Hemodialysis patients have an enhanced risk for cardiovascular events. Cardiac biomarkers provide useful information for stratifying their risk. However the prognosis value of creatine kinase MB isoenzyme (CKMB) has not yet been validated in this population. The aim of the present study is to determine the predictable value of CK-MB in hemodialysis.
MethodsA cohort of 211 hemodialysis patients (58.3% male, median age 73 (60–80) years) were followed for 39 (19–56) months. Cardiac biomarkers including CKMB were recorded at baseline. Factors associated to CKMB and prognosis value of this biomarker was studied.
ResultsThe median value of CKMB was 1 (1–2) ng/mL with no patient exceeding normal laboratory values. Previous heart disease, diabetes mellitus, peripheral vascular disease and systolic and diastolic dysfunction were associated with higher levels of CKMB. Ninety-four patients (44.5%) cardiovascular events were recorded. CKMB levels ≥2ng/mL was independently associated to cardiovascular events during the follow up after adjusting. Adding CKMB to a model including several variables for predicting cardiovascular events, resulted in 17% improvement in risk discrimination (IDI) with a relative IDI of 9.9% (p=0.04).
ConclusionsCKMB is a good marker for stratifying cardiovascular risk in hemodialysis patients and adds prognosis information to other well known independent predictors for cardiovascular events.
Los pacientes en hemodiálisis presentan un riesgo cardiovascular elevado. Los biomarcadores cardiacos otorgan información útil para estratificar dicho riesgo cardiovascular. Sin embargo, el valor pronóstico de la isoenzima MB de la creatincinasa (CKMB) no ha sido aún validado en esta población. El objetivo del presente trabajo es evaluar el valor predictivo de CKMB en una población en hemodiálisis.
MétodosUna cohorte de 211 pacientes en hemodiálisis (58,3% varones, con una edad media de 73 [60–80] años) fueron seguidos durante 39 (19–56) meses. Se recogieron basalmente los valores de diferentes biomarcadores cardiacos incluyendo CKMB. Se evaluaron los factores asociados a niveles más elevados de CKMB, así como su valor predictivo independiente.
ResultadosLa mediana de CKMB fue de 1 (1–2) ng/mL. Todos los pacientes presentaron valores dentro de los establecidos de referencia en la población normal. Los antecedentes de cardiopatía, diabetes mellitus, enfermedad periférica y la disfunción diastólica y sistólica se asociaron a niveles más elevados de CKMB. Un total de 94 pacientes (44,5%) presentaron un evento cardiovascular. Los niveles de CKMB≥2ng/mL se asociaron de manera independiente a presentar eventos cardiovasculares durante el seguimiento tras el ajuste para diferentes factores. La adición de CKMB a un modelo predictor con diferentes factores generó una mejoría del 17% en la estimación de la probabilidad de forma lineal (IDI) con un IDI relativo del 9,9% (p=0,04).
ConclusionesCKMB es un buen marcador para estratificar el riesgo cardiovascular en los pacientes de hemodiálisis y añade información en cuanto al pronóstico cuando se combina con otros predictores de eventos cardiovasculares.
Chronic kidney disease (CKD) is a major cardiovascular risk factor similar to congestive heart failure, and this risk increases with the decline of renal function, being maximum in dialysis.1,2
Several strategies have been proposed in order to detect those patients at high risk for developing cardiovascular events and for detecting subclinical alterations to be treated.3 Current guidelines recommend measuring cardiac biomarkers, specifically troponins and natriuretic peptides, as they are usually increased in our patients.1,4 If possible, ecocardiography should be performed periodically although recommended intervals vary in function of the guideline. Some studies have found a strong association between cardiac biomarkers, ecocardiographic findings and prognosis.3,5
On one hand, serum cardiac biomarkers are increased in virtually all CKD patients especially in dialysis and those who have higher values have poorer prognosis. On the other hand, when these biomarkers are adjusted by ecocardiographic findings (for example, diastolic and systolic dysfunction and left ventricular hypertrophy) they lose their independent prediction value, suggesting their role as observer of cardiac damage, usually subclinically.5,6 Most importantly, increased cardiac biomarkers seems to be universal with high sensitivity assays, but those with higher values have a better association to postmortem cardiac damage or with coronary lesions demonstrated by angiography.7,8
However, and although several authors have proposed different cut-offs for stratifying the cardiovascular risk, lack of agreement has been reached, suggesting that probably those markers must be interpreted as continuous variables.
