Introducción: Los resultados de los trasplantes efectuados con donantes con criterios expandidos (DCE) son inferiores a los obtenidos con donantes con criterios estándar (DCS). Para optimizar su evolución, se podría reducir su tiempo de isquemia fría (TIF) reduciendo su daño de preservación. Comparamos los resultados obtenidos al aplicar TIF <15 horas tanto a DCE como a DCS. Material y métodos: Realizamos un estudio unicéntrico, de cohortes, prospectivo, de casos incidentes de trasplante renal de cadáver entre junio de 2003 y diciembre de 2007. El tiempo mínimo de seguimiento fue de 12 meses. Comparamos los datos de los donantes, de los receptores y de la evolución de los trasplantes efectuados con DCE frente a los de los DCS. Resultados: El TIF para los DCE (N = 24) y para los DCS (N = 50) fue, respectivamente, de 9,3 ± 2,5 y 8,3 ± 3,3 horas (p = 0,18). No encontramos diferencias significativas entre los receptores de DCE y DCS en cuanto a: no función primaria del injerto 4,2 vs. 4%, retardo en la función del injerto 16,7 vs. 10%, complicaciones quirúrgicas 25 vs. 16% y rechazos agudos 8,3 vs. 2%. El filtrado glomerular estimado al año para los DCS fue de 65,8 ± 14,9 ml/min y para los DCE de 49,4 ± 12,5 ml/min (p <0,0001). La supervivencia renal al año fue del 95,8% para los receptores de DCE y del 94% para los DCS (p = 0,75). Conclusiones: La aplicación de TIF cortos a los DCE permite conseguir una evolución similar a la de los DCS, aunque su función renal sea en todo momento inferior.
INTRODUCTION
An ECD is defined as a donor of a kidney whose relative risk of failure is 1.7 times greater than that of a kidneyprovided by an ideal donor.1 ECD implants have an estimated survival rate of 92.3% at three months, 84.5% at one year, and 68% at three years, while standard criteria donor (SCD) kidney transplants have survival rates of 94.6, 90.6 and 79.4%, respectively, for the same time periods.2
Despite having a worse prognosis, the use of ECD kidneys has been completely justified ever since it was shown that the survival time for the recipient of this type of graft is greater than that of a person remaining on the waiting list3. ECD organs are increasingly common, and their use is practically mandatory if we are to maintain the current transplant rates in order to satisfy the continuous and increasing demand for grafts.
With this in mind, we must adopt the measures that are necessary in order to optimise ECD transplant results.4 To do so, we have proposed selecting recipients correctly and attempting to reduce damage during organ preservation.
Regarding the recipient selection aspect, these organs are not recommended for repeat transplants or for patients younger than 40,5 but are recommended for recipients with a low metabolic demand (the elderly and patients with a low body mass index) and for patients with little immunologic risk (low PRA score).6 There has even been a proposal to match up donors and recipients according to estimated survival profiles.7
There are two strategies for attempting to limit damage during preservation: preserving organs with machine perfusion8 or reducing cold ischaemia times (CIT).9
Prolonged CIT is associated with delayed organ function, and both factors lead to increased rejection rate and hospitalisation time, worse renal function and a decreased long-term survival rate. CIT has also been described as an independent risk factor in organ survival with donors younger than 50 (SCDs).10 As a result, it seems reasonable to attempt to optimise the results of ECD organs using short CITs.
In this study, we will compare the evolution of transplanted ECD and SCD organs in a transplant programme that uses short CITs in order to reduce both the delay in graft function as well as the acute rejection rate, thus improving survival and renal function.
MATERIAL AND METHODS
A prospective cohort study was performed using incident cases of kidney transplants performed on patients at a single centre between June 2003 and December 2007. All transplants involved adult recipients and donors, and none of the organs came from a living donor. The study excluded transplants performed with a CIT >15 hours. All of the recipients underwent follow-up for a minimum of one year after the transplant or until loss of the organ or death; data collection was completed in December 2008.
