Antecedentes: En la poliquistosis renal autosómica dominante (PQRAD), la hematuria macroscópica por la rotura de quistes renales es una manifestación habitual, que puede ser espontánea o el resultado de un traumatismo, cálculos renales, un tumor o una infección. El sangrado del quiste puede verse prolongado por la activación local de la fibrinólisis por uroquinasa. El manejo de la hematuria, en la mayoría de los pacientes con PQRAD, suele ser conservador con reposo, transfusiones, corrección de las coagulopatías, uso de desmopresina y agentes estimulantes de la eritropoyesis. En algunos casos de hematuria incoercible, puede ser necesaria la embolización y hasta la nefrectomía. Otras modalidades terapéuticas han tratado de evitar la hospitalización prolongada y la nefrectomía, intentando preservar la función renal con la administración de agentes antifibrinolíticos. Objetivo: El objetivo del estudio fue analizar, de forma prospectiva, la respuesta a la administración de ácido tranexámico en pacientes con PQRAD y con hematuria masiva incoercible o que no cede con tratamiento convencional. Resultados: Se evaluó su acción sobre la función renal, así como el desarrollo de efectos secundarios. Un total de ocho pacientes recibieron ácido tranexámico por vía oral o i.v., durante de 2 a 5 días, y en todos los casos la hematuria cedió de forma inmediata o en el plazo de pocos días, manteniendo estables los niveles de hemoglobina y de función renal. No se observaron complicaciones tromboembólicas o de otro tipo. En esta serie de casos, que hasta ahora es el mayor estudio prospectivo que se ha comunicado y el único que incluye diferentes grados de función renal, el ácido tranexámico se confirma como una herramienta prometedora para tratar la hematuria debida a hemorragia quística en la PQRAD. Conclusiones: El ácido tranexámico puede utilizarse de forma segura para el tratamiento de la hematuria sin respuesta a la terapia convencional en la PQRAD. El tratamiento resultó eficaz, tanto en los pacientes con función renal conservada como en aquéllos con insuficiencia renal crónica. El fármaco puede administrarse por vía oral o i.v., siendo necesario ajustar las dosis según el grado de función renal. La terapia con ácido tranexámico puede preservar la función renal directamente, al parar y espaciar los episodios acumulativos de hematuria, o indirectamente, evitando la embolización y la nefrectomía. Es necesario realizar estudios prospectivos controlados y aleatorizados para obtener conclusiones más definitivas.
Background: Gross haematuria is a common manifestation of autosomal dominant polycystic kidney disease (ADPKD). It can be spontaneous or the result of trauma, renal calculi, tumour, or infection. Spontaneous cyst bleeding is important in this particular group of patients, since it can be prolonged by local activation of fibrinolysis by urokinase. The management of haematuria in ADPKD is usually conservative, including bed rest, blood transfusion, correction of blood disorders, and use of vasopressin and erythropoiesis-stimulating agents. In some patients, the management of gross or life-threatening haematuria may require embolisation and/or nephrectomy. Nonetheless, other methods have been tried to avoid prolonged hospitalisation and nephrectomy and preserve kidney function, such as the use of anti-fibrinolytics. Tranexamic acid was recently suggested as a tool to treat gross haematuria in ADPKD in isolated cases. Objective: The aim of this study was to evaluate prospectively the response to tranexamic acid in a group of 8 patients with ADPKD and gross haematuria unresponsive to conventional treatment. Results: The massive bleeding stopped within 2 to 5 days in all patients. The haemoglobin level and renal function subsequently stabilised. There were no side effects or thromboembolic events. In this case series, the largest prospective study so far published and the only one including different degrees of renal function, tranexamic acid is confirmed as a promising tool for treating haematuria due to intracystic bleeding in ADPKD. Conclusions: In summary, tranexamic acid can be used safely in ADPKD patients with chronic renal impairment or preserved renal function to treat severe haematuria poorly responsive to conventional therapy. Tranexamic acid can be administered orally or IV; and dose adjustment for renal impairment is important. Tranexamic acid therapy may preserve renal function in ADPKD directly, by stopping haematuria episodes, or indirectly, by preventing embolisation and/or nephrectomy. The major limitation of this study is the small sample size and the lack of an untreated control group. We suggest a prospective, randomised controlled study to confirm the efficacy of this treatment, its long-term safety, and the optimal dosage. Further larger and multicentre studies are needed to evaluate the cost-benefit ratio and the limits of this therapy in the clinical setting.
