Acute kidney injury (AKI) represents an independent, prognostic risk factor for chronic kidney disease development and progression, cardiovascular morbidity and mortality and all-cause mortality both in the in-hospital and in the outpatient setting1–4. Patients with diabetes mellitus experience an increased risk for AKI compared to non-diabetic subjects, while lower estimated glomerular filtration rate and higher albuminuria are associated with greater risk of AKI5.
In a recently published retrospective cohort study it was demonstrated that sodium-glucose co-transporter-2 (SGLT-2) inhibitors do not correlate with increased risk of AKI compared to other glucose-lowering drugs6. Two previous meta-analyses of the initial “hallmark” cardiovascular outcome trials, namely the Empagliflozin Cardiovascular Outcome Event Trial in Type 2 Diabetes Mellitus Patients-Removing Excess Glucose (EMPA-REG OUTCOME), the Canagliflozin Cardiovascular Assessment Study (CANVAS) and the Dapagliflozin Effect on Cardiovascular Events-Thrombolysis in Myocardial Infarction 58 (DECLARE-TIMI 58) trial, with the addition of the Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation (CREDENCE) trial, have shown that SGLT-2 inhibitors confer a robust decrease in the risk of AKI, overcoming initial worries regarding increased risk7,8.
During last year, publication of the Dapagliflozin and Prevention of Adverse Outcomes in Heart Failure (DAPA-HF) demonstrated for the first time the substantial cardiovascular benefits with SGLT-2 inhibitors in patients with heart failure with reduced ejection fraction (HFrEF) regardless of concomitant diabetes mellitus, establishing a new indication for the administration of this drug class. Recently, we were delighted to read the results of the ertugliflozin cardiovascular outcome trial (VERTIS CV) and the trial assessing the cardiovascular and renal effects of empagliflozin on patients with HFrEF with or without diabetes (EMPEROR-Reduced). In addition, we have also welcomed the results of the trial evaluating the effects of dapagliflozin on patients with chronic kidney disease and albuminuria, with or without diabetes (DAPA-CKD), along with the sotagliflozin trials, namely the Sotagliflozin on Cardiovascular and Renal Events in Patients With Type 2 Diabetes and Moderate Renal Impairment Who Are at Cardiovascular Risk (SCORED) trial and the Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure (SOLOIST-WHF) trial. Therefore, we sought to determine the effect of SGLT-2 inhibitors on AKI regardless of diabetes status across the hallmark cardiovascular and renal outcome trials.
Two independent reviewers (D.P. and C.P.) extracted the data from the eligible reports, by using a pilot tested, data extraction form. We preferred utilizing data from intention-to-treat analyses.
As we assessed only a dichotomous variable, difference was calculated with the use of risk ratio (RR), with 95% confidence interval (CI), after implementation of the Mantel–Haenszel (M–H) random effects formula. Statistical heterogeneity among studies was assessed by using I2 statistics. Heterogeneity was considered to be low if I2 was between 0% and 25%, moderate if I2 was between 25% and 50%, or high if I2 was greater than 75%9. All analyses were performed at the 0.05 significance level, while they were undertaken with RevMan 5.3 software.
Two independent reviewers (D.P. and C.P.) assessed the quality of the included randomized controlled trials, by using the Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) for the primary safety outcome10. Discrepancies between reviewers were solved by discussion, consensus or arbitration by a third senior reviewer (M.D.).
