Aims: To describe PD outcomes over 25 years in a single centre, comparing hospitalisation rate, technique withdrawal, and survival between diabetic (DM) and non-diabetic (NonDM) patients. Differences between type 1 (DM1) and type 2 (DM2) diabetics were also analysed. Patients and methods: One hundred and eighteen DM patients (52 year old average, 74 men, 44 female) and 117 Non-DM (53 year old average, 64 men, 53 female), with at least 2 months on PD, 25±20 (2-109) and 29.4±27 (2-159) months respectively, were included. Diabetics were divided in 66 DM1 and 52 DM2. The survival and hospitalisation study was also analysed in two different time periods: before 1992 (1981-1992) and after 1992 (1993-2005). Results: 93% Non-DM and 75% DM were self-sufficient to manage the PD technique (P<.001) as well as 65% of 44 blind patients. 28% of Non-DM and 15% of DM received a renal allograft (P<.001). There was no difference in transfer to haemodialysis. 18.6% of DM and 4.3% of Non-DM patients presented >4 comorbid factors on starting PD (P<.001). Hospitalisation (admissions/year) was higher in DM than in Non-DM patients (3.4 vs 1.8, P<.01) and also hospitalisation length (46 vs 22 days/year, P=.01), without differences between DM1 and DM2. Admissions due to cardiovascular events, infections, technical problems and peritonitis were more frequent in DM2 than in Non-DM and DM1 patients (P<.05). However, DM2 patients admitted to hospital for peritonitis did not spend more days in hospital than Non-DM or DM1 patients. Mortality was 48% in DM and 22% in Non-DM (P<.001). Survival adjusted for comorbidity was higher in Non-DM (P<.001). Cerebrovascular disease was the highest risk factor for mortality in DM. Mortality was higher in DM2 than in DM1 and Non-DM(P<.001). Age (HR 1.052, P=.001), DM2 (HR 1.96, P<.01) and cerebrovascular disease (HR 4.01, P<.001) were the most important risk factors. In the post-1992 period, the hospitalisation rate and survival improved in DM1 and Non-DM patients. Conclusions: DM patients more often require outside assistance to perform PD and have more comorbidity, lower survival, and higher admissions than Non-DM, but there is no difference in HD discontinuation. Age and cardiovascular comorbidity are the factors involved in mortality. Technological advances and cumulative center experience may achieve dialysis outcome improvements in diabetic patients.
¿
Objetivos: Describir la experiencia de 25 años de tratamiento con diálisis peritoneal (DP) en un solo centro, comparando la hospitalización, abandono de la técnica y supervivencia entre pacientes diabéticos (DM) y no diabéticos (NoDM) y analizando las diferencias entre diabéticos tipo 1 (DM 1) y tipo 2 (DM 2). Material y métodos: Se incluyen 118 DM (52 años, 74 hombres y 44 mujeres) con, al menos, 2 meses de permanencia en DP y media de 25 ± 20 meses (2-109), divididos en 66 con DM 1 (45 años) y 52 con DM 2 (65 años) y 117 NoDM (53 años, 64 hombres y 53 mujeres), con un tiempo en DP de 29,4 ± 27 meses (2-159). Por el largo período estudiado, en el análisis de hospitalización y de supervivencia se evalúa, además, el seguimiento en dos períodos: 1981 a 1992 (pre-92) y 1993 a 2005 (post-92). Resultados: El 93% de los NoDM y el 75% de los DM fueron autosuficientes para realizar DP (p <0,001) y también el 65% de 44 pacientes ciegos. Han sido sometidos a trasplante el 28% NoDM frente al 15% DM (p <0.001) y no hay diferencia en la transferencia a HD. El 18,6% de los DM frente al 4,3% de los NoDM (p <0.001) presentan cuatro o más factores comórbidos al iniciar DP. La hospitalización (ingresos/año) fue mayor en DM (3,4 frente a 1,8) que en NoDM (p <0,01) y también los días/año (46 frente a 22; p <0,01), sin que exista diferencia entre DM 1 y DM 2. Los ingresos por causas cardiovasculares, infecciones, problemas técnicos e infección peritoneal fueron más frecuentes en DM 2 (p <0,05) que en NoDM y DM 