INTRODUCTION
Haematuria is a common problem in autosomal dominant polycystic kidney disease, more than 60% of patients experiencing at least one episode of gross haematuria.1-4 These episodes are normally managed with conservative medical treatment and rarely require surgery. Although they are self-limiting, successive episodes of gross haematuria may have a detrimental effect on renal function in the long term.3,4 On the other hand, imaging techniques such as magnetic resonance (MRI) and computed tomography (CT) have become widespread, and we may now observe intracystic haemorrhages that would never have been noticed before. This is an important step forward, since it is widely known that patients with ADPKD who suffer from frequent haematuria episodes or show signs of intracystic haemorrhage develop chronic renal failure (CRF) more quickly.5,6 Conservative treatment for haematuria in ADPKD involves bed rest, blood transfusion, IV fluid, correcting blood disorders and use of vasopressin and erythropoiesis-stimulating factors. At present, there is no effective medical therapy for patients with intractable haematuria7 other than invasive procedures such as embolisation or nephrectomy. Nevertheless, other treatment methods have been tested with the aim of preserving renal function and avoiding long hospital stays, embolisation, and nephrectomy. These methods include the use of antifibrinolytics such as aprotinin and epsilon-aminocaproic acid,8 and more recently, tranexamic acid.9,10
Tranexamic acid is a synthetic lysine analogue with strong anti-fibrinolytic activity. It is commonly used to treat a number of bleeding disorders, including CRF.11-16 Several studies have shown it to reduce blood loss in patients with primary menorrhagia and those who have undergone cardiopulmonary bypass surgery, prostatectomy, hip replacement, and liver transplant procedures.17 However, we have not yet determined the effect of low doses of tranexamic acid on managing cyst bleeding and on renal function in ADPKD patients with a relatively preserved renal function or at a certain stage of CRF. In addition, since systemic and local hyperfibrinolysis may play a role in cyst bleeding in ADPKD,13 tranexamic acid was administered to a cohort of patients with different levels of renal function and intractable haematuria that did not respond to conventional treatment.
PATIENTS AND RESULTS
An observational study was conducted between 2009 and 2011 that included 8 patients with ADPKD (5 men and 3 women aged 23-51 years) who were referred to us due to intractable haematuria and suspected cyst bleeding, and who received treatment with tranexamic acid (Amchafibrin®, Rottapharm, Spain). In all cases, ultrasound (US), CT and/or MR imaging were used for diagnosis and follow-up. Medical histories and imaging studies were reviewed in order to determine the cause of the cyst bleeding. All patients showed a very high total renal volume measured by manual segmentation, and MRI and CT images showed several cysts with signs of intracystic haemorrhage. Case 4 was administered initial treatment with 4 doses of recombinant Factor VII (Novoseven®, Novo Nordisk, Spain), which was ineffective. Case 5 was also diagnosed with sickle-cell anaemia and had a history of recurrent haematuria. Three of the 8 patients were treated with IV tranexamic acid, hydration therapy, analgesics and blood transfusions. They were discharged 1 or 2 days after haematuria stopped and haemoglobin levels stabilised. The other 5 patients were treated with oral tranexamic acid on an outpatient basis. In all cases, haematuria resolved immediately or within a few days; haemoglobin levels and renal function remained stable. No thromboembolic or other complications were observed. There were no new haematuria episodes in the 3 months following discharge.
