.37 (1.20), 2.10, and (2.5) for each of the groups, respectively. This meant that approximately 40% of the patients in each group had acute kidney failure during hospital admission. Thus, additional factors were involved other than direct complications of HIV infection, such as the haemodynamic status and the use of nephrotoxic medicines. In this regard, it should be noted that the creatinine levels at the time of death of the patients in group III (HAART) were significantly higher than that of the other groups, even though the incidence of acute kidney failure was similar in the three groups.
It is difficult to make comparisons between different studies because of differences in ethnic composition, HIV transmission routes and the classification of anatomopathological lesions, in addition to the study period. As in other autopsy cohorts, the main anatomopathological diagnoses derived from tubulointerstitial lesions were ATN and septic nephritis.
The disease is also very heterogeneous with regard to glomerular changes. In the study of our pre-HAART patients (group I), similar to the Italian study published by Monga et al.,13 there was a predominance of the forms of mesangial proliferative glomerulonephritis, which may be related to the large amount of circulating antigens and the formation of immune complexes that occur in these patients. This is consistent with the study by Gutierrez et al.,31 which highlights the growing incidence of glomerular diseases associated with immune complexes, compared with HIV-associated nephropathy (HIVAN). However, in this study, among the glomerular diseases associated with immune complexes, we highlight the frequent membranoproliferative glomerulonephritis (MPGN) associated with HCV infection. This disorder is in contrast with the results of our study, since, despite the high prevalence of HBV and HCV coinfection found in our samples, there was only one case of membranoproliferative glomerulonephritis in group I and one case of membranous glomerulonephritis in group II (in a patient coinfected with both viruses). This low prevalence rate of glomerulonephritis associated with viral liver diseases was also described in the Spanish study by Martinez et al.12
In our study, the 4 patients with focal segmental glomerulosclerosis met the criteria for HIVAN.22 This constitutes a higher incidence rate than in cases described in previous studies, especially when all the patients were Caucasian (there were no cases described in the Spanish study by Martinez et al.12 and in the Dutch study by Van der Rejden;8 while the rate was 0.4% in the Swiss study by Hailemariam et al.16 and 0.8% in the Italian study by Monga et al.13). However, in studies carried out in the North American population, where there is a clear predominance of African Americans, there is a HIVAN incidence rate of up to 12% of the patients.6,7,9,10,23,30
Concerning treatment, some studies have shown a benefit from the use of HAART, such as the study by Atta et al.,32 where patients with HIVAN who were treated with HAART had a slower progression compared with those who did not receive HAART. In contrast, there are also studies in the literature that do not associate slower progression to kidney failure with the use of HAART, as is the case of the study by Szczech et al.33 ACE inhibitors and ARA II (Angiotensin II Receptor Antagonists) have been shown to decrease proteinuria and progression to kidney failure. In the study by Wei et al.,34 the patients with HIVAN who received fosinopril had a stable renal function, while those patients who were not treated with fosinopril had to undergo haemodialysis. The results of the study by Smith et al.,35 where corticosteroids were administered to 20 patients with HIVAN, show that 17 of them improved considerably, although relapse occurred after discontinuation of the therapy. In addition, 6 patients developed infectious complications. In this context, this means that currently this treatment is contraindicated or at least questioned. In our case, of the 4 patients with HIVAN only one presented the typical progression with proteinuria and renal function impairment. This patient received HAART (zidovudine, lamivudine and saquinavir), corticosteroids and ACE inhibitors, resulting in a slowing of the progression to the terminal stage of the disease.
Nephrocalcinosis, a common finding in patients with HIV that has been associated with treating patients with amphotericin B24 and with the infection caused by Pneumocystis jirovecii,25 was rare in our study, with rates ranging between 1.6% and 6.7 %.
The results of this study provide important and unique information about the spectrum of kidney disease in patients with HIV, since they have not been selected for the presence of kidney disease. In addition, this study constitutes one of the most extensive renal anatomopathological studies published so far, which includes patients treated with HAART and those untreated. The study’s limitations include a lack of information about abnormalities in the urinalysis in most patients, as well as the fact that renal function was assessed according to creatinine levels, following the same methodology of the previous study.17 It should be noted that it was not possible to carry out an immunofluorescence study, while the diagnosis was made only by means of direct view, using optical microscopy together with the previously described staining methods. This has prevented the best classification of glomerulonephritis, especially mesangial, found in our study. However, the advantage of autopsy studies is that more tissue is available than on guided biopsies, which allow for the diagnosis of early-stage kidney diseases. It should be remembered that, as previously noted in our study, there is a predominance of tubulointerstitial disease.
According to our results, there are some possible differences between patients treated with HAART and those untreated, especially with regard to a possible increased interstitial disease. Similarly, the number of cases of acute tubular necrosis could be higher in these patients, which may be related to renal damage attributed to other causes, especially the patients’ haemodynamic status and the nephrotoxic medicines administered prior to death.
Finally, it is important to note that there is a significant change in the causes of death in patients with HIV, with a decrease in opportunist and non-opportunist infections and an increase of deaths caused by liver diseases and neoplasias. In addition, the average age at the time of death is significantly higher in the current stage. These results are consistent with our previous study on the progression of the causes of death in patients with HIV and with other studies published in different countries.26-29
According to our data, we can say that people with HIV have a high susceptibility to the development of renal damage, especially tubulointerstitial, which is favoured both by general factors and virus-specific factors. In this study, we outline some possible differences between patients treated with HAART and those untreated, particularly in relation to a possible increased interstitial disease in those treated with HAART. This could be influenced by the advanced age of the patients in group III, and the fact that they have a longer survival and a slower time of progression of HIV. On the other hand, the number of cases of acute tubular necrosis could be higher in these patients, where – in addition to HAART, the age at the time of death and the time of progression of HIV – we should consider other causes, in particular the patients’ haemodynamic situation and the nephrotoxic medicines administered prior to death. The special characteristics of the patients who were included in this study, their critical status at the time of death, the volume depletion and the use of potentially nephrotoxic medicines could constitute factors that would favour tubular damage. This is why anatomopathological studies need to be carried out in patients treated with HAART who are in a stable condition, so as to assess the real impact of these treatments on the occurrence of renal lesions.
Table 1. Sample characteristics
Table 2. Description of renal lesions observed
Table 3. Diagnoses of renal lesions