Introducción: La hiperfosfatemia (fósforo sérico ≥ 5,5 mg/dl) es un factor independiente de mortalidad en la población en diálisis. Comparamos el transporte peritoneal de fósforo, creatinina y urea para demostrar diferencias y señalar la relevancia de estos parámetros en el control del fósforo sérico. Material y métodos: Se incluyeron 60 pacientes en diálisis peritoneal y se determinó el índice dializante/plasma de fósforo (D/P P) y creatinina (D/P Cr), el aclaramiento semanal de fósforo (AclP) y creatinina (AclCr), Kt/V de urea semanal y la excreción peritoneal de fósforo (ExP). Resultados: El D/P P fue superior en pacientes con normofosfatemia, comparados con los que presentaron hiperfosfatemia, 0,61 ± 0,13 frente a 0,54 ± 0,10 (p = 0,035). Se observó una adecuada correlación entre el D/P P y el D/P Cr, r = 0,90, p < 0,05, pero una pobre concordancia entre ambos, con un límite inferior de −0,17 (−0,24 a −0,09 IC 95 %) y límite superior de 0,47 (0,39-0,54 IC 95 %) para el D/P Cr respecto al D/P P. El AclP tuvo una adecuada correlación con el D/P P en pacientes con Kt/V ≥ 1,7 (r = 0,384, p = 0,04) y en anúricos (r = 0,392, p = 0,04), pero no con el D/P Cr. Hubo una pobre concordancia del AclCr respecto al AclP con límite inferior de −13,54 l/sem/1,73 m2 SC (−21,68 a −5,4 IC 95 %) y límite superior de 58,98 l/sem/1,73 m2 SC (50,84-67,12 IC 95 %). La ExP total se relacionó con el AclP (r = 0,643, p < 0,05), mientras que no lo hizo con el AclCr (r = 0,222, p = 0,23). Mediante el método CHAID se realizó un árbol de clasificación del transporte de fósforo con base en su D/P, obteniendo 5 nodos (≤ 0,5, 0,51-0,55, 0,56-0,66, 0,67-0,76, > 0,76), mostrando diferencias estadísticamente significativas entre nodos para niveles séricos de fósforo, AclP total y peritoneal, así como Kt/V de urea semanal. Conclusiones: Las mediciones de D/P P y AclP no concuerdan con las mediciones de D/P Cr y AclCr, respectivamente, por lo que su determinación es una herramienta clínica para el control del nivel de fósforo sérico.
Background: Hyperphosphataemia (serum phosphorus ≥5.5mg/dl) is an independent mortality factor for the dialysis population. We compared phosphorus, creatinine and urea peritoneal transport to demonstrate differences and indicate the relevance of these parameters in the control of serum phosphorus. Material and method: We included 60 patients on peritoneal dialysis and determined the dialysate/plasma phosphorus (D/P P) and creatinine (D/P Cr) ratios, weekly creatinine clearance (CrCl) and phosphorus clearance (PCl), weekly Kt/V of urea, and peritoneal phosphorus excretion (PEx). Results: D/P P was higher in patients with normal phosphataemia, compared with those who were hyperphosphataemic 0.61±0.13 versus 0.54±0.10 (p=.035). We observed an adequate correlation between D/P P and D/P Cr, r=0.90, p<.05, but poor concordance between both, with a lower limit of −0.17 (−0.24 to −0.09 95% CI) and an upper limit of 0.47 (0.39-0.54 95% CI) for D/P Cr with respect to D/P P. PCl had an adequate correlation with D/P P in patients with a Kt/V ≥1.7 (r=0.384, p=.04) and in anuric patients (r=0.392, p=.04), but not with D/P Cr. There was poor concordance of the CrCl with respect to PCl with a lower limit of −13.54l/week/1.73m2 BSA (−21.68 to −5.4 95% CI) and an upper limit of 58.98l/week/1.73m2 BSA (50.84-67.12 95% CI). Total PEx was related to PCl (r=0.643, p<.05), while it was not related to CrCl (r=0.222, p=.23). Using the CHAID method, we created a classification tree for phosphorus transport based on its D/P, obtaining 5 nodes (≤0.5, 0.51-0.55, 0.56-0.66, 0.67-0.76, >0.76), with statistically significant differences between nodes for serum phosphorus, total and peritoneal PCl and weekly Kt/V of urea. Conclusions: D/P P and PCl are not concordant with D/P Cr and CrCl respectively and therefore their determination is a clinical tool to control serum phosphorus levels.
INTRODUCTION
Cardiovascular disease is the main cause of mortality in population with advanced renal disease, and hyperphosphatemia (serum phosphate > 5.5 mg/dl) is an independent risk factor that can range from 30 to 40% of the population subjected to renal replacement therapy, resulting amongst other complications, calcification of the tunica media of blood vessels, particularly in coronary arteries, which precipitates acute cardiovascular events, with fatal consequences1-10. For these reasons, controlling serum phosphorus is imperative in the management of chronic kidney disease patients.
