To the Editor,
CASE REPORT
44-year-old patient, nurse, with no history of interest except hypertension (AHT) of 10 years progression, being treated with enalapril 30 mg, nicardipine 10 mg and hydrochlorothiazide 25 mg, without regular monitoring of blood pressure (BP). The patient sought consultation due to raised levels of BP (160-180/95-100mmHg) for 4-5 months with frequent hypertensive crisis that caused admission to the Emergency Department. The patient did not show signs of taking non-steriodal anti-inflammatory drugs, nor history of lithiasis or urinary infection, although chronic inguinal pain on the left side contributed to dysmenorrhoea and no study was carried out. Examination revealed: body mass index 29 kg/m2, BP 167/94 mmHg, 87 bpm, normal cardiopulmonary auscultation, enlarged abdomen and minimal bilateral malleolar oedema.
Complementary examinations were also performed, with the following results:
- Ambulatory BP monitoring (ABPM): diurnal average 151/80mmHg, 61bpm, pulse pressure 70.6mmHg; nocturnal average 137/70mmHg, 57bpm, pulse pressure 66.8mmHg. Maximum diurnal BP 183/99mmHg, maximum nocturnal BP 171/95mmHg. Non-dipper BP pattern.
- Electrocardiogram: sinus rhythm 72bpm; Sokolow-Lyon index <35mm; asymmetric reversal of T-wave.
- Echocardiogram: interventricular septum 12mm.
- Normal thoracic radiographs.
- Normal funduscopy.
- Abdominal ultrasound: right kidney (RK) 13cm, normal differentiation; left kidney (LK) 25cm, hydronephrotic, without corticomedullary differentiation.
- Biochemical tests showed: normal haemogram, hepatic lipid profile, uric acid, ions and glycemia. Urea: 31mg/dl, creatinine 0.79mg/dl, glomerular filtration rate (MDRD-4) >60ml/min/1.73m2.
- Plasma renin activity (PRA) 1.35ng/ml/h (normal radioimmunoassay range 0.2-3.3ng/ml/h).
- Urine: proteinuria 0.11g/24hr, creatinine 161mg/d, Na 241mEq/l, fractional excretion of Na 0.78%. Sediment: 5 red blood cells/field, 30 leukocytes/field, negative nitrites; creatinine clearance 113ml/min.
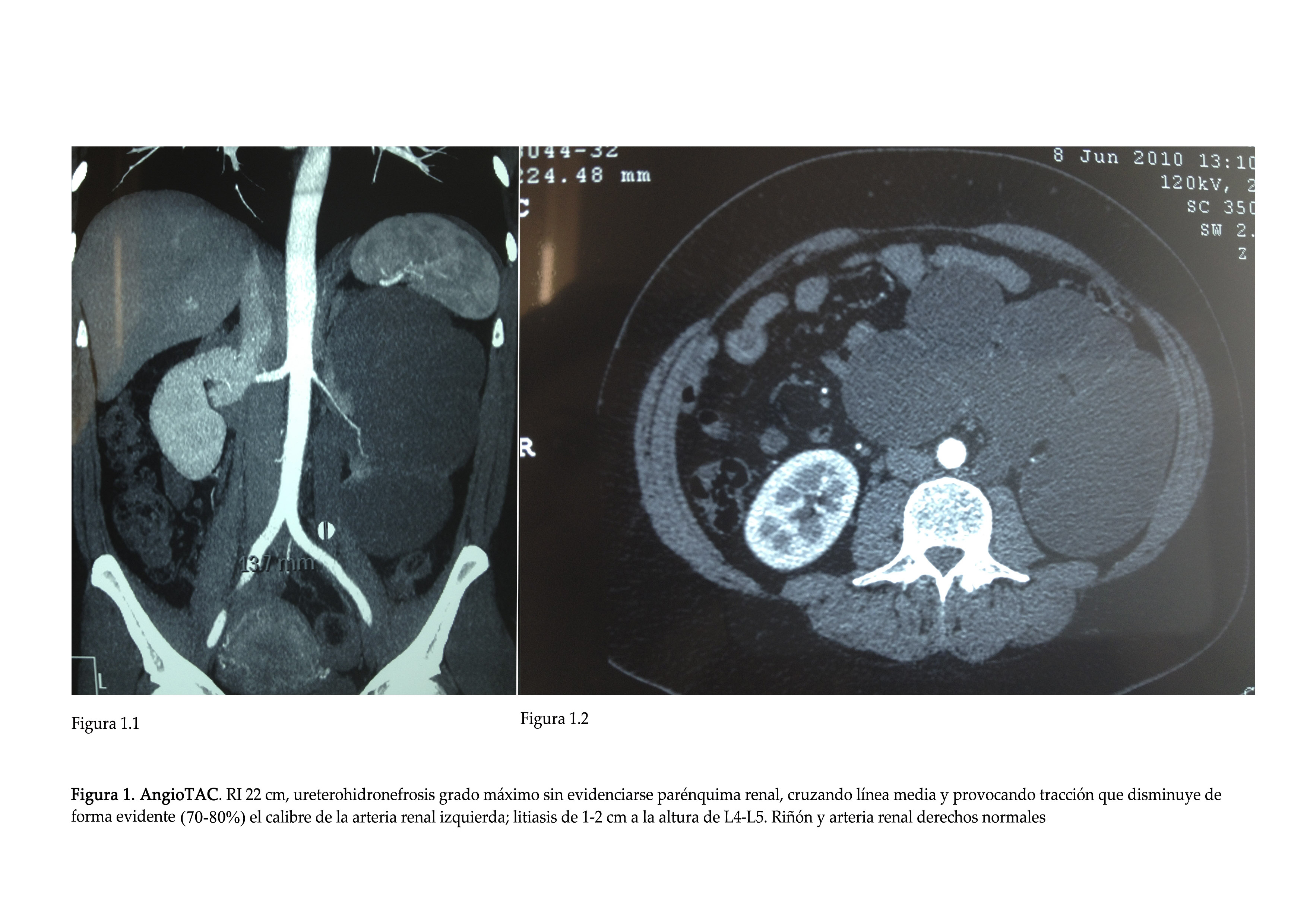
- CT Angiogram (axial computed tomography): lithiasis and maximum ureterohydronephrosis on the left-side that caused traction of the ipsilateral renal artery (Figure 1).
- Technetium Tc 99m Dimercaptosuccinic Acid Scintigraphy: DR 100 %, IR 0 %.
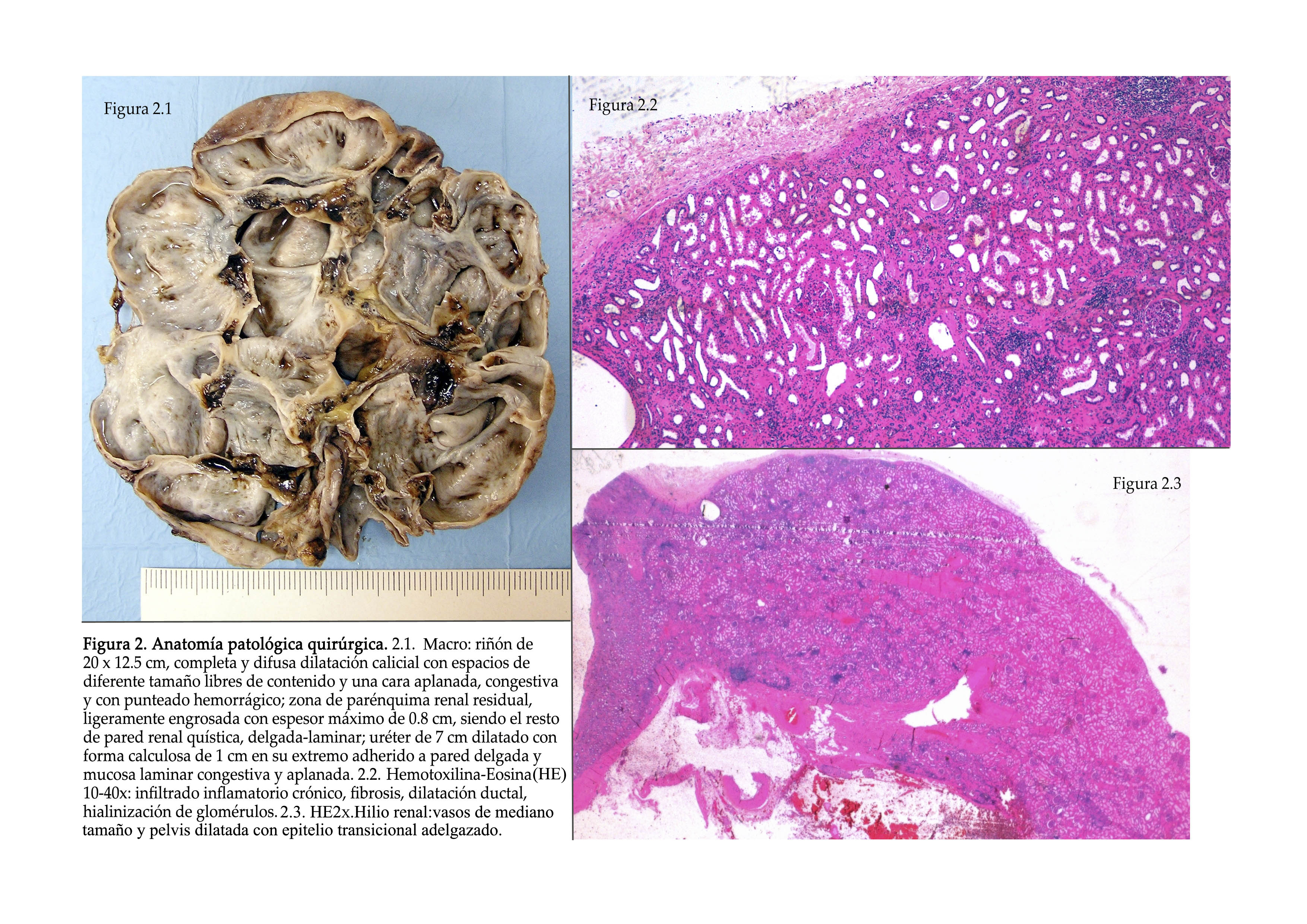
The patient was diagnosed with moderate risk stage 2 HBP, initially of renovascular aetiology and failure of the RK, and it was decided to carry out laparoscopic nephrectomy on the left side which went smoothly. The anatomical pathology of the specimen reported a hydronephrotic kidney with lithiasis, chronic pyelonephritis in the stage of exacerbation, acute fibrino-hemorrhagic pyelonephritis, uriniferous cysts and chronic erosive urethritis (Figure 2).
Progress from a clinical point of view was favourable. Renal clearance and creatinine slightly and temporarily deteriorated following nephrectomy, returning to their baseline levels in less than 15 days. BP dropped signficantly. Postoperative renogram revealed a RK with good vascularisation and radiotracer uptake capacity, as well as adequate response to the diuretic. Sequential images and the renographic curve showed adequate elimination, without ectasia or obstructive behaviour. At present, three years after surgery, BP remains well controlled (self-measurement and ABPM) with lecarnidipine 20mg/day (10mg in the summer), and presents 1.1mg/dl of creatinine, clearance of 71ml/min and proteinuria 0.09g/day. The patient has not re-experienced inguinal pain.