One important biomarker has not been widely studied in CKD patients until date, creatine kinase MB isoenzyme (CKMB). In general population, its sensitivity in acute coronary syndromes is inferior to troponins. However, due to its small half-life, current guidelines recommend its use for monitoring cardiac damage after revascularization as they present a good correlation with re-infarction.9,10 Many patients with CKD have been excluded from studies about CKMB due to its difficult interpretation when renal function is impaired. Published series yield controversial data in terms of prevalence of raise serum values and their use in ischemic heart disease. However, it seems to have the same value as prognosis marker in re-infarction in patients with renal impairment.11,12 The aim of the present study was to analyze the prognosis value of CKMB in a cohort of dialysis patients and also its related factors.
Materials and methodsPatientsA total of 211 patients on hemodialysis in a single center were enrolled in the restrospective study. Stable patients with no cardiovascular events in the 4 weeks before serum determinations were included. During the follow up [39 (19–56) months], patients with changes in hemodialysis parameters, transferred to another center or transplanted were censored. Investigations were in accordance with the Declaration of Helsinki.
Baseline characteristics and measurementsBaseline characteristics were recorded, including age, sex, presence of diabetes mellitus, peripheral vascular disease, previous cardiovascular disease (congestive heart failure determined by echocardiography within the three previous months, myocardial infarction, cerebrovascular disease), dialysis vintage and data regarding the vascular access. Basally, we measured C-reactive protein (CRP), high sensitivity troponin T (hsTnT), CK-MB and N-terminal prohormone of brain natriuretic peptide (Nt-proBNP). All included patients had the same hemodialysis therapy protocol: 4h, three times per week. Routine clinical and biochemical variables were measured by standardized methods on autoanalyzers. CKMB and HsTnT were measured on a Roche/Hitachi Cobas E411 analyzer. Factors related to higher values of CKMB were analyzed.
EchocardiographyEchocardiography was recorded in stable patients who had a less than 6-month one before obtaining the sample and within 24h after the last hemodialysis on the day between mid-week dialysis sessions. Diastolic dysfunction was defined as e′ (early mitral annulus velocity) less than 8cm/s, average E (early mitral flow)/e′ over 8, LAVi (left atrium volume index) over 28mL/m2 or Ar−A (time difference between duration of the pulmonary venous atrial reversal wave and duration of the A wave) over 30ms. Systolic dysfunction in turn was defined as a left ventricle ejection fraction of under 45%. The left ventricular mass index (LVMi) was estimated by Devereux's formula.13
OutcomesNew cardiovascular events (ischemic or hemorrhagic cerebrovascular accident, cardiac event [including myocardial infarction and/or congestive heart failure], peripheral vascular events and other ischemic events) were recorded during follow-up. We analyzed the predictor value of CKMB for cardiovascular events.
Statistical proceduresValues are expressed as the mean (SD) or median (IQR). We established linear regression models for evaluating the distribution of the studied variables according to CKMB levels. To assess the diagnosis value of the different cardiac markers we used the receiving operator characteristics (ROC) curve for each one. Correlations between cardiac markers were performed using Pearson test. Multivariate analysis was performed by Cox regression. Variables were analyzed and only those considered confounders were entered in the final Cox regression model. Different models were used, including CKMB for its different values, in order to determine the best cut-off to assess cardiovascular risk. Cardiovascular events were analyzed using Kaplan–Meier plots, and survival curves were compared using the log-rank test. We used the integrated discrimination improvement (IDI), as described by Pencina et al.14 to interpret the incremental value of CKMB≥2ng/mL added to a risk prediction model including age, sex, previous heart disease, peripheral vascular disease, diabetes mellitus, NtproBNP and HsTnT. IDI is a measure of improvement in model performance and represents the difference between discrimination slopes of two competing models. We calculated the relative IDI, which expresses the relative increase in separation of events and non-events from the separation achieved in the base model (i.e., the difference in discrimination slopes is expressed as a proportion of the discrimination slope of the base model).15 All statistical analyses were performed with the SPSS® 18.0 statistical package (Chicago, IL, USA). A p-value <0.05 was considered statistically significant.