There is little information with respect to weekly phosphorus clearance (PCl) and dialysate/plasma phosphorus (D/P P). Sedlacek et al. found a high correlation between dialysate/plasma creatinine (D/P Cr) and D/P P, as well as their respective clearances, concluding that the clearance of both solutes is closely linked, and as such, the main determinant for weekly creatinine clearance (CrCl) and PCl is D/P Cr11. In a study of paediatric patients on automated peritoneal dialysis, they found a good correlation between both D/P, but PCl was related better to D/P P than to D/P Cr, with the conclusion that the former is a better predictor12. Bernardo et al. classified phosphorus transport by its D/P based on Twardowski’s creatinine transport classification13 and they observed that the distribution of patients based on D/P P in the different categories was different to that presented for D/P Cr, suggesting that, despite there being a correlation between D/P P and D/P Cr, the latter measure is not adequate enough to classify patients in accordance with the transport of phosphorus14. This study evaluates concordance between D/P Cr and D/P P, and between CrCl and PCl, using the Bland and Altman method15, with the objective of finding out how much one test may differ from the other and thus determine whether D/P Cr and CrCl can continue to be reliable replacements of peritoneal phosphorus transport. Furthermore, we evaluated peritoneal phosphorus transport in a dialysis population with the aim of highlighting its role in phosphataemia control beyond dietary restriction and the use of phosphate binders.
MATERIALs AND METHODs
We carried out a cross-sectional, observational and comparative study in which we included 60 patients of the peritoneal dialysis outpatient clinic. Inclusion criteria: patients older than 16 years of age who were on renal replacement therapy for at least one month through continuous ambulatory peritoneal dialysis or nocturnal automated peritoneal dialysis (NAPD). Exclusion criteria: having presented with peritonitis at the time of the study, with this being considered to be the presence of 100 or more leukocytes per field in the cytological study of dialysate, who completed antibiotic treatment within the two previous months or patients who presented or had presented in the previous month with an incapacitating medical condition. For the peritoneal equilibration test (PET), we included as exclusion criteria ultrafiltration failure, defined as lower than 400ml after the application of a 4.25% 2000ml bag after 4 hours in the peritoneal cavity, catheter mechanical problems, defined as a fluid entry time greater than 10 minutes and an exit time greater than 30 minutes or having greater D/P P than the unit. The following were counted as exclusion criteria in solute clearance measurement: NAPD mode (reasons unrelated to the interests of this study), peritonitis, the 24-hour dialysate effluent volume being lower than the infused volume and a lack of adherence to the dialysis prescription, confirmed subjectively by the patient.
We performed a PET on all patients using a 4.25% solution to determine D/P P and D/P Cr. We carried out a cytology of the dialysate in order to rule out an active infectious condition. We calculated weekly the Kt/V of urea and weekly PCl and CrCl expressed in litres per week adjusted to body surface area (l/week/1.73m2 BSA), all in their renal, peritoneal and total forms using standard methods16, as well as the normalized protein nitrogen appearance using the Randerson formula17. The urine samples were only collected for those with diuresis equal to or greater than 200ml/day; if this was not the case, they were considered anuric. The Department of Nutrition, though the food frequency questionnaire of the Nutritional Habits and Nutrient Consumption Evaluation System18 determined the average daily phosphorus intake.
STATISTICAL ANALYSIS
The results were expressed as means and standard deviations in the case of numeric variables or as proportions for categorical variables. The comparison of means between the two groups was carried out with Student’s t-test for independent groups or with its non-parametric alternative (U-Mann-Whitney), in accordance with the distribution of each variable. The comparison of proportions was carried out using the χ2 test. Variables were associated using Spearman’s correlation coefficient.
We carried out a concordance analysis between two tests (D/P Cr and D/P P; CrCl and PCl) using the Bland and Altman method. We created a graph showing the difference between the two methods against their average. 95% of differences are found in this graph between two limits that define the confidence interval (CI). The concordance limits are displayed with their respective 95% CI.
To compare more than two means, we carried out a one-way ANOVA test with the Bonferroni test. In order to divide up the database in the best way possible with regard to the D/P P variable, we used a classification tree, taking the CHAID (Chi Squared Automatic Interaction Detector) method as the form of growth.
Values of P<.05 were considered to be statistically significant. The statistical package SPSS version 15 for Windows was used.
RESULTS
We included 60 patients, of which 4 PET were excluded due to having a greater D/P P than the unit and we eliminated 10 solute clearance measurements due to the following: one due to inadequate sample collection, one due to not requiring dialysis after recovery from acute renal damage, four due to a 24-hour ultrafiltration volume lower than the infused volume, two due to being in NAPD mode and two due to not having a stable dialysis dose because of poor adherence to their prescription.