1, pero no los días de ingreso por peritonitis. El 48% de los DM y el 22% de los NoDM fallecen (p <0,001). La supervivencia ajustada a factores de comorbilidad es mayor en NoDM (p <0,001), con la enfermedad cerebrovascular como factor mayor de impacto en la mortalidad de DM. La mortalidad es mayor en DM 2 que en DM 1 y NoDM (p <0,001). La edad (HR 1,052; p <0,001), la condición de DM 2 (HR 1,96; p <0,01) y la enfermedad cerebrovascular (HR 4,01; p <0,001) son los más importantes factores de riesgo. En el período post-92 mejora de manera importante la tasa de hospitalización y la supervivencia de pacientes NoDM y, sobre todo, de DM 1. Conclusión: Los pacientes con DM precisan más frecuentemente ayuda para realizar la DP y presentan más comorbilidad, menor supervivencia y mayor hospitalización que los pacientes NoDM, mientras que es comparable la tasa de abandono de la técnica. La edad y las complicaciones cardiovasculares (sobre todo cerebrales) son los factores implicados en la mayor mortalidad. Los avances tecnológicos y la mayor experiencia de los centros pueden mejorar las expectativas de los DM en diálisis.
INTRODUCTION
At present, diabetic nephropathy is the leading cause of inclusion of patients with chronic kidney disease in dialysis programmes.1,2 Because of the difficulty of creating vascular access for haemodialysis (HD) and the haemodynamic instability in diabetic patients, during the eighties there were abundant descriptions of promising experiences with patients in continuous ambulatory peritoneal dialysis (CAPD).3-6 Good haemodynamic tolerance and good glycaemic control with intraperitoneal insulin were the keys to optimism in using CAPD as preferential treatment in diabetics. The frequency of complications in diabetic patients treated with peritoneal dialysis (PD) in all its forms was higher than in non-diabetic patients, in relation to the severity of multi-organ systemic diseases such as diabetes mellitus (DM). After a period of frequent publications on the development of diabetic patients on dialysis, in recent years literature searches only reveal reviews of the topic and there are few original articles. However, the characteristics of diabetic patients starting haemodialysis (HD) or PD in recent years have changed, and unlike the eighties and nineties, the inclusion of diabetic patients on dialysis today occurs if they are suffering from Type 2 diabetes. DM has become a true pandemic, with a higher prevalence of adult DM.7 This difference can change the outcome of patients on renal replacement therapy.
The aim of this study is to provide the experience of a single centre over 25 years of treating diabetic patients with PD, analysing survival and hospitalisation in relation to non-diabetic patients, and studying the difference between Type 1 and Type 2 diabetics.
PATIENTS AND METHOD
After years of intermittent use of PD in a small number of patients and the description of CAPD in 1977, the Hospital Clínico San Carlos (Madrid) started the CAPD programme in 1981 and in 1982 they included the first diabetic patient. It is a retrospective, observational study on patients who started the PD programme at this centre, since the programme's inception until 2005. Data has been collected from 235 patients with PD stays over 2 months and with sufficient documentation to follow-up (in 12 patients the clinical history data were insufficient to evaluate), 118 diabetics (50.2%) who met the criteria for diabetic nephropathy and 117 non-diabetics (Non-DM) (49.8%). Demographic data on DM and Non-DM are shown in Table 1. Age, gender distribution, PD time and accumulated follow-up time were not different between DM and Non-DM patients. The DM patient group consisted of 66 Type 1 DM, 44.9±10.4 years (41 men and 25 women) and accumulated follow-up time of 2.08 years, and 52 Type 2 DM, 62.6 years (33 men and 19 women) and 2.1 years accumulated follow-up time.