We used the following immunosuppressant protocol: until June 2005, triple immunosuppressant therapy with corticosteroids in decreasing doses, mycophenolate mofetil (MFM) and tacrolimus (TAC) with target levels between 10 and 15ng/ml during the first month. In June 2005, we added induction therapy with basiliximab in patients with low immunologic risk (PRA < 50%) and timoglobulin in hyperimmune patients (PRA > 50%). After induction therapy, target levels of TAC were lowered to 5-10ng/ml during the first post-transplant month. When digestive intolerance appeared in response to MMF, the drug was replaced with enteric-coated mycophenolic acid (EC-MPA). Immunosuppressants were not adjusted according to the donor type (ECD or SCD). In cases of suspected acute rejection, a kidney biopsy was performed and empirical treatment begun with 6-methylprednisolone bolus; where the diagnosis was not confirmed, the corticosteroids were discontinued. If the rejection was corticosteroid-resistant, it was treated with timoglobulin. Humoral rejection was treated with plasmapheresis and immunoglobulins. The biopsies were classified according to Banff-97 criteria.
An ECD is defined as any donor older than 60 years or between 50 and 59 years with at least two of the following conditions: a history of hypertension, death due to stroke and creatinine above 1.5mg/dl prior to organ removal.1 According to this definition, the recipients are classified in two groups according to the type of donor providing the organ: those receiving an organ from a standard criteria donor and those with an expanded criteria donor.
The variables in the study were collected prospectively, and data from both donors and recipients were collected in addition to data on transplant evolution.
The following donor information was collected: age, sex, weight, history of hypertension, cause of death, creatinine level prior to kidney removal and estimated glomerular filtration rate (eGFR) calculated with the Cockcroft-Gault formula.11 Additionally, the transplant prognosis was rated according to the Nyberg score.12
For recipients, the following information was collected: age, sex, the cause of the chronic kidney disease, body mass index, type of dialysis and its duration, number of transplant being received, number of incompatibilities between donor and recipient, the PRA score at the time of transplant and the maximum peak in historical serum levels, considering patients with PRA > 50% to be hyperimmune. CIT was counted from clamping time in the donor up to unclamping time in the recipient.
The initial evolution of the transplants included the record of surgical complications: Arterial or venous thrombosis, haemorrhage requiring secondary surgery, urological fistulae, stenosis of the ureter leading to deterioration of renal function, and lymphoceles that produced secondary complications due to their size or location. Patients were classified in four groups according to evolution of renal function over the immediate post-op period: no primary function (NPF), delayed graft function (DGF), delayed graft function without dialysis (DGF-WOD) and immediate renal function (IRF). The NPF group included those with a lack of function at any time, due to any cause. The DGF group included all patients who underwent dialysis during the first week following the transplant. The DGF-WOD and IRF groups were defined according to the creatinine reduction rate.13,14 Where the rate exceeded 30%, the patient was included in the IRF group, and where it was less, in the DGF-WOD group. In addition, we recorded the number of days required in order for the creatinine to drop below 3mg/dl, and the creatinine level and eGFR calculated by the MDRD-415 formula on the sixth day following the transplant.
Upon discharge, we recorded the number of days admitted, the proteinuria, creatinine, eGFR calculated by the MDRD-4 formula and the tacrolimus levels.
During follow-up, analytical data for all patients was recorded prospectively at 3, 6 and 12 months, and annually thereafter. The organ function stability was calculated for the first year (eGFR at 12 months - eGFR at 6 months) as well as any acute rejection episodes. All cases of kidney loss and exitus were also recorded throughout the follow-up.
Statistical analysis
We initially performed a descriptive analysis of the study variables by comparing the normality of the quantitative variable distributions using the Kolmogorov-Smirnov test. Following this, we carried out a comparative bivariate analysis using parametric tests (Student-t test and Chisquared with Fisher¿s exact probability test) or nonparametric tests (Mann-Whitney U test) as applicable. In addition, we used the log-rank test to compare survival curves. Statistical significance was established for p values < 0.05. Statistical analysis was carried out using SPSS software, version 15.0 (SPSS Inc., Chicago IL, USA).
RESULTS
Of the 87 transplants performed during the study period, 13 were excluded for having a CIT of more than 15 hours (only three with SCDs): eight organs were sent from other centers (CIT 21.3 ± 3.2 hours) and five were from our center (CIT 16.8 ± 1.2 hours). Of the 74 remaining patients (mean CIT 9.1 ± 3.6 hours), 50 received SCD organs and 24 received ECD organs.