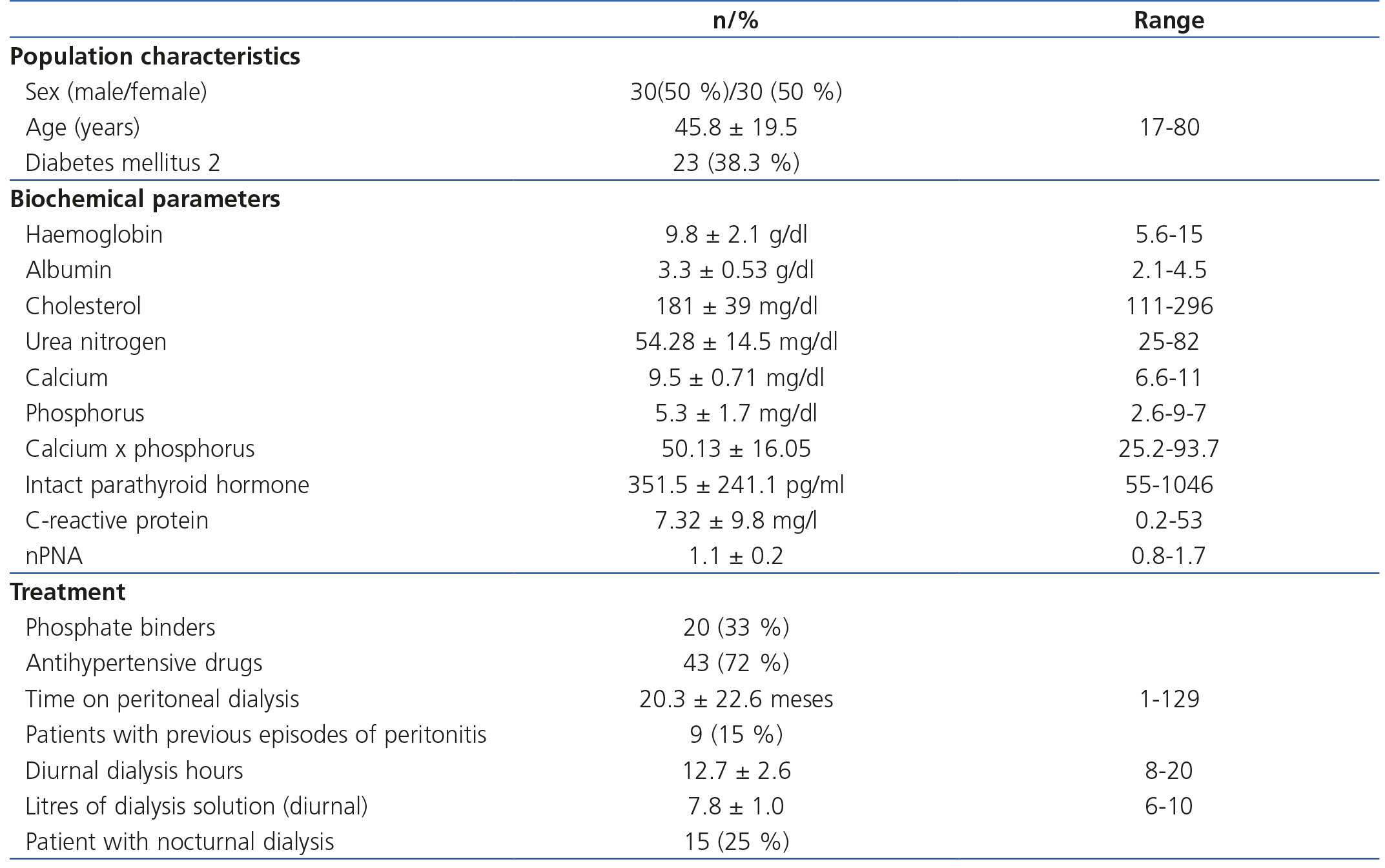
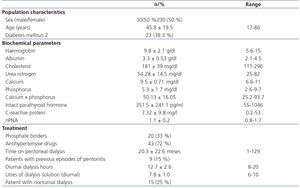
The population characteristics and the biochemical and treatment parameters can be observed in Table 1.
Phosphorus
Mean serum phosphorus in our population was 5.3±1.7mg/dl (2.6-9.7mg/dl), with 21 (31.3%) cases of hyperphosphataemia, with there being a low inverse correlation between phosphorus levels and D/P P, r=−0.273, p=.04 and a statistically significant difference in D/P P in those with normal phosphataemia when we compared them with hyperphosphataemic patients, 0.61±0.13mg/dl versus 0.54±0.10mg/dl (p=.035). Daily estimated mean phosphorus intake was 1290±472mg/day (522-2794mg/day). We found that those who consumed a quantity lower than or equal to 1000mg/day (n=18, 32%) had significantly lower serum phosphorus when we compared them with those who consumed more than 1000mg/day (4.8±1.6mg/dl versus 5.6±1.7mg/dl, p=.039).