The aetiologies of the renal disease most common in Non-DM patients are divided into interstitial 14.4%, 12.6% vascular-ischaemic, 6.5% glomerular, 4.8% polycystic disease, and the rest belong to other causes and unknown origin.
Due to changes that occur over such a long period of follow-up in terms of material, techniques and experience, the study of hospitalisation and survival is also performed by dividing the 25 years of experience into two periods: the first period, from 1981 to 1992 (pre-92) and the second period, from 1993 to 2005 (post-92). Major developments that occurred from the nineties and that justify this division include erythropoietin, which began to be used in 1990, CAPD double bag systems that were introduced in 1992 and automated PD (APD) with cyclers, which began to be implemented in Spain in the early nineties.
In the results sections the following aspects are evaluated: 1) self-sufficiency in carrying out PD, 2) number and frequency of comorbid conditions at the start of PD, such as obesity, hypertension, heart failure, heart disease, cerebral vascular disease and peripheral vascular disease, 3) discontinuation of the technique, 4) hospitalisation and causes of admission, 5) patient survival, and 6) causes of death. In all cases, we compared data from patients with DM against those without DM, and between Type 1 and Type 2 DM.
Statistical Analysis
The continuous variables are expressed as average and standard deviation (SD). Comparisons were carried out using the Student's t or Chi-squared tests according to the nature of the variables. Survival was analysed using the Kaplan-Meier log rank test and confidence intervals (CI), considering other events as appropriate and the forward conditional Cox regression model to identify the influence of risk factors. In terms of patient mortality, the event is death; and leaving the programme for any other reason (transplant, transfer, etc.) is considered a loss. Discontinuation of the technique includes the move to HD, transplantation and recovery of renal function; the failure of the technique only includes transfer to HD.
The heterogeneity of the groups were analysed with the Chi-squared test for N-1 degrees of freedom with an alpha of .05 for statistical significance. Data are expressed as mean survival probability with 95% CI. Data were processed with the SPSS 16.1 statistical software for Windows.
RESULTS
Prevalence
DM prevalence and type changed over the two periods analysed, so that in the period from 1981 to 1992, 58% of patients were diabetic and in the period from 1993 to 2005 the percentage dropped to 40.5%. Meanwhile, the type of diabetes changed in the two periods, with a DM1 percentage of 39.5% and DM2 of 18% in pre-92, which is reversed to 16.5 and 25.9%, respectively, in the post-92 period. 93% of Non-DM and 75% of DM were self-sufficient for PD (P<.001) and also 65% of 44 blind patients or with severe impairment of visual acuity (legally blind). At the beginning of PD, DM patients had high comorbidity, higher than Non-DM patients (Table 1). 18.6% of DM compared to 4.3% of Non-DM (P<.001) had four or more risk factors.
Admissions
DM patients are admitted more than Non-DM (1.38±1.1 vs 0.88±0.9 admissions/year, P<.001) and have more days of accumulated stay (20.7±25.4 versus 13.2±19.0 days/patient/year, P=.018). Peritoneal infection is the leading cause of hospital admission in all patient groups, and has a higher rate for DM than Non-DM (33 vs 28%, P<.05) (Table 2). This is due mainly to the subgroup of Type 2 diabetes, with 46.2% of admissions due to peritoneal infection compared to 22.7% of Type 1 DM (P<.001) (Table 2). However, no significant differences were found between Type 1 DM and Type 2 DM in the accumulated number of stays per year due to peritoneal infection (11.1±18.6 versus 7.8±14.0 days/patient/year, P=.150). The percentage of patients hospitalised due to infectious peritonitis, cardiovascular or dialysis technique-related problems, is shown in Table 2.
When analysing separately the two periods into which we divided the study, we see a progressive tendency towards reduced admissions in all subgroups of patients. Therefore, the Non-DM move from 1.2±1.1 in the first period (pre-92) to 0.63±0.64 in the second period (post-92) (P<.01), with a reduction in inpatient days (32.8±25.8 compared with 15.1±22.6, P<.01). The same applies to DM patients, whose admission rate decreases from 1.58±1.18 in pre-92 to 1.13±1.0 in post-92 (P<.01), with a consequent reduction of days of accrued stay (51±61 versus 40.6±48.7, P<.01).