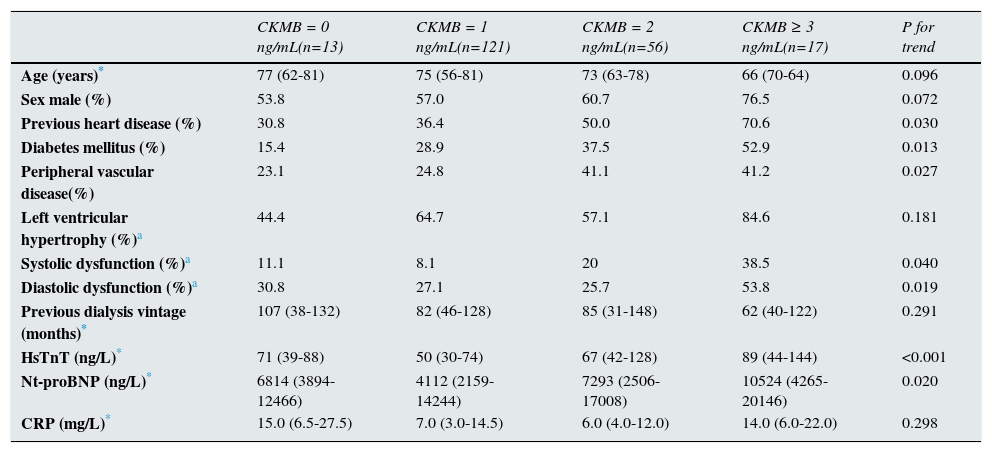
ResultsBaseline characteristics and factors associated with increased levelsA total of 211 prevalent hemodialysis patients were included in this study to be followed for 39 (19–56) months. One hundred and twenty-three patients (58.3%) were male, with a median age of 73 (60–80). Baseline characteristic are showed in Table 1. The median value of CKMB was 1 (1–2) ng/mL, with the following distribution: 13 patients (6.3%) have basal levels of 0ng/mL, 121 (58.5) have 1ng/mL, 56 (27.1%) have 2ng/mL, 13 (6.3%) have 3ng/dL and only 4 (1.9) have 4ng/dL. In Table 2, baseline characteristics are shown in the different strata according to CKMB values. No patients exceeded the normal range of our laboratory (0–4ng/mL). Univariate analysis revealed that higher levels of CKMB were associated to previous heart disease, diabetes mellitus, peripheral vascular disease and systolic and diastolic dysfunction (data shown in Table 3).
Baseline characteristics.
| Age (years) | 73 (60-80)* |
| Sex male,n(%) | 123 (58.3)a |
| History of heart disease,n(%) | 90 (42.7)a |
| Diabetes mellitus,n(%) | 68 (32.2)a |
| Peripheral vascular disease,n(%) | 64 (30.3)a |
| Previous echocardiogram,n(%) | 166 (78.7)a |
| - Left ventricular hypertrophy,n(%) | 106 (63.9)a |
| - Systolic dysfunction,n(%) | 24 (14.4)a |
| - Diastolic dysfunction,n(%) | 60 (36.1)a |
| Previous dialysis vintage (months) | 83 (43-128)* |
| Vascular access- Autologousn(%)- PTFEn(%)- Permanent cathetern(%) | 116 (55)a65 (31)a30 (14)a |
| HsTnT (ng/L) | 56 (35-90)* |
| CKMB (ng/mL)- 0 ng/mL,n(%)- 1 ng/mL,n(%)- 2 ng/mL,n(%)- 3 ng/mL,n(%)- 4 ng/mL,n(%) | 1 (1-2)*13 (6.3)a121 (58.5)a56 (27.1)a13 (6.3)a4 (1.9)a |
| Nt-proBNP (ng/L) | 4994 (2237-15036)* |
| CRP (mg/L) | 7.0 (4.0-15.0)* |
Descriptive of baseline characteristics according to CKMB values.