Dialysate/plasma phosphorus and creatinine ratios
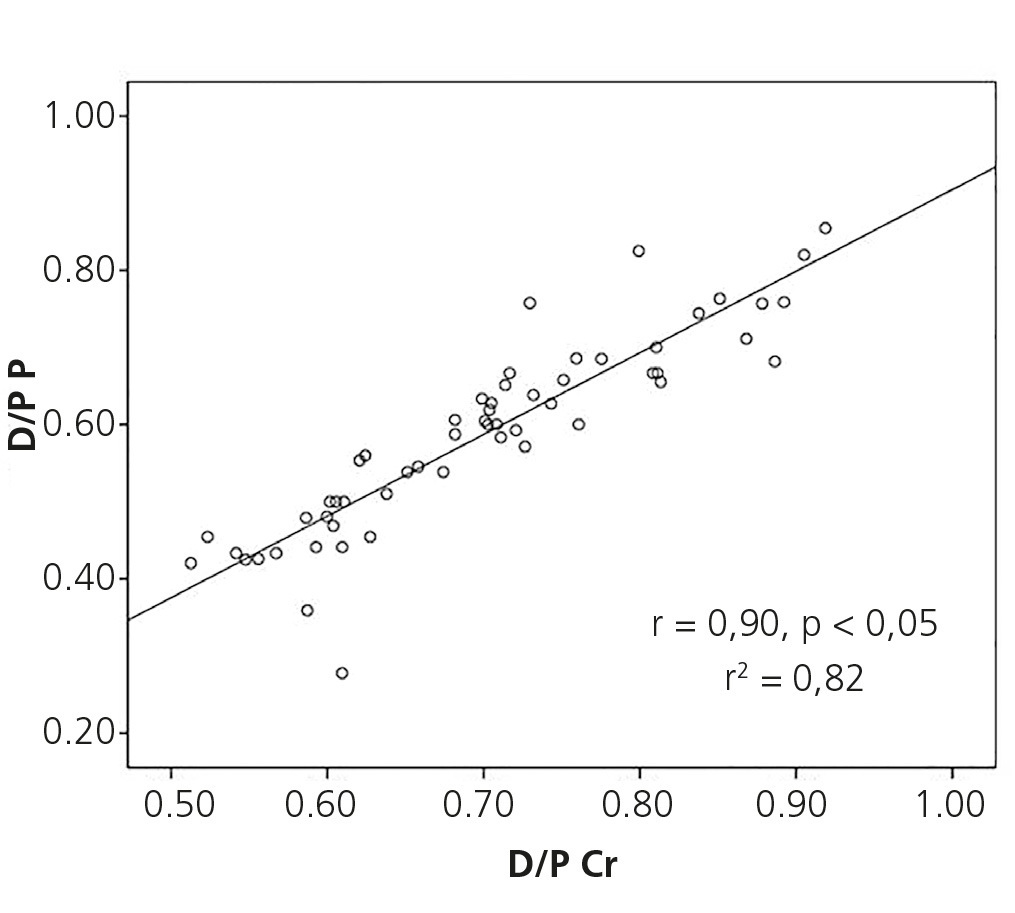
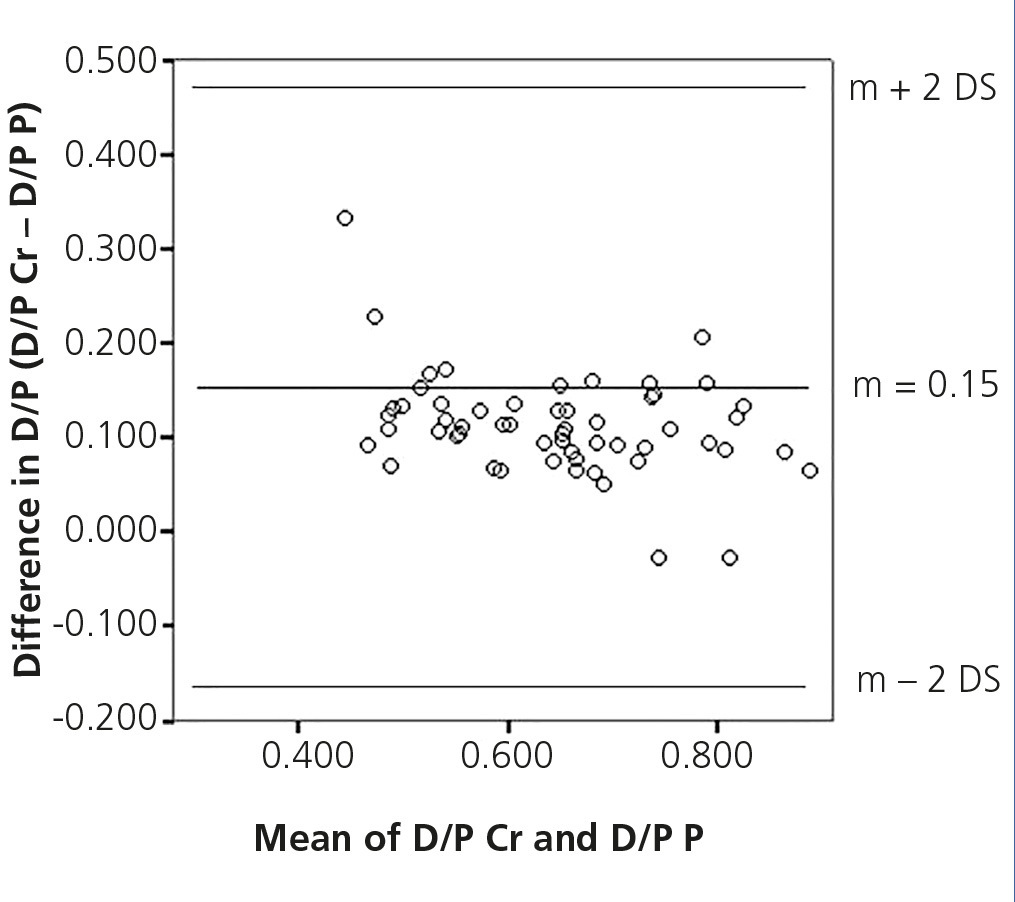
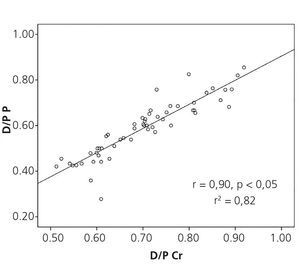
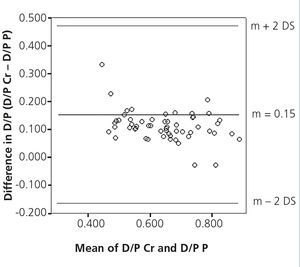
The mean D/P Cr for our population was 0.70±0.10 (0.51-0.92) and the mean D/P P was 0.58±0.12 (0.28-0.85), and we observed an adequate correlation between the two, with r=0.90, p<.05 (Figure 1). However, we observed a poor concordance between the two methods with upper and lower limits of D/P Cr with respect to D/P P of −0.17 (−0.21 to −0.13 95% CI) and 0.47 (0.35-0.59 95% CI), respectively (Figure 2).
Phosphorus clearance, creatinine clearance and Kt/V of urea
We observed a low correlation between PCl and D/P P (r=0.33, p=.02), and between PCl and D/P Cr (r=0.30, p=.03). However, when we carried out a subanalysis that only included those with a Kt/V of urea ≥1.7, we found a correlation between PCl and D/P P (r=0.384, p=.04), but not between PCl and D/P Cr (r=0.348, p=.06). We also explored this association in anuric patients, where we similarly observed that PCl was significantly related to D/P P (r=0.392, p=.04), but not to D/P Cr (r=0.358, p=.52).