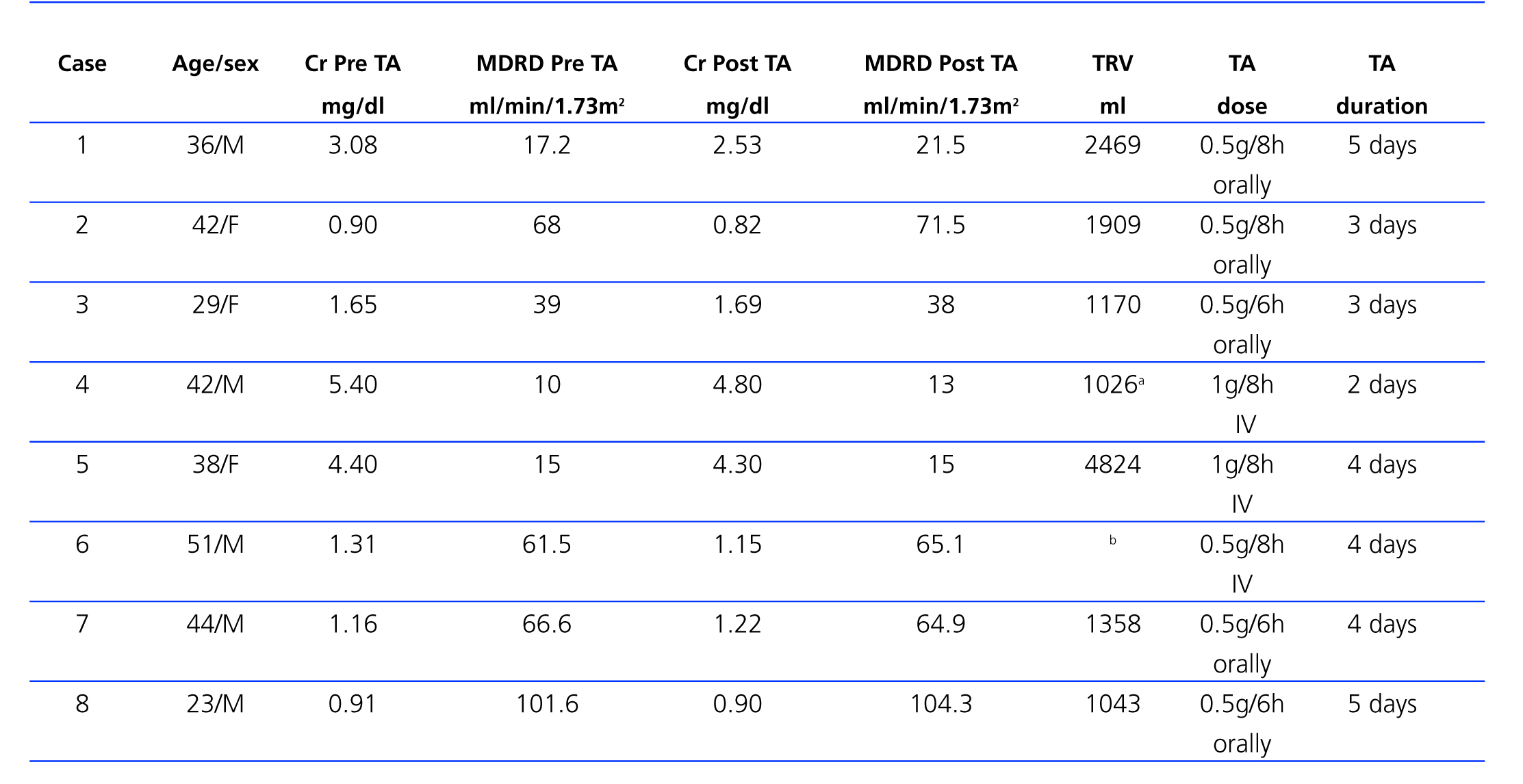
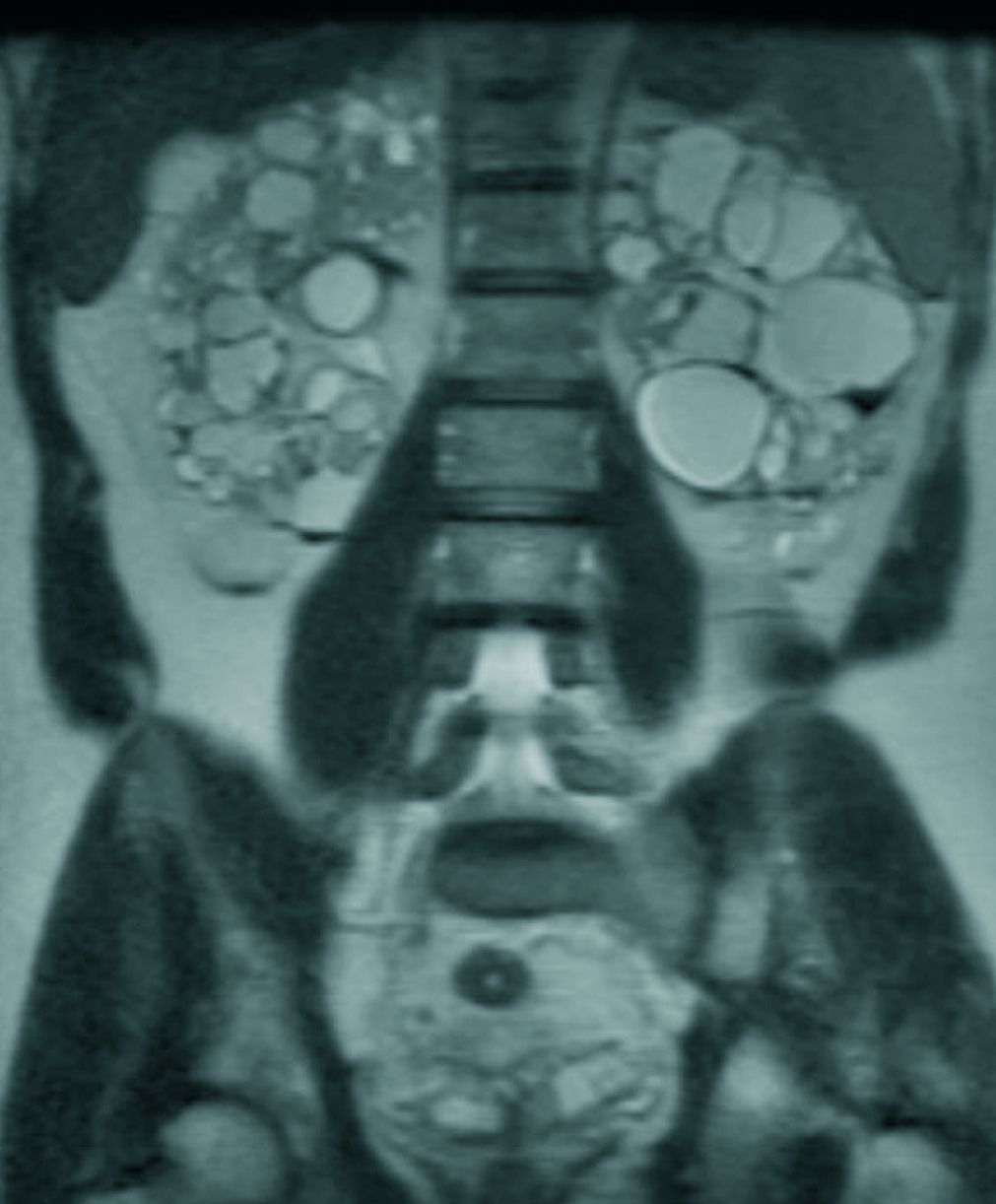
Table 1 provides a summary of the clinical and evolution data of the 8 patients treated with tranexamic acid. Figure 1 shows an example of renal cysts with signs of intracystic haemorrhage.
DISCUSSION
Gross haematuria caused by the rupture of renal cysts is a common manifestation of ADPKD. Haematuria is the first sign of the disease in 35% of all cases, and either gross or microscopic haematuria is detected in 60% of this patient population.1-6 Risk of haematuria seems to be associated with the presence of hypertension and cyst size increase. Although most patients report trauma or strenuous exercise as possible triggers, a precise association has never been proven between such triggering mechanisms and the condition. While polycystic kidneys are quite resistant to traumatic damage, mild trauma18-20 may give rise to intrarenal or retroperitoneal bleeding, accompanied by intense pain which may require administration of narcotics.21,22 Cysts are associated with excessive angiogenesis evinced by fragile vessels stretched across their distended walls.23,24 These vessels present an array of malformations, including aneurysms and spiral shapes. These vessels may leak blood into the cyst, either spontaneously or due to trauma; and as a result, the cyst will expand rapidly, causing intense pain. If bleeding continues, the cyst may rupture into the collecting system, which causes gross haematuria. Another possibility is that the cyst will break into the subcapsular compartment and eventually dissect through the renal