Technique Change
The analysis of technique changes are detailed in Table 2. There were no differences in the transition to HD between DM and Non-DM but surprisingly, the move to HD is more frequent in Type 1 DM (39.4%) than in Type 2 (13.5%), (P<.001). As expected, kidney transplantation was the main reason for discontinuing DP. As such, the amount of Non-DM patients who discontinued DP for this reason was twice that of DM patients (Table 2). Among diabetic patients who underwent transplantation, 22.7% have Type 1 DM compared to only 5.8% of Type 2 DM.
Survival
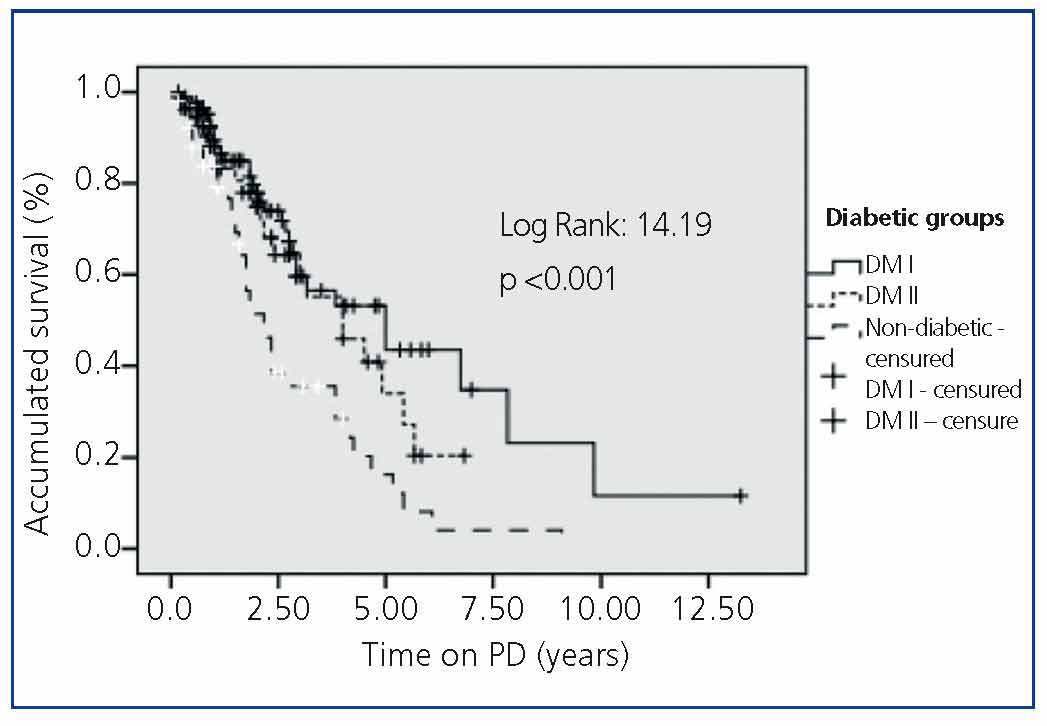
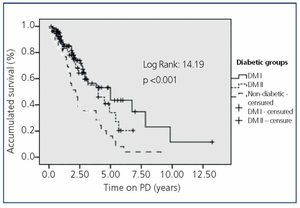
Some 48.3% of DM patients died in PD during the study period, compared to 27.4% of Non-DM patients (P<.001), with a HR of 1.96 (95% CI, 1.1-3.3). The Kaplan-Meier analysis (Figure 1) reflects a higher survival rate for Non-DM patients, although for up to 4 years they show a similar survival as Type 1 diabetics, about 60%, and always higher than Type 2 DM. The HR of Type 2 DM patients compared with Non-DM is 2.18 (95% CI, 1.042-4.51). If we carry out a forward, stepwise multivariate Cox regression analysis, based on the age and added comorbidity, the presence of Type 2 diabetes with a HR of 1.96 (95% CI, 1.13-3.39, P<.01), along with age with a HR of 1.052 (1.019 to 1.079) (P<.001) and cerebrovascular disease (HR 4.013, 95% CI, 2.119-7.601, P<.001) are the factors with greater weight in terms of mortality.