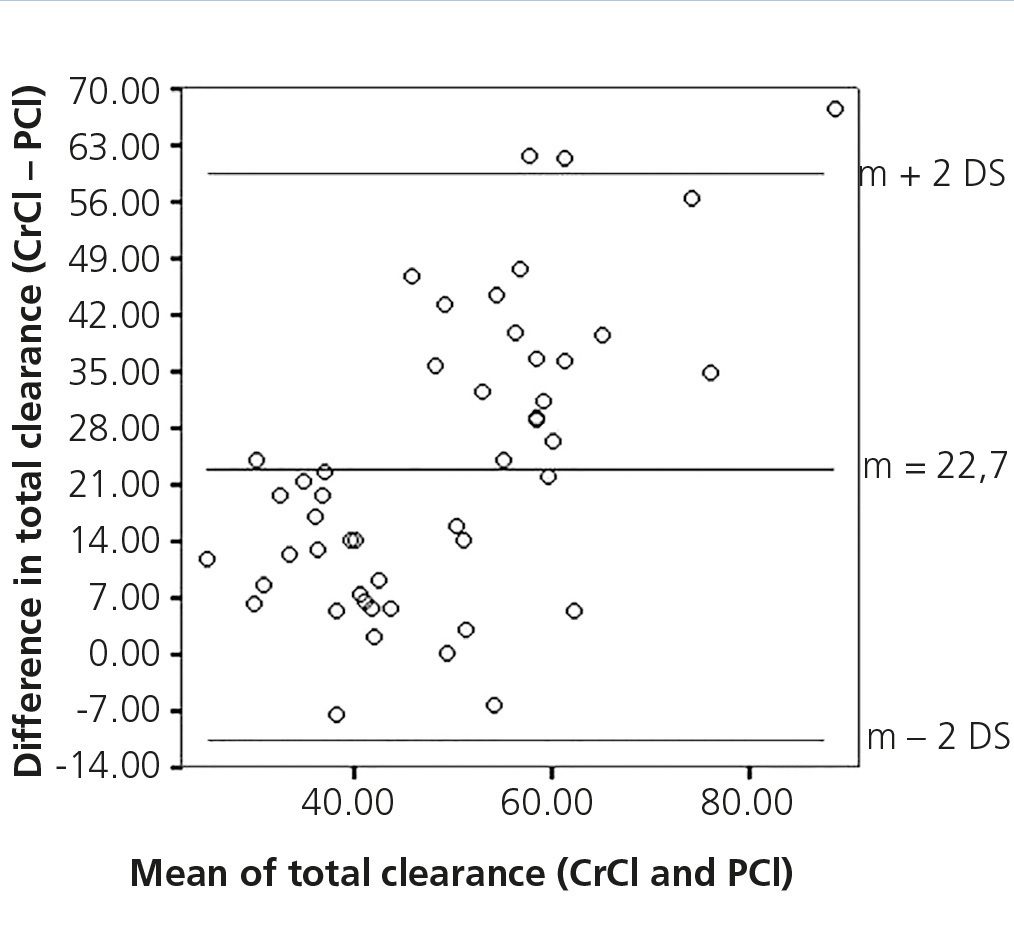
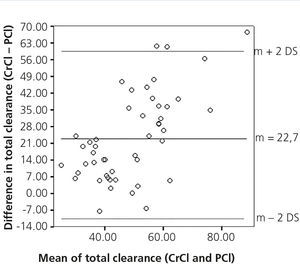
We attempted to find an association between the Kt/V of urea, CrCl and PCl in their total, peritoneal and renal measurements and we found the following: Kt/V of urea had significant correlations with CrCl in its total, peritoneal and renal measurements (r=0.68, p<.05; r=0.78, p<.05; r=0.47, p=.03, respectively); PCl showed significant correlations with CrCl in its total, peritoneal and renal measurements (r=0.45, p<.05, r=0.59, p<.05 and r=0.67, p<.05, respectively). However, the concordance analysis showed that it is poor, with lower and upper concordance limits of −13.54l/week/1.73m2 BSA (95% CI −21.68 to −5.4) and of 58.98l/week/1.73m2 BSA (95% CI 50.84-67.12), respectively, of total CrCl with respect to total PCl (Figure 3). Moreover, renal PCl showed an almost significant association with Kt/V of renal urea (r=0.43, p=.05), while total and peritoneal PCl did not display a significant association with Kt/V of total and peritoneal urea (r=0.18, p=.31 for both).
Phosphorus clearance, serum phosphorus levels and residual renal function