| CKMB = 0 ng/mL(n=13) | CKMB = 1 ng/mL(n=121) | CKMB = 2 ng/mL(n=56) | CKMB ≥ 3 ng/mL(n=17) | P for trend | |
|---|---|---|---|---|---|
| Age (years)* | 77 (62-81) | 75 (56-81) | 73 (63-78) | 66 (70-64) | 0.096 |
| Sex male (%) | 53.8 | 57.0 | 60.7 | 76.5 | 0.072 |
| Previous heart disease (%) | 30.8 | 36.4 | 50.0 | 70.6 | 0.030 |
| Diabetes mellitus (%) | 15.4 | 28.9 | 37.5 | 52.9 | 0.013 |
| Peripheral vascular disease(%) | 23.1 | 24.8 | 41.1 | 41.2 | 0.027 |
| Left ventricular hypertrophy (%)a | 44.4 | 64.7 | 57.1 | 84.6 | 0.181 |
| Systolic dysfunction (%)a | 11.1 | 8.1 | 20 | 38.5 | 0.040 |
| Diastolic dysfunction (%)a | 30.8 | 27.1 | 25.7 | 53.8 | 0.019 |
| Previous dialysis vintage (months)* | 107 (38-132) | 82 (46-128) | 85 (31-148) | 62 (40-122) | 0.291 |
| HsTnT (ng/L)* | 71 (39-88) | 50 (30-74) | 67 (42-128) | 89 (44-144) | <0.001 |
| Nt-proBNP (ng/L)* | 6814 (3894-12466) | 4112 (2159-14244) | 7293 (2506-17008) | 10524 (4265-20146) | 0.020 |
| CRP (mg/L)* | 15.0 (6.5-27.5) | 7.0 (3.0-14.5) | 6.0 (4.0-12.0) | 14.0 (6.0-22.0) | 0.298 |
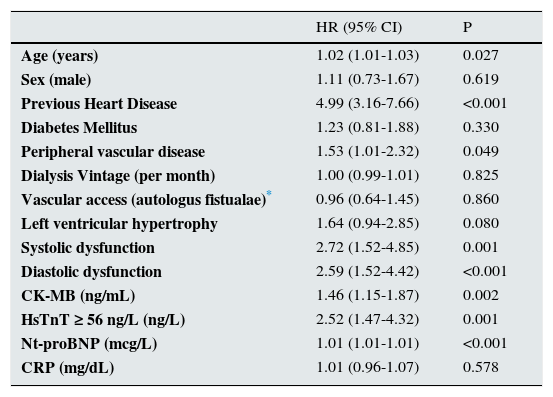
Factors associated with cardiovascular events during follow up (unadjusted Cox regression).
| HR (95% CI) | P | |
|---|---|---|
| Age (years) | 1.02 (1.01-1.03) | 0.027 |
| Sex (male) | 1.11 (0.73-1.67) | 0.619 |
| Previous Heart Disease | 4.99 (3.16-7.66) | <0.001 |
| Diabetes Mellitus | 1.23 (0.81-1.88) | 0.330 |
| Peripheral vascular disease | 1.53 (1.01-2.32) | 0.049 |
| Dialysis Vintage (per month) | 1.00 (0.99-1.01) | 0.825 |
| Vascular access (autologus fistualae)* | 0.96 (0.64-1.45) | 0.860 |
| Left ventricular hypertrophy | 1.64 (0.94-2.85) | 0.080 |
| Systolic dysfunction | 2.72 (1.52-4.85) | 0.001 |
| Diastolic dysfunction | 2.59 (1.52-4.42) | <0.001 |
| CK-MB (ng/mL) | 1.46 (1.15-1.87) | 0.002 |
| HsTnT ≥ 56 ng/L (ng/L) | 2.52 (1.47-4.32) | 0.001 |
| Nt-proBNP (mcg/L) | 1.01 (1.01-1.01) | <0.001 |
| CRP (mg/dL) | 1.01 (0.96-1.07) | 0.578 |
Abbreviations: HR (95% CI) = Hazard ratio (95% Confidence interval). (F/M)=(female/male). Creatinekinase-MB(CK-MB), N-terminal prohormone of brain natriureticpeptide (NT-proBNP),High sentivity troponin T (hsTnT), C- reactive protein (CRP).