capsule to fill the retroperitoneal space. In the case of massive bleeding, blood may reach the skin covering the flank and abdomen, manifested as subcutaneous ecchymosis (Grey Turner's sign). Intracystic haemorrhages in the kidney may be diagnosed by using US, CT or MRI. However, diagnosis is difficult in the presence of a persistent, but very slight haemorrhage. On the other hand, positron emission tomography-CT allows us to distinguish haemorrhages from cystic infection.25-27 Evidence from CT scans suggests that intracystic haemorrhage, shown as hyperdense subcapsular cysts, is present in 90% of patients with ADPKD.28,29 There are often dozens of superficial cysts that may cause intracystic haemorrhage. Direct inspection of the “hyperdense” cysts has revealed them to be filled with cellular debris derived from the breakdown of blood products.
Patients with a history of renal haemorrhage, as demonstrated by repeated episodes of gross haematuria, have larger kidneys1-6 and develop renal failure sooner than patients with no history of renal haemorrhage. In a retrospective study, Gabow et al1,2 found that male athletes with ADPKD who participated in contact sports had more haematuria episodes and developed kidney failure faster than those who did not participate in such sports. Renal haemorrhage caused by ruptured cysts may occur in patients of all ages, and this decreases their quality of life. There is currently no medical treatment for ADPKD patients with severe intractable haemorrhage, apart from conservative treatment and the use of invasive procedures such as embolisation and/or nephrectomy.