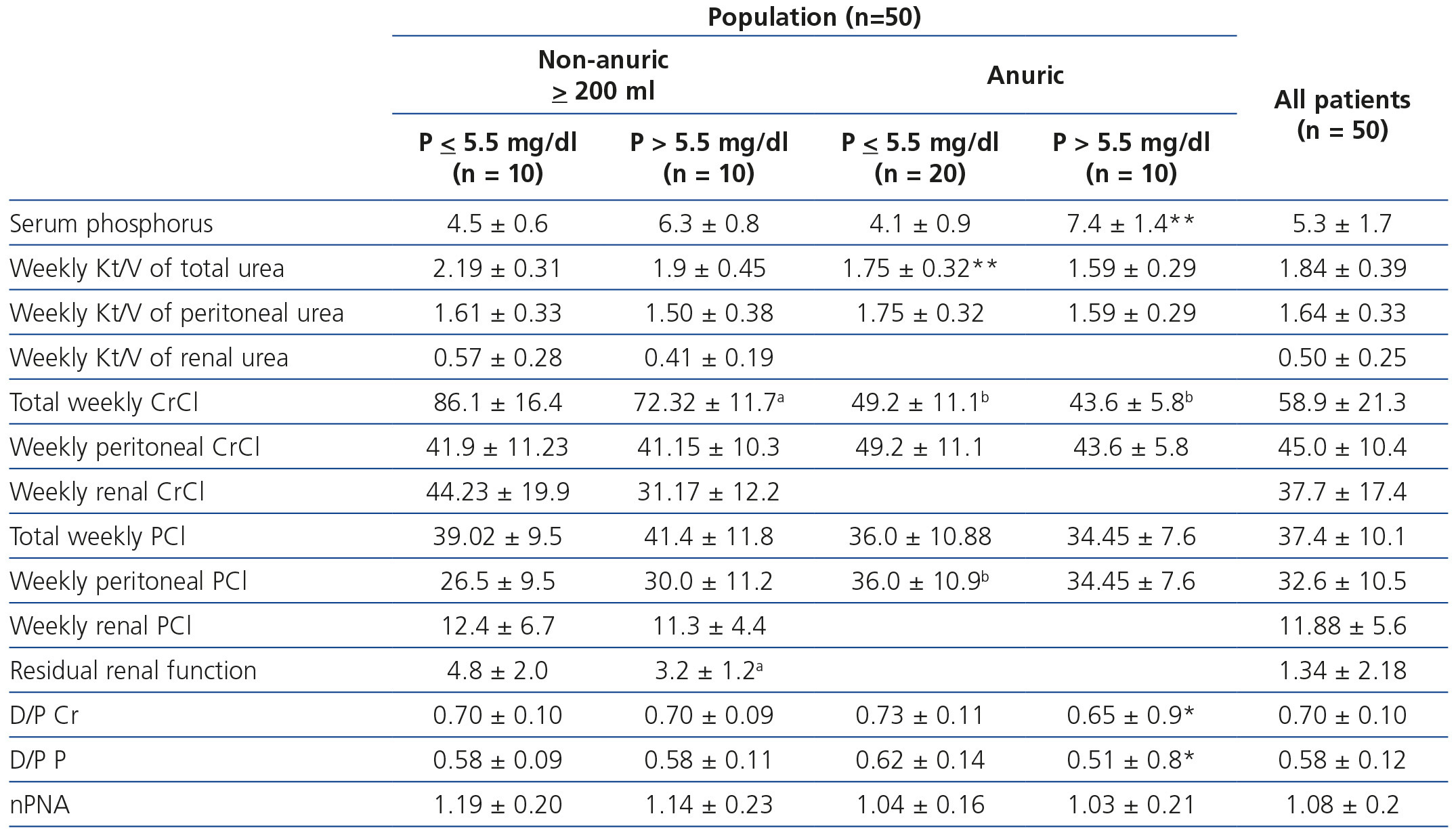
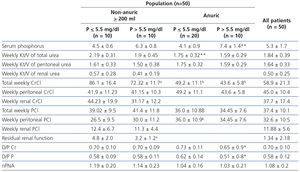
When we divided patients in accordance with diuresis and subdivided each group into normal phosphataemia and hyperphosphataemia (Table 2), we could observe that mean serum phosphorus for anuric patients with hyperphosphataemia was significantly higher than those with hyperphosphataemia who maintained diuresis (7.4±1.4mg/dl versus 6.3±0.8mg/dl, p=.03). D/P P was significantly lower in anuric patients with hyperphosphataemia, compared with those who had normal serum phosphorus levels (0.51±0.8 versus 0.62±0.14, p=.006). Although total PCl was higher in patients with diuresis compared with the anuric patient subgroup (40.2±10.4l/week/1.73m2 BSA vs. 35.6±9.6l/week/1.73m2 BSA, p=.12), it was not significantly different. However, peritoneal PCl in anuric patients was significantly higher when we compared it with patients who maintained diuresis (35.5±9.8l/week/1.73m2 BSA vs. 28.3±10.25l/week/1.73m2 BSA, p=.01). Residual renal function was significantly lower in patients with hyperphosphataemia compared with those with normal phosphataemia (3.2±1.2ml/min vs. 4.8±2.0ml/min, p=.048).