DISCUSSION
Nephrolithiasis, in spite of techniques such as ureteroscopy, lithotomy or lithotripsy, is one of the causes that lead to the development of chronic renal failure.1 The asymptomatic form is more frequent than expected, reaching 11 % prevalence in a study carried out in potential donors.2 The evolution of these “healthy carriers” of nephrolithiasis has not been studied, the risk of chronic kidney disease being controversial, which seems to relate to the size, the composition and the location of the calculus, among other factors.3-7 The same occurred with the asymptomatic bacteriuria, whose need for antiobiotic and/or surgical treatment is dependant on, in addition to microbiology and the age of the patient, the existence or not of morphological changes in the urinary tract, since it seems that these are one of the main factors that directly influence the perpetuation of renal failure.8 In our case, there was no possibility of this approach, since the diagnosis of both disorders was carried out simultaneously and the renal failure of the suffering kidney was already established. The microbiology and the composition of the calculus was unknown in our case (histological examination of the lithiasis was not carried out because urine calcium oxalate was normal). In addition, there was no family history of lithiasis and no prevous imaging of the reno-ureteral system. Thus, we did not know whether the liathisis was the cause of the infections or viceversa.9
In terms of imaging diagnosis, in the ultrasound there was no clear image of the nephrolithiasis, likely due to the location of the calculus obstruction, clearly showing up in the CT scan and in the surgical specimen. The CT has, likewise, greater sensitivity than the ultrasound to detect nephrolithiasis <5mm, as well as other renal anomolies.10 A Doppler ultrasound was not carried out because of the anthropometric characteristics of the patient (obesity), directly indicating CT-angiogram under protocol of nephroprotection, given the presumption of unilateral functional nullity.
We understand the pathophysiology as to why this patient developed HBP at 34 as multifactorial: a) the stretching of the renal artery due to traction of the hydronephrotic kidney caused its stenosis, and subsequent activation of the renin-angiotensin-aldosterone system (RAAS), similar to what happens in some cases of renal ptosis.11 Although we do not have previous images to establish a differential diagnosis of hypoplastic renal artery, the patient presented ultrasound pregnancy check-ups without changes; b) nephrolithiasis determined a state of obstructive uropathy and with that the formation of uriniferous cysts that compressed the renal parenchyma, also activating RAAS;12 c) interstitial inflammation as an immune response to the urinary infection reduced the area of the peritubular capillaries, hindering the mechanism of natriuresis because of pressure.13 The PARA of the patient was within the normal range because of its suppression in the contralateral kidney, as well as, in the majority of individuals, this figure being normal once HBP was established.14 The elevated excretion of sodium in the urine indicated increased ingestion, and its fractional excretion < 1 %, an increase in its tubular reabsorption (obstructive uropathy that can also present itself in the non-functioning kidney with 0 % of participation in the scintography), despite presenting low doses of thiazide diuretic.
The laparoscopic nephrectomy was carried out routinely in patients with complicated lithiasis and also in renal artery stenosis, both separately.15,16 What is unique about our case is the conjunction of both illness and the mechanical aetiological component of arterial stenosis, not giving an option for endovascular treatment.
Having revised the histology of the specimen, although the extensive fibrosis did not allow the aetiology of the chronicity to be well seen, it seems that the predominant factor of nephropathy could be the tubulo-intersitial, caused by primary infection and/or obstruction. Ischaemic nephrosclerosis, reflected as areas of haemorrhage and intimal thickening, seems to remain, at least, in the background in this case.
CONCLUSION
Different factors can cause HBP. Giant hydronephrosis can cause stenosis by stretching the renal artery, causing renovascular hypertension. Nephrectomy is the main therapeutic option in cases of functional nullity. Urinary infections and nephrolithiasis can also contribute to the development of HBP, but the necessary approach when faced with bacteriuria and/or nephrolithiasis remains in controversy, unless morphological changes develop which are perpetuated by the factors that predispose the development of chronic renal disease.
Conflicts of interest
The authors declare that they have no conflicts of interest related to the contents of this article.
Figure 1. CT Angiogram.
Figure 2. Surgical anatomical pathology.