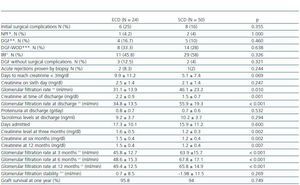
Demographic data for ECDs and SCDs, as well as the characteristics of the recipients of either type of organ are shown in table 1. Compared with SCDs, ECDs were significantly older, more frequently hypertensive, and a larger percentage died from a cerebrovascular event. Although creatinine levels in ECDs were lower than in SCDs, their eGFR calculated by the Cockcroft-Gault formula was significantly less.
All of the ECD organ transplants had a Nyberg score12 of more than 20 points. 87.5% of these transplants belonged to group C (between 20 and 29 points) and 12.5% belonged to group D (more than 30 points). All SCD organ transplants scored below 20 points. Of these, 58% belonged to group B (between 10 and 19 points) and 42% belonged to group A (between 0 and 9 points).
We did not find any significant differences between the recipients of the ECD organs and recipients of the SCD organs. We should point out that although ECD organ recipients were older, the difference was not statistically significant (p = 0.052).
The evolution of both recipient groups is described in table 2. We did not find any significant differences in the percentage of surgical complications. Neither were there any differences in the percentage of patients classified as NPF, DGF, DGF-WOD or IRF for the two groups.
In the first month following the transplant, four patients in the ECD group were treated for suspected acute rejection (15.4%), which was proven by a renal biopsy in two patients (8.3%): one was a case of acute humoral rejection in a hyperimmune patient and the other, a case of cellular rejection (Banff-97 classification IIA); both reverted with treatment. In the SCD group, six patients (12%) were initially treated with corticosteroid boli due to suspected acute rejection; the biopsy ruled out the suspected diagnosis in five cases, and the other patient presented a borderline rejection.
The hospital stay durations were similar in both groups, and during this time renal function in recipients of SCD organs was significantly better than that of ECD organ recipients.
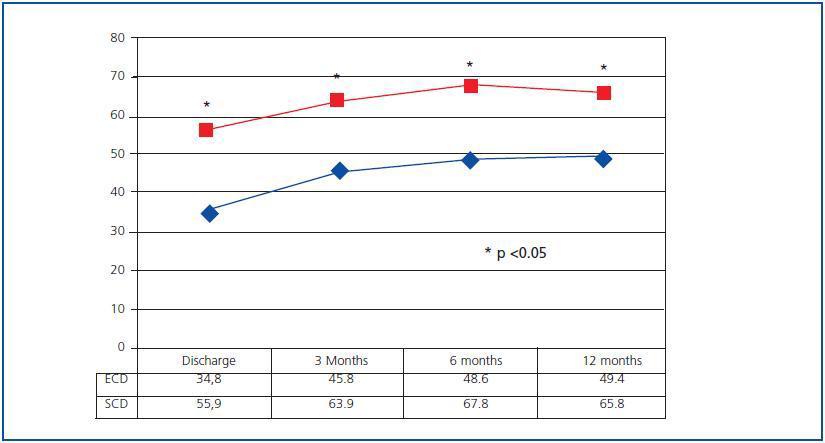
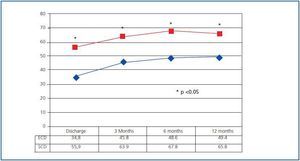
Evolution of renal function was significantly better in SCD recipients. Since the discharge date, eGFR calculated by MDRD-4 has been significantly better in SCD organ recipients than in ECD organ recipients, and this difference has been present throughout the entire follow-up (figure 1). However, organ function stability throughout the first year has been similar for both groups, and has been above zero even among ECD organ receptors (which indicates an improvement in eGFR between the 6 and 12 month marks), while in SCD organ recipients, it was below zero (suggesting a certain decrease in eGFR).
In the SCD group, two patients presented NPF in the organ due to vascular problems during surgery, and another lost the organ three months after the transplant due to the recurrence of a glomerular disease. No other transplant was lost and none of the patients died; survival rates at 12, 24 and 36 months were 100% for patients and 94% for organs. In the ECD group, one patient presented NPF, which was attributed to problems during organ preservation; no other graft was lost and none of the patients died, so survival rates at 12, 24 and 36 months were 100% for patients and 95.8% for grafts. Therefore, there were no differences for renal survival at 12, 24 and 36 months (log-rank, p = 0.749) between the recipients of ECD and SCD organs. There were also no differences in patient survival between the two groups during the same period of time.