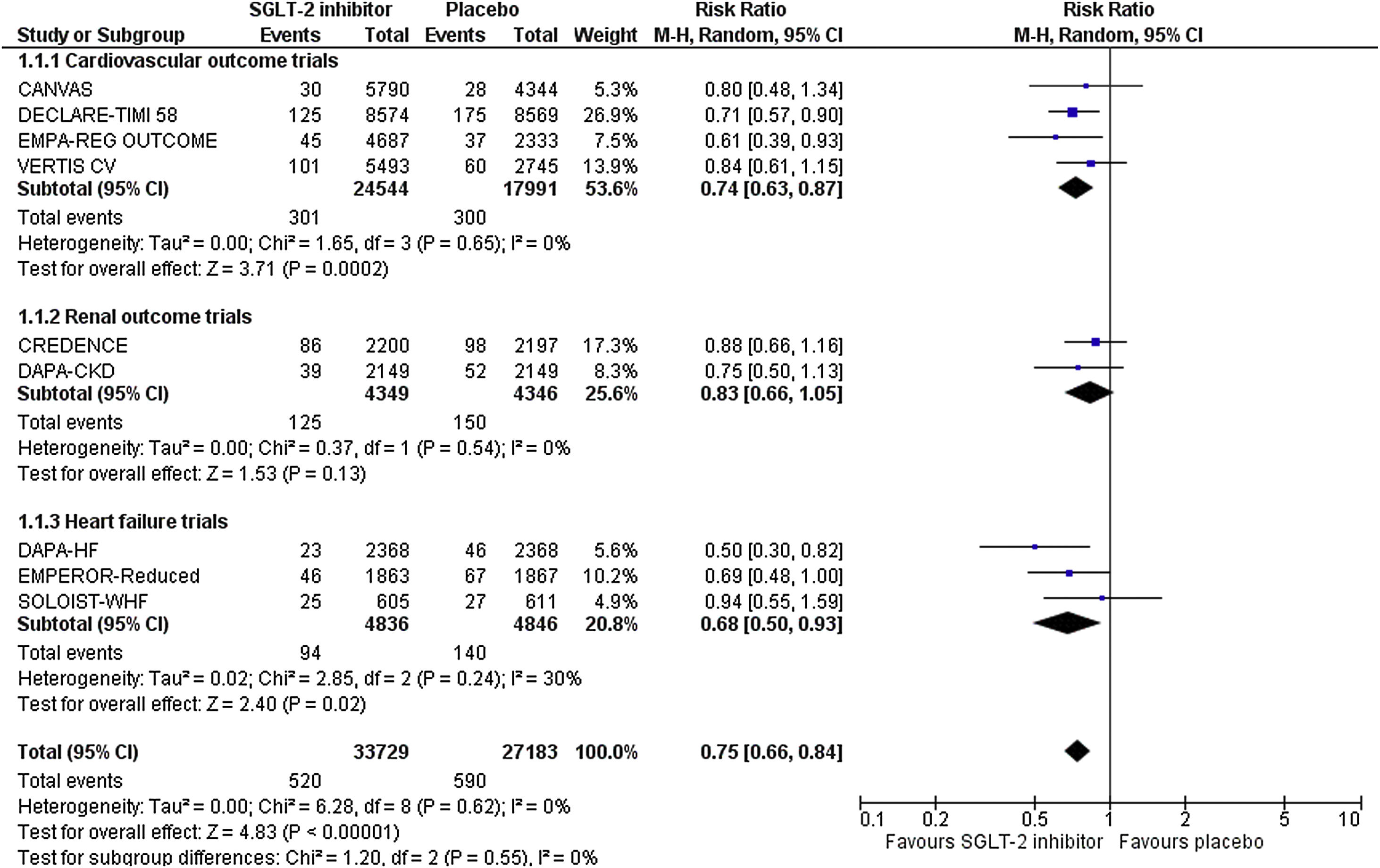
Besides trialists of the SCORED trial who did not report any data regarding the incidence of AKI in the two treatment arms, the rest cardiovascular and renal outcome trials and trial performed in the heart failure population provided corresponding data. Specifically, trialists of the EMPEROR-Reduced trial provided relevant data in a recently published, post-hoc analysis. Therefore, we pooled available data from nine randomized controlled trials in a total of 60,912 enrolled participants. Risk of bias is considered as low across all included trials.
Overall, SGLT-2 inhibitor treatment resulted in a decrease in the risk of AKI equal to 25% (RR=0.75, 95% CI; 0.66–0.84, I2=0%), as shown in Fig. 1. This effect was driven by the cardiovascular outcome trials (RR=0.74, 95% CI; 0.63–0.87, I2=0%) and trials performed in the heart failure population (RR=0.68, 95% CI; 0.50–0.93, I2=30%). In contrast, no effect was observed in the dedicated renal outcome trials (RR=0.83, 95% CI; 0.66–1.05, I2=0%).
Collectively, it seems that the observed protective effect of SGLT-2 inhibitors against AKI in the high or very high-risk patients enrolled in the aforementioned cardiovascular and renal outcome trials and trials in the heart failure population is very important and should influence decision making in clinical practice. Of note, close monitoring of patients after prescription of a SGLT-2 inhibitor is always required, especially in patients with risk factors for presenting with AKI, since manifestation of adverse events such as volume depletion phenomena or hypotension could potentially predispose to the development of AKI in the context of the induced osmotic diuresis, even though incidence of these events remains quite low.
Conflict of interestNone to declare.