Correlation between cardiac biomarkers showed a positive and significant one between CKMB and NtproBNP (0.163, p=0.020) and between NtproBNP and HsTnT (0.635, p<0.001). No significant correlation was established between CKMB and HsTnT.
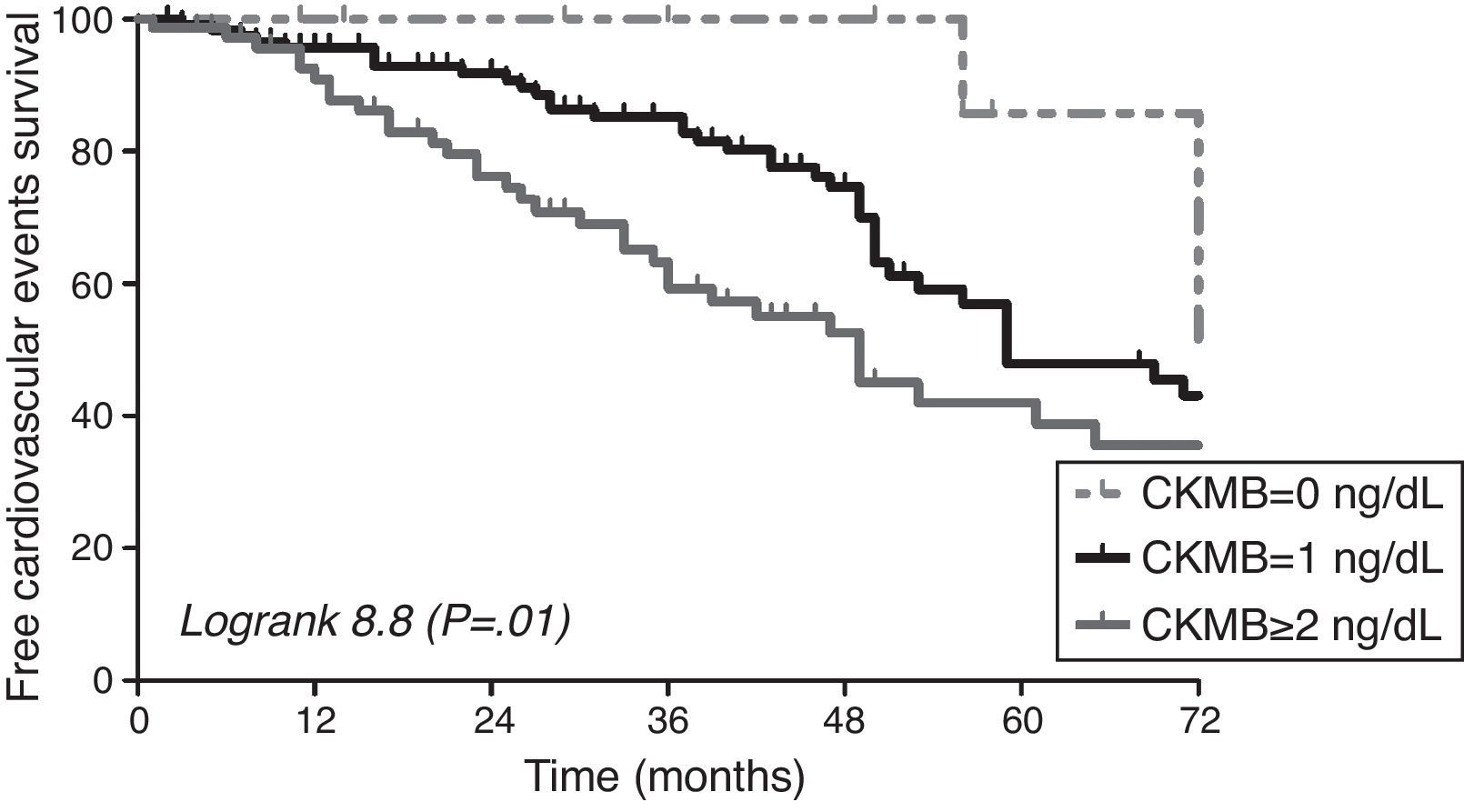
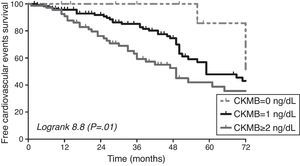
Cardiovascular events and predictive value of CKMBA total of 94 cardiovascular events were recorded during the follow up. Cardiac event was the most common event (79.8%), followed by peripheral vascular disease (8.5%), cerebrovascular event (6.4%). Diabetic patients did not show higher incidence of cardiovascular events in our cohort (p=0.3), although they have a trend of higher prevalence of diastolic dysfunction (p=0.056). The area under the ROC curve for cardiovascular events was greater for HsTnT (0.723) and Nt-proBNP (0.688) than CKMB (0.587). CKMB levels were associated to the development of cardiovascular events during follow up [HR 1.46 95% CI (1.15–1.87), p=0.002] as well as the other factors shown in Table 3. In Fig. 1, the survival curve shows the association between cardiovascular events and the different CKMB values confirming that higher levels condition worse prognosis (logRank 8.8, p=0.01). Multivariate Cox regression model adjusted for several cofounders and variables showed that CKMB levels ≥2ng/mL independently increased cardiovascular risk in our cohort of hemodialysis patients. A non-significant trend was observed if the CKMB cut-off was ≥1ng/mL (Table 4).