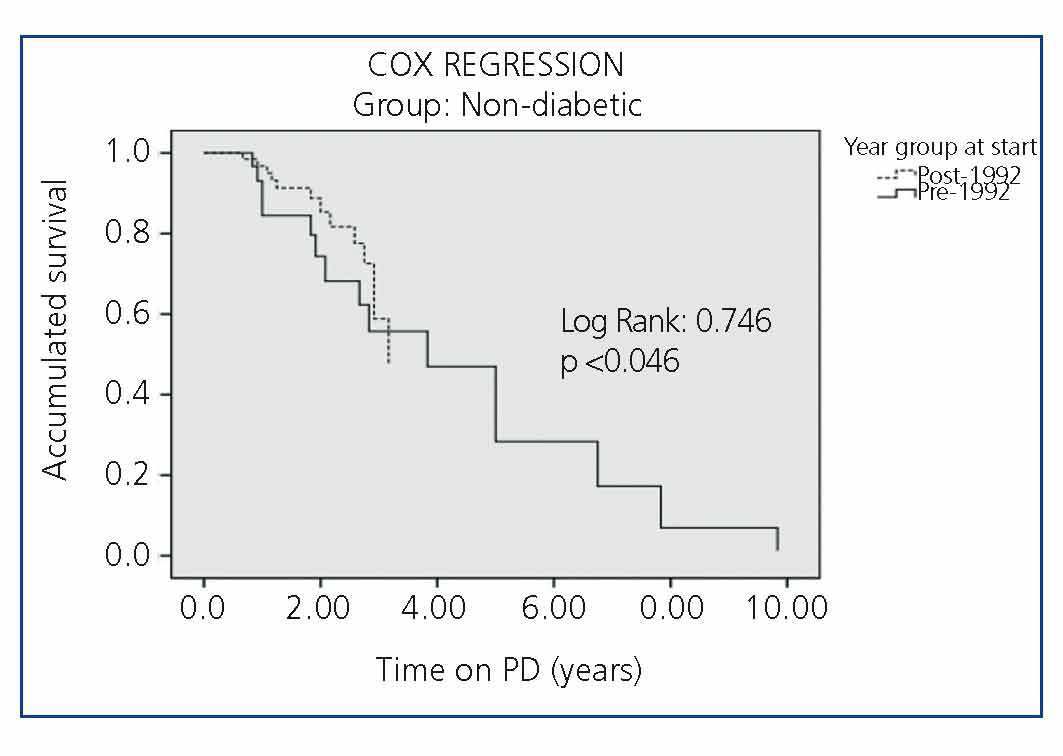
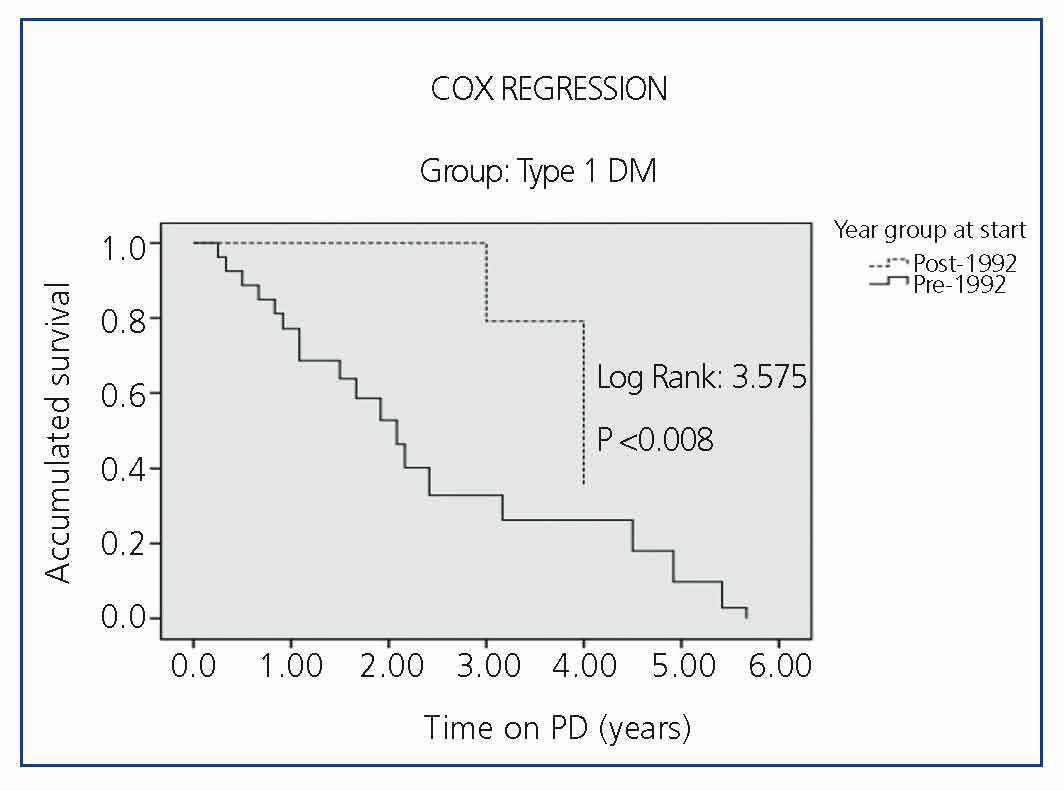
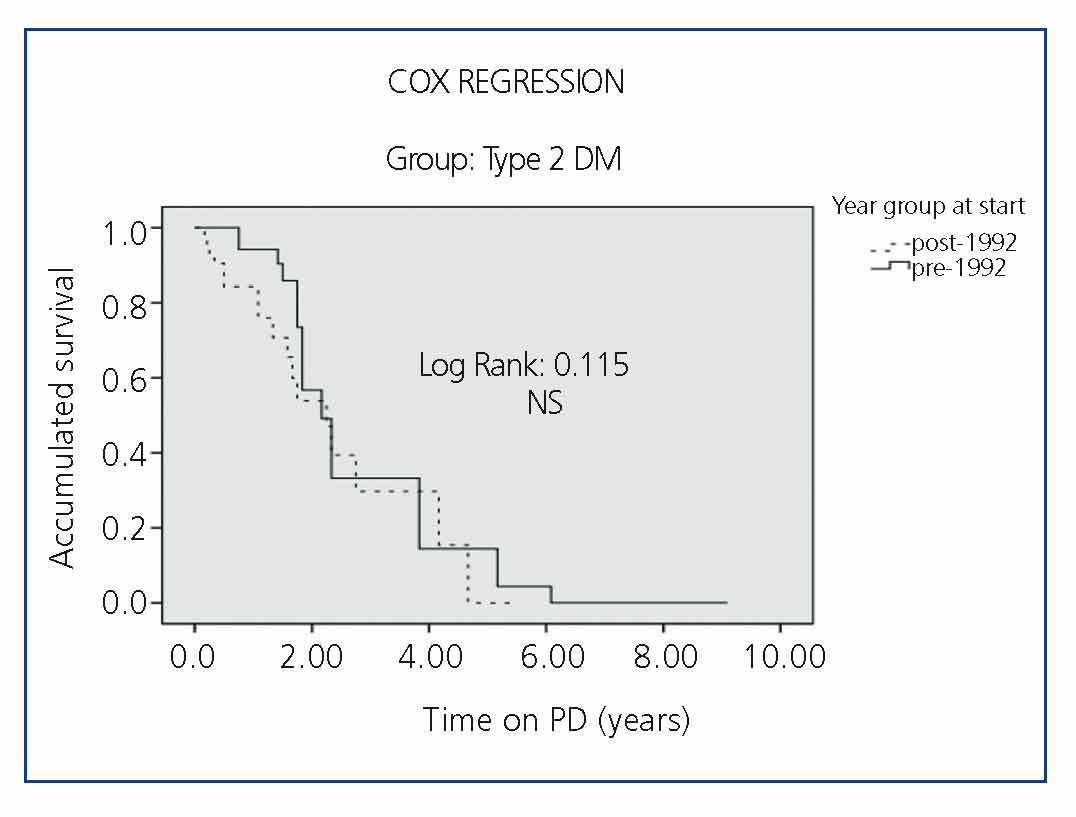
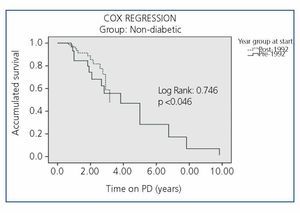
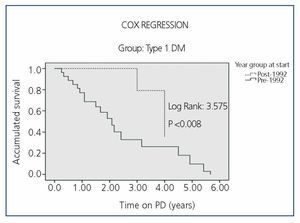
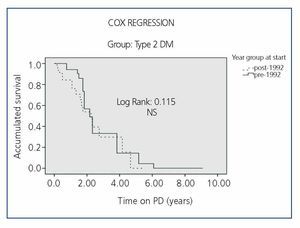
Survival by periods for each of the subgroups (DM type 1, DM Type 2 and Non-DM) is shown in Figure 2, Figure 3 and Figure 4, where we can see a slight difference in survival between the two periods in Non-DM patients (P<.046) and a significant increase in DM1 patients (P<.008), without significant differences between the two periods in DM2 patients. In the Cox regression analysis by periods, Non-DM patients hypertension (HR 1.6, 95% CI, 1.17-1.86, P=.017) and stroke (HR 4.7, 95%, 1.6 to 14.4, P<.001) mark the difference between the two periods. In DM1 patients it is ischaemic heart disease (HR 2.6, 95% CI, 0.70-9.92, P<.001). In DM2 patients, it is heart failure (HR 1.64, 95% CI, 0.67-3.93, P<.001) and stroke (HR 6.94, 95% CI, 2.32-20.7, P<.001) in both periods.