Tranexamic acid (trans-4-(aminomethyl)cyclohexanecarboxylic acid) is commonly used for the treatment of a number of bleeding disorders.11-16 It is a synthetic lysine analogue with strong anti-fibrinolytic activity. The plasminogen binds to fibrin to form plasmin, which in turn breaks down fibrin molecules into fibrin degradation products. Tranexamic acid blocks plasminogen's lysine binding sites and prevents its interaction with fibrin. As a fibrinolysis inhibitor, it is 7 to 10 times more potent than epsilon aminocaproic acid. Some clinical trials have proven it to reduce blood loss in patients with primary menorrhagia and those who have undergone cardiopulmonary bypass surgery, prostatectomy, hip replacement and liver transplant procedures.17 It was recently reported to be effective in treating haematuria in patients with sickle-cell anaemia as well.30 Tranexamic acid is well-tolerated in general. Although a few cases of renal cortical necrosis have been reported,31,32 its side effects are mainly gastrointestinal and include nausea, vomiting, diarrhoea and abdominal pain. Myoclonus and encephalopathy due to overdose have been described in only one case.33 Tranexamic acid is excreted through glomerular filtration, and therefore accumulates in the presence of low renal function. Patients with CRF will require dose titration. On the other hand, the effectiveness of haemodialysis in tranexamic acid elimination has not been studied.15,16 The normal dose is 10mg/kg given 3 or 4 times daily. Andersson et al34 recommend reducing dosages in patients with decreased renal function as follows: for serum creatinine levels of 120-250µmol/l (1.36-2.83mg/dl), 10mg/kg IV twice daily; for serum creatinine levels of 250-500µmol/l (2.83-5.66mg/dl), 10mg/kg IV once daily; for serum creatinine levels ≥500µmol/l (5.66 mg/dl), 10mg/kg IV every 48 hours. Similar doses have been used with good results in patients on dialysis with upper digestive tract haemorrhage.16
Treatment options for controlling severe haemorrhage from polycystic kidneys are limited.35-39 As far as we are aware, there have been only two published studies of tranexamic acid as treatment for haematuria due to cyst bleeding in ADPKD, and both were isolated cases.9,10 In one of the cases, the initial dose was 20mg/kg IV, which was later maintained by the oral route.9 In the other case, which was treated with tranexamic acid on multiple occasions, the dosage varied between 15mg/kg IV 3 times daily and 10mg/kg once daily orally.10 Treatment duration also varied from 3 to 5 days in the first case and 1 to several weeks in the other one. While results from this study are preliminary, they suggest that in the most severe cases of uncontrolled haemorrhage it would be reasonable to administer anti-fibrinolytic treatment with tranexamic acid. Our patients received doses ranging from 15mg/kg 3 times daily IV to 10mg/kg 3 times daily orally with treatment lasting between 2 and 5 days. All courses of treatment were equally effective. No side effects were observed at any time. However, we recommend administering oral or IV tranexamic acid at the lowest possible effective dosage and during the shortest possible period in order to lessen the risk of adverse effects. In our patients, this dose was 10mg/kg 3 times daily orally during 3 to 4 days. It is possible that treatment lasting less than 3 days could be just as effective, but this type of dose was not studied. Lastly, early at-home use of tranexamic acid by patients with ADPKD and recurrent haematuria could be very cost-effective if it prevented the need for hospitalisation. The main limitations of this study are a small sample size and the lack of an untreated control group.
In summary, tranexamic acid may be used safely in selected ADPKD patients with severe or intractable haematuria that does not respond to conventional treatment. The drug can be administered orally or intravenously, and the dose must be adjusted in patients with kidney failure. Treatment with tranexamic acid in ADPKD may preserve renal function both directly, by stopping cyst bleeding and decreasing the frequency of haematuria episodes, and indirectly, by preventing embolisation and/or nephrectomy.
Acknowledgements
This study was partially funded by a grant awarded to R.P. by Instituto de Salud Carlos III, the Spanish Ministry of Science and Innovation (EC08/00236) and the research activity intensification programme (Programa Intensificación Actividad Investigadora) at IdiPAZ and Lain-Entralgo/CM.
Conflicts of interest
The authors affirm that they have no conflicts of interest related to the content of this article.
Table 1. Clinical data from ADPKD patients before and after medical therapy with tranexamic acid
Figure 1. Magnetic resonance of Case 6 showing numerous renal cysts with signs of intracystic bleeding and fluid-fluid levels