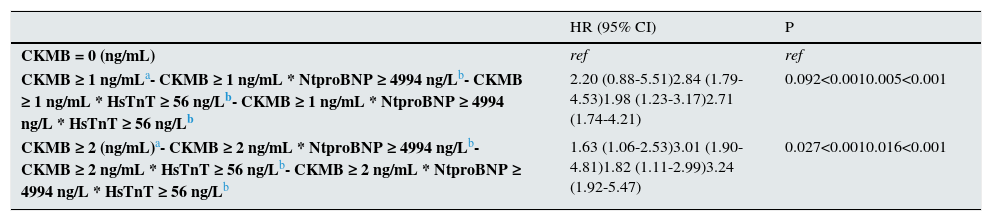
Predictor value of the different values of CK-MB for cardiovascular events during follow up (adjusted Cox regression) alone and in combination with the other cardiac biomarkers.
| HR (95% CI) | P | |
|---|---|---|
| CKMB = 0 (ng/mL) | ref | ref |
| CKMB ≥ 1 ng/mLa- CKMB ≥ 1 ng/mL * NtproBNP ≥ 4994 ng/Lb- CKMB ≥ 1 ng/mL * HsTnT ≥ 56 ng/Lb- CKMB ≥ 1 ng/mL * NtproBNP ≥ 4994 ng/L * HsTnT ≥ 56 ng/Lb | 2.20 (0.88-5.51)2.84 (1.79-4.53)1.98 (1.23-3.17)2.71 (1.74-4.21) | 0.092<0.0010.005<0.001 |
| CKMB ≥ 2 (ng/mL)a- CKMB ≥ 2 ng/mL * NtproBNP ≥ 4994 ng/Lb- CKMB ≥ 2 ng/mL * HsTnT ≥ 56 ng/Lb- CKMB ≥ 2 ng/mL * NtproBNP ≥ 4994 ng/L * HsTnT ≥ 56 ng/Lb | 1.63 (1.06-2.53)3.01 (1.90-4.81)1.82 (1.11-2.99)3.24 (1.92-5.47) | 0.027<0.0010.016<0.001 |
IDI analysis was performed to assess the improvement in risk discrimination of adding CKMB (≥2ng/mL) to a cardiovascular event risk prediction model including age, sex, previous heart disease, peripheral vascular disease, diabetes mellitus, NtproBNP and HsTnT. This analysis comprised all individuals and used NtproBNP and HsTnT as dichotomous variables. Adding CKMB resulted in 17% improved risk discrimination for cardiovascular events (IDI 0.026 [0.004–0.053]; relative IDI 9.9%; p=0.04).