Phosphorus excretion
We also evaluated the potential association between total and peritoneal phosphorus excretion with total and peritoneal CrCl and PCl, and we found the following: total phosphorus excretion showed a better correlation with total PCl (r=0.62, p<.05) than with total CrCl (r=0.360, p<.05). Peritoneal phosphorus excretion also had a better correlation with peritoneal PCl (r=0.687, p<.05) in comparison with peritoneal CrCl (r=0.466, p<.05). Lastly, in patients with a Kt/V of urea ≥1.7 the correlation between total PCl and total phosphorus excretion was significant (r=0.643, p<.05), but was not between total CrCl and total phosphorus excretion (r=0.222, p=.23).
Categorization of phosphorus transport
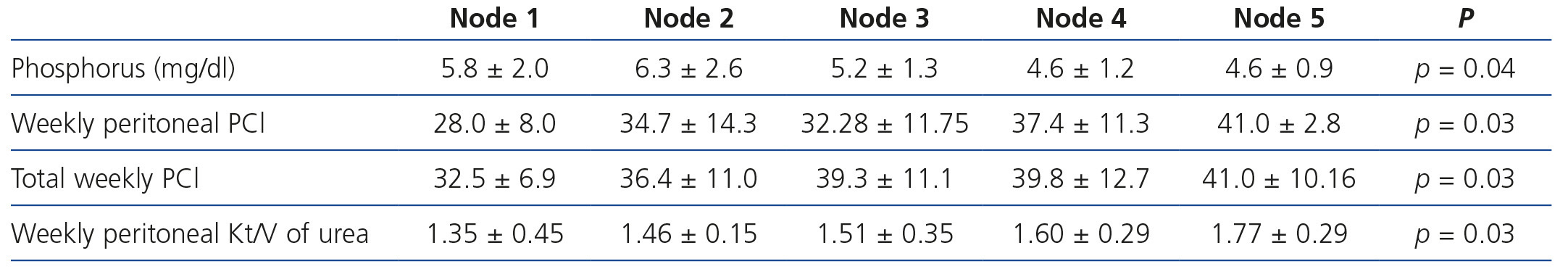
On the basis of D/P P distribution using the CHAID method, we created a classification tree and obtained the following nodes with their respective ranges: node 1: ≤0.5, node 2: 0.51-0.55, node 3: 0.56-0.66, node 4: 0.67-0.76, node 5: >0.76, and we found statistically significant differences between nodes in terms of serum phosphorus levels, total and peritoneal PCl, and Kt/V of urea (Table 3).