The leading cause of death in DM patients is a cardiovascular event. Some 15.3% died due to heart problems compared with 8.5% of Non-DM (P<.001) and stroke (8.5% in DM compared to 2.6% in Non-DM, P<.01). Peritoneal infection as a cause of death is equal in both DM and Non-DM (5.1 vs 5.1%).
DISCUSSION
Our study describes the experience in treating PD at a single centre over 25 years. Overall we found the best results in non-diabetic patients in the most recent period (post-1992). Although previous studies highlight the weight of DM as a prognostic factor, it is important to have data from our area and for such a long period.
Most diabetic patients had injury in various organs and systems at the time of starting PD, which determines a high comorbidity. This, in turn, can influence their adaptation to the technique, their maintenance on it and their survival. With regard to adaptation, it is relevant that we have achieved a sufficient level of self-care so that the patient is responsible for their dialysis, even with the large number of DM patients who are blind. As for continuation of the technique and survival, our data shows increased mortality, especially cardiovascular, as other authors have also reported.8, 9
One of the merits of this study is that it brings together a long experience in a single centre, more than 25 years. This has allowed us to differentiate two time periods, in which progressive improvement is seen, and although DM patients have a worse prognosis in both periods, in post-92 an improvement is seen in survival in Non-DM and DM1 patients. The worst prognosis of DM patients is often reported in the medical literature until the late nineties10-13 and more sporadically in recent years.14-16 Accordingly, the long-term trends described here are influenced by changes in the prescription of dialysis over time, improvements in technology and greater experience in treating these patients.