Discussion
Our findings indicate that it is possible to obtain excellent results with ECD organs when using short CITs, and that these results can be similar to those for SCD organs, also with short CITs. However, in our experience, it is not possible to use ECD grafts and achieve a renal function comparable to that provided by SCD organs. The fact that both patient groups have similar renal survival rates could be related to the renal function stability that we reached between 6 and 12 months, considering that this parameter has been related to the survival of organs from older donors.16
Prolonged cold ischaemia favours a delay in graft function,17,18 and this delay is a risk factor for the survival of renal transplants.19 Furthermore, ischaemic damage is a determining factor for the appearance of acute rejection.20 Experimental models for kidney transplants have shown that prolonged ischaemia and advanced donor age are closely related to organ malfunction.21 Due to these reasons, the tendency in the last few years has been to shorten CIT times for ECD organs in order to improve transplant results. Europe¿s Eurotransplant Senior Programme, which applies the concept of ¿old for old¿, has reduced CIT and obtained excellent results (mean CITs of 10.6 ± 3.9 hours).22 CITs have also been reduced in the USA by applying the UNOS criteria for ECD organs.23 In this study, our CIT for ECD organs was 9.3 ± 2.5 hours. With this CIT, we achieved an NPF rate of 4.2%, a DGF rate of 16.7%, a 25% rate of initial surgical complications and an 8.3% rate of acute rejection in the first month. These results are similar to those obtained by the Eurotransplant Senior Programme22 with similar CITs. These results are not as good as those obtained with SCD organs and similar CITs (8.3 ± 3.3 hours), but they are not significantly lower: Compare with an NPF rate of 4%, a DGF rate of 10%, a 16% rate of surgical complications and a 2% rate of acute rejection proven by biopsy (although this was actually a single case of borderline rejection).
The renal function provided by the ECD organs is acceptable, although significantly less than that provided by SCD organs under the same conditions. This difference is apparent from the first week after the transplant and is still present after one year. We must take into account that the SCD group¿s evolution is very good, with a 2% acute rejection rate and only a 4% DGF rate if we exclude patients whose DGF was due to surgical complications. We believe this is what has resulted in creatinine levels of 1.22 ± 0.4mg/dl and an eGFR (MDRD-4) of 65.8 ± 14.9ml/min one year after the transplant. We calculated glomerular filtration rate with the MDRD-4 formula, which is the most precise estimate according to some studies.24,25 However, if we use the Cockcroft-Gault formula, at one year after transplant the SCD group has an eGFR of 76.7 ± 17.6ml/min, which is higher than that obtained in other studies over the same period of time and using similar immunosuppressant methods.26
One year after transplant, ECD organs present creatinine levels of 1.5 ± 0.4mg/dl and an eGFR calculated by MDRD-4 of 49.4 ± 12.5ml/min (61.4 ± 17.6ml/min by Cockcroft- Gault). This renal function is better than that obtained in other studies of ECD organs.6,16,22,27 Better renal function in ECD organ receptors has been described at one year of evolution when the CIT is less than 12 hours, compared with CITs ranging from 12 to 24 hours. However, this improvement only becomes noticeable when the ECD belong to group C on the Nyberg scale (scoring between 20 and 29 points). Our ECD organ recipients mostly belong to group C (87.5%), which probably explains their favourable evolution with short CITs. The concept of ECD implies a binary distinction between donor classes: either ECD or SCD. So, the same definition includes patients with an ample spectrum of risks having to do with losing the graft and a risk level between 1.7 and 2.691 of that of the ideal donor. This enormous difference must be reflected in the different evolutions and responses to the conditions that we impose in a transplant situation, and may generate large disparities in study results according to the type of ECD organ that is included.28 As a result, it seems necessary to develop more precise classification systems, as some have suggested.29,30
Renal survival in the ECD organ group was good: 95.8%. These good results may have been predictable, since it has been shown that in a rejection-free population, the age of the donor and the DGF rate are determining factors for graft survival;31 in our ECD group, the incidence rates for acute rejection and DGF were relatively low. In addition, we have attained a good level of renal function stability between the 6 and 12-month marks, even showing some eGFR improvement, which could be very important to these kidneys¿ long-term survival.16 Furthermore, we should not forget that one of the best parameters for predicting graft survival is the creatinine level one year after the transplant,32 and our ECD organ recipients had creatinine levels of 1.5 ± 0.4mg/dl at the one-year mark.