DISCUSSION
Phosphorus clearance is the result of the complex interaction between diffusion, convection, osmotic and chemical factors, electrical gradients, and active transmembrane phosphorus transporters19,20. It has been demonstrated that absorption by lymphatic convection has no impact and that solutions with a higher concentration remove a higher quantity of phosphorus and are attributed to the convection factor. However, it was not possible to establish for certain whether ultrafiltration influences clearance21. Messa et al. demonstrated in 35 patients that glucose concentration was one of the predictors of phosphorus clearance and this was independent of the ultrafiltration volume22, while Granja et al. calculated that 11% of the phosphorus eliminated was due to ultrafiltration23.
In our population, we found that the frequency of hyperphosphataemia was 31.3%, which is similar to that reported in the literature7. In previous studies, an adequate correlation was found between D/P Cr and D/P P, and between CrCl and PCl, suggesting equivalence between them12,13. In this study, we demonstrated that although, D/P Cr and D/P P displayed adequate correlation, the concordance was poor, since the concordance limits were very broad. With such great variability of D/P Cr or CrCl with respect to the phosphorus parameters, there would be a very significant impact on clearance, excretion and therefore serum phosphorus levels, and as such, these measurements could not be considered equivalents. For example, a patient with a D/P P of 0.4 may have a D/P Cr from 0.16 to 0.94, values that are very different from the value of D/P P. Likewise, a patient with PCl of 35l/week/1.73m2 BSA could have CrCl from 13.32l/week/1.73m2 BSA to 102.12l/week/1.73m2 BSA. Given the poor concordance that exists between the values studied and that they cannot be taken as replacements, we proposed a phosphorus transport classification tree with five nodes based on its D/P P, amongst which we identified statistically significant differences between groups for total and peritoneal PCl.
Our results confirm the role of diuresis in maintaining phosphorus levels at low values compared to anuria. In anuric patients, in whom the peritoneum has an exclusive role in phosphorus clearance, the determination of phosphorus transport indicators is particularly important for two factors: 1) D/P P is significantly lower in hyperphosphataemic patients; 2) Peritoneal PCl is significantly higher compared with those who maintain diuresis (Table 2).
The current recommendations for the prescription of peritoneal dialysis in accordance with the 2006 K/DOQI clinical guidelines24, based on the results of three major studies25-27, set a Kt/V of urea target of ≥1.7, which has not managed to reduce mortality in this population. As such, the search for new markers of targets is vitally important. The current recommendations are to maintain the phosphorus level within the normal limits28, although this is an arduous challenge for patients and physicians, since beyond dietary restriction and the adequate use of binders, the doctor must know the role that dialysis exercises on PCl. This study demonstrates the independence of phosphorus transport with respect to urea and creatinine and it leads to a series of questions such as the PCl dose necessary for helping patients achieve the serum phosphorus targets, whether this value is different in patients with diuresis and anuric patients, whether the dialysis adjustment to achieve an established PCl target improves mortality compared with the current Kt/V of urea targets or whether a low D/P P is associated with greater mortality, amongst others. As such, studies must be carried out to determine the benefits of the adjustment of peritoneal dialysis based on D/P P with different PCl doses, establish the mid- and long-term morbidity and mortality, and thus compare them with the current recommendations on dialysis.
CONCLUSIONS
This study shows that D/P P and PCl measurements are not concordant with those of D/P Cr and CrCl, respectively, despite having an adequate correlation. As such, given the current lack of knowledge about phosphorus transport in peritoneal dialysis in clinical practice, in a group of patients in whom phosphataemia control is imperative, it is necessary to give continuity to the research of these indicators, since they are useful tools for physicians and may guide dialysis prescription in search for a better control of serum phosphorus.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Table 1. Population characteristics: medical and dialysis management
Table 2. Transport of solutes, dialysis dose and phosphataemia in accordance with residual uresis
Table 3. Phosphorus and clearance characteristics between transport nodes
Figure 1. Correlation between the dialysate/plasma creatinine ratio and the dialysate/plasma phosphorus ratio.
Figure 2. Concordance between the dialysate/plasma creatinine ratio and the dialysate/plasma phosphorus ratio.
Figure 3. Concordance between creatinine clearance and phosphorus clearance.