Dividing the study into two phases, with a time point in 1992, coincides with major changes in PD technology, such as the consolidation of double-bag systems and the introduction of new cyclers that have allowed an increasing number of patients in ADP. A learning curve is common in almost all complex medical activities, but here it is clear that this improvement in results is not limited to the first months or years of application of the technique, but persists in time and can be maintained for years.17 This improvement in overall performance is more evident in younger patients with Type 1 diabetes, which have reduced mortality in the comparison between the two periods.
In one of the few recent studies on the evolution of DM patients on PD, Fang et al.15 indicate advanced age as the most important factor affecting mortality in diabetic patients. Our results are along the same lines, and age, along with cardiovascular comorbidity are the most significant factors with regards the mortality of our patients. Other authors report similar results, describing how heart disease primarily affects DM2 patients.14,16 In our study the CV event with the greatest weight on outcome is stroke (CVA). Type 2 DM patients are older and have a higher prevalence of stroke, and the three factors combine to worsen the prognosis of these patients. Others have suggested the prognostic value of cerebrovascular disease in DM patients, both in PD18 and in HD.19 In a previous study by our group on survival in DM patients and the renal function with which they started PD, cerebrovascular disease and heart failure also appear as the factors with greatest impact on mortality.20
The Type 1 and Type 2 DM patient profiles are completely different, and therefore we analysed both groups separately. Type 1 DM patients had similar survival rates to the Non-DM in the first 4 years of treatment, as shown in Figure 1, and improved survival and hospitalisation in the post-92 period, which is not seen in the DM2 patients.
We found a higher hospitalisation rate in diabetic patients, especially in Type 2 diabetics, although some studies do not report differences in hospitalisation between DM and Non-DM on PD.14 We have previously discussed how technical advances can have lead to the improved results for the second period. One recent study, with a similar design which divides a long-term monitoring in two periods of time, explained how technological advances in PD may influence the results.21 A significant percentage of DM patients underwent kidney transplantation, although due to their higher comorbidity, it was a lower number than Non-DM. The transfer to HD due to failure or fatigue of the technique is, however, same for the DM and Non-DM patients.
This study has several limitations. On one hand, a long evolution involves the incorporation of changes and improvements in treatment, but the prognosis for DM is maintained in both periods. On the other hand, as it is a single-centre study, we cannot ensure that results can be generalised. This is a retrospective study with asymmetry in the size of the groups, which limits the survival analysis. However, we have included all patients who have gone through the PD programme, data has been reviewed and a correct analysis was carried out. Therefore, this study provides a good description of PD treatment in the real world, away from the constraints of clinical trials.
In conclusion, in the 25 year monitoring, diabetic patients had worse clinical status at the start of PD and had a poorer outcome in overall results such as hospitalisation and patient survival. The leading cause of death is a cardiovascular event, and it is possible that the vascular damage present before the start of the PD affects these results. Therefore, DM patients require special attention from the CKD early stages. The programme experience and developments in PD may be responsible for the better results with Type 1 DM patients in the second half of the period.
Acknowledgements
Thanks to Dr J. Portolés for his comments and advice on the statistical treatment of the study.
Figure 1. Survival of Patients with Type 1 Diabetes (DM1), Type 2 (DM2) and Non-Diabetic (ND) in Peritoneal Dialysis (PD)
Figure 2. Comparison of the Survival of Non-Diabetic Patients in the Two Study Periods
Figure 3. Comparison of the Survival of Type 1 Diabetic (DM1) Patients in the Two Study Periods
Figure 4. Comparison of the Survival of Type 2 Diabetic (DM2) Patients in the Two Study Periods
10553_108_10786_en_w4777105871110553_t1__en.ppt
Table 1. Characteristics of diabetic and non-diabetic patients on PD and risk factors upon initiation of PD
10553_108_10787_en_w4777105871010553_t2_en.ppt
Table 2. Causes of Hospitalisation and PD Discontinuation in Diabetic and Non-Diabetic Patients