DiscussionOur study demonstrates that CK-MB is a useful marker for stratifying cardiovascular risk in dialysis patients, even in normal range values. Importantly for acute situations, hemodialysis patients do not have basally elevated higher levels, so this marker could be useful in the differential diagnosis of chest pain as marker of acute ischemia. Supporting this fact, and different from other cardiac markers, CKMB does not appear to be influenced by the dialysis, so its levels remain stable after and before.16 However, and although CKMB has a short half-life, its levels are correlated to HsTnT and NtproBNP and their values are associated to other cardiovascular risk factors. One study conducted by McCullong et al. that included 817 consecutive patients with chest pain, shown that those with confirmed acute myocardial infarction (AMI) suffer a CKMB rise irrespective of the renal function.17 Curiously, in this study, those patients without final diagnosis of AMI showed significant different basal levels of CKMB between groups (inversely correlated with glomerular filtration and maximum in dialysis patients), unlike those with final diagnosis of AMI.
The first report regarding the influence of hemodialysis in CKMB levels was published in 1984 by Jaffe et al.18 In this study that included 88 patients, authors demonstrated normal levels of CKMB for the most part of the studied sample. Although some authors differ in their opinions on this regard, the CHANCE study confirmed not only this situation but also that levels of CK-MB could be influenced by the presence of history of ischemic heart disease.19–22 Our results agree with this finding, and with the association to age (we could only demonstrate a trend with this value) and troponins. Cardiovascular risk factors as peripheral vascular disease, diabetes mellitus and Nt-proBNP seem to be associated to CKMB, above all with values ≥3ng/mL.
We assessed the predictive value of CKMB for cardiovascular events. Our data suggests that CKMB≥2ng/mL give a poor cardiovascular prognosis in dialysis patients. Results of CHANCE study agree with our results, showing an increased risk of major cardiovascular events in those patients with CKMB≥3ng/mL, in a 2-year follow up.11
However, CKMB is not the only marker but the less studied in this regard, although it is cheap and easily applicable in routine clinical. In adjusted multivariate analysis, those patients with HsTnT and NtproBNP over the median and CKMB≥2ng/mL enhanced its cardiovascular risk more than threefold, in comparison to CKMB≥2ng/mL alone. Several studies have found an association between cardiac markers and prognosis, and now we know that this situation reveals a true clinical or subclinical cardiac damage.7,23 In fact, in our study, CKMB demonstrated an association with previous heart disease and also with systolic and diastolic dysfunction. Previous published data has only been able to remark an association to LVH in patients with renal function impairment.
The present study has some limitations. Firstly, the typical limitations of a retrospective design. Secondly, not all the patients had a echocardiograph image and they were performed by different specialists. This bias was partially avoided by the use of the same criteria for the definitions of each entity (systolic and diastolic dysfunction and LVH). Thirdly, vascular calcifications were not assessed as recommended in recent guidelines.24 Lastly, the study was performed in one center, and results must be confirmed in bigger sample size.
In conclusion, CKMB is a good marker for stratifying cardiovascular risk in hemodialysis patients even in the normal range of their values. We recommend measuring several cardiac markers, at least two, in order to get better predictive values. As basal levels of CKMB are not elevated in these patients, further studies are required to confirm their value in acute coronary syndromes.
Conflict of interestsThe authors declare no conflict of interest.
We would like to thank Vilma E. Pacheco for proofreading the manuscript.