This study has certain limitations. It is a single-centre cohort study, with a limited number of patients and it still has a short evolution time, which is why we did not apply regression methods. We have not been able to make any comparisons with a significant number of transplant patients with an organ CIT > 15 hours, since we have very few patients of this description. However, we feel that our results indicate that efforts should be made to reduce CITS in order to improve transplant results, and that these efforts should be maximised in the case of ECD organs. In this way, we will be able to optimise results from this graft type and offer recipients the best possible scenario for both initial evolution and for graft survival and renal function.
In conclusion, the use of short cold ischaemia times enable us to transplant kidneys from expanded criteria donors with a low incidence rate of delayed graft function and acute rejection, which also results in good survival rates and a good medium-term renal function. It also enables us to obtain a very stable renal function, which may be a determining factor for excellent longterm survival.
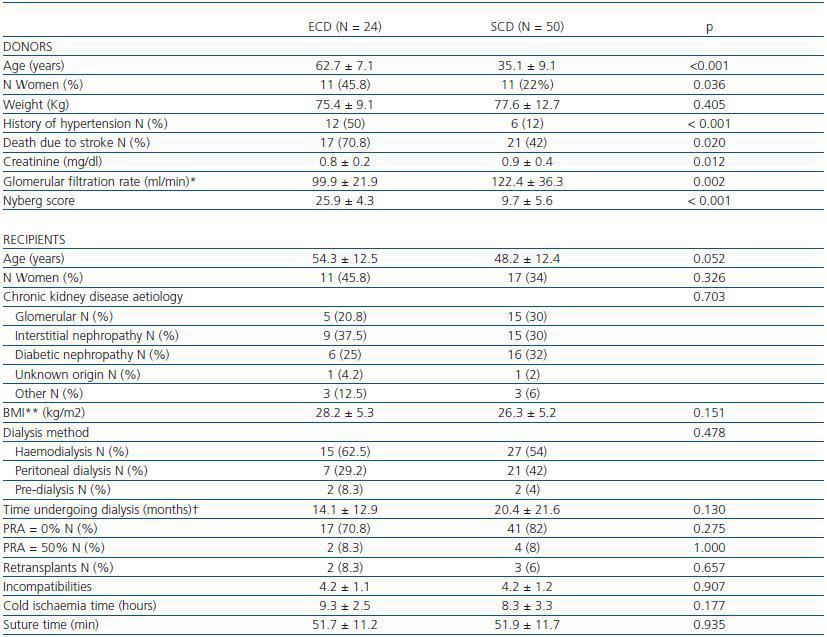
Table 1. Characteristics of expanded criteria donors (ECD) and standard criteria donors (SCD). Characteristics of recipients.
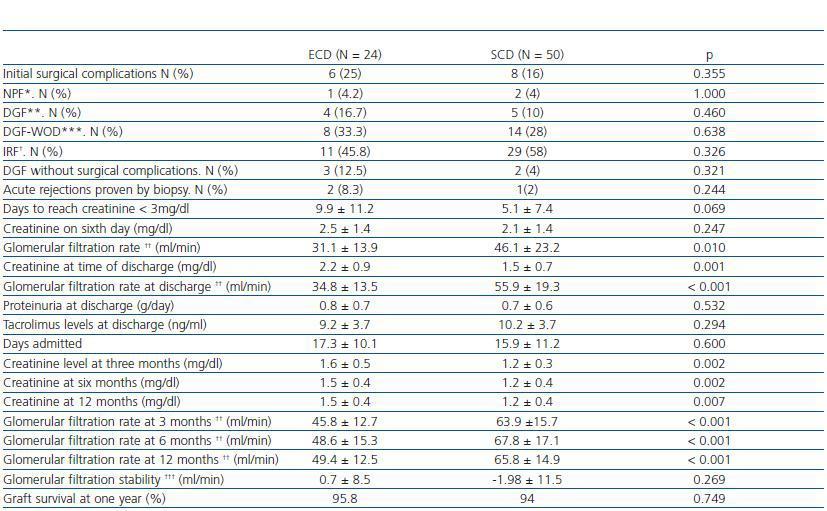
Table 2. Evolution of ECD and SCT transplant recipients
Figure 1.