The use of proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i) has changed the clinical practice of dyslipidemia and their effects in cardiovascular events. Nowadays two active principles have been commercialized (evolocumab and alircumab) with indication in patients with low-density lipoprotein (LDL) cholesterol over 100mg/dl, history of cardiovascular events and higher statins doses prescribed. Clinical trials with PCSK9i have included chronic kidney disease (CKD) patients, although glomerular filtration rate has been limited up to 20ml/min/1,73m2 or higher. The published sub-analysis of them have demonstrated at least the same efficacy (in reduction of LDL and cardiovascular events) and safety in patients with CKD. However, as these drugs are very powerful in reducing LDL cholesterol, some safety concerns appeared in the past. Once safety is probed, we conclude that PCSK9i are beneficial for CKD patients.
El uso de fármacos inhibidores de la proprotein convertase subtilisin/kexin type 9 (iPCSK9) ha supuesto un cambio en el tratamiento de la dislipemia en sus efectos sobre los eventos cardiovasculares. Actualmente disponemos de dos principios activos comercializados (evolocumab y alircumab) que tienen indicación en los pacientes que no alcanzan unos valores de lipoproteína de baja densidad (LDL) inferior a 100mg/dl habiendo presentado un evento cardiovascular previo y con dosis máximas o intolerancia a las estatinas. Los ensayos clínicos con iPCSK9 han incluido a pacientes con enfermedad renal, aunque con límites en el filtrado glomerular de hasta 20ml/min/1,73m2 en el menos restrictivo de ellos. Los subanálisis de estos ensayos clínicos han demostrado eficacia en los pacientes con insuficiencia renal tanto en la mejoría del perfil lipídico como en la reducción de eventos cardiovasculares. Derivado de su intenso efecto en la reducción de LDL colesterol han surgido algunas dudas sobre su seguridad que actualmente están resueltas y que confieren a los iPCSK9 un evidente beneficio cardiovascular también pacientes con enfermedad renal.
The leading cause of mortality in patients with chronic kidney disease (CKD) in the world is cardiovascular.1,2 Patients with renal dysfunction have a high cardiovascular risk due to a combination of classical and non-classical factors.3 Among the traditional cardiovascular risk factors, dyslipidemia has been associated (not without some controversy) with a worse cardiovascular prognosis in patients with CKD and kidney transplant patients, but not in dialysis patients (Table 1).4–9
Clinical trials of patients with chronic kidney disease.
| N | Randomization | Tracing | Results | |
|---|---|---|---|---|
| UK-HARP I | 448 (242 ERC, 73 D, 133 Tx) | Simvastatin 20mg vs. placebo | 12 months | Decrease in LDL, total cholesterol and TG |
| UK-HARP II | 203 (152 ERC, 51 D) | Simvastatin 20mg+ezetimibe vs. simvastatin 20mg | 6 months | Decrease in LDL and total cholesterol |
| DAWN | 2,773 D | Rosuvastatin 10mg vs. placebo | 3.2 years | No differences in major CVDa |
| 4D | 1,225 DM, D | Atorvastatin 20mg vs. placebo | 4 years | No differences in CVD greater than. LDL decreased |
| SHARP | 9,270 (6247 ERC, 3,023 D) | Simvastatin 20mg+ezetimibe 10mg vs. placebo | 4.9 years | Reduction of major cardiovascular events in patients with CKDb |
| ALERT | 2,102 Tx | Fluvastatin 80mg vs. placebo | 6.7 years | Reduction of major cardiovascular events in patients with CKDc |
D, dialysis; DM, diabetes mellitus; CKD, chronic kidney disease; CVD, cardiovascular events; TG, triglycerides; Tx, transplantation.
For this reason, current Nephrology guidelines practically universally recommend the use of statins or statins with ezetimibe in patients with CKD but not in those undergoing some type of renal replacement therapy (unless they have already been prescribed it previously). The recommendation is extended with less evidence to kidney transplant patients.10
The marketing of the new proprotein convertase subtilisin/kexin type 9 (iPCSK9) inhibitor drugs, added to the recent revision of the European cardiology and atherosclerosis guidelines, with specific recommendations in the renal population, has proposed a change in the paradigm of the treatment of dyslipidemia. Additionally, the ability of these agents to lower low-density lipoprotein (LDL) levels may raise safety concerns when reaching these extremely low levels.
Kidney effects of dyslipidemia: lipoid nephrotoxicityThe bilateral effects of dyslipidemia in the kidney and of renal diseases on the lipid profile have been studied mainly in nephrotic syndrome.11 In this, lipoproteins are altered both in quantity and composition. While high-density lipoprotein (HDL) cholesterol molecules are not particularly altered in nephrotic syndrome, levels of LDL cholesterol, very low-density lipoproteins, and triglycerides are elevated. The proposed mechanism includes defects in the clearance of lipoproteins (due to decreased activity of liver and tissue lipoprotein lipase) but also an increase in the PCSK9 enzyme responsible for the recycling of hepatic LDL cholesterol.11,12 Renal failure itself also generates changes in the lipid profile, making it more atherogenic. Alterations consisting of a decrease in HDL, an increase in triglycerides and oxidation of LDL particles become more evident with the deterioration of the glomerular filtration rate (GFR).13,14
On the other hand, lipids can be deposited in the kidney leading to some alterations that are included under the term lipoid nephrotoxicity.11 High LDL cholesterol levels and the oxidation of these particles are trapped by mesangial cells, promoting their proliferation and, finally, glomerular sclerosis. Free fatty acids have demonstrated their ability to induce direct damage to the podocyte, especially in the presence of albuminuria.15 Additionally, saturated fatty acids can leach out causing tubulointerstitial damage.16
All these renal toxic actions of the different particles of lipid metabolism are increased when obesity is associated with dyslipidemia due to increased oxidative stress, inflammation or overactivation of the renin-angiotensin-aldosterone system.17
Together, these alterations theoretically would have the ability to promote the development of progressive renal failure. However, published clinical studies present conflicting results. On the one hand, the association between the incidence of CKD and alterations in the lipid profile in the general population has been proven in epidemiological studies.18 On the other hand, the predictive capacity of the lipid profile to aggravate the progression of renal failure seems proven in association with obesity, but it is doubtful when analyzed independently.19,20
Recommendations of the clinical guidelinesThe clinical guidelines published to date are highly controversial regarding the management and target LDL levels in patients with renal failure (Table 2).21 Based on the KDIGO guidelines, patients with CKD should receive a statin in any situation without having to monitor LDL cholesterol levels.10 This strategy, known as “fire and forget”, is in contrast to what the recently published European guidelines on Cardiology and Atherosclerosis establish as a recommendation.22 In them, the presence of CKD means raising the cardiovascular risk of patients to high in those with an estimated GFR (eGFR) between 30 and 59ml/min/m2 and they mark an LDL target lower than 70mg/dl, already very high in eGFR less than 30ml/min/m2, placing the LDL cholesterol target at 55mg/dl. Studies prior to the publication of the latest guidelines reveal the difficulty of achieving these goals in the vast majority of patients with CKD. Up to 60% of the population with renal failure is unable to achieve an LDL lower than 70mg/dl, so it seems clear that statins may not be sufficient in the management of dyslipidemia in these patients.23,24
Recommendations of the clinical guidelines for dyslipidemia in patients with kidney disease.
| Treatment indication | Treatment objective | |
|---|---|---|
| KDIGO 2013 | Patients older than 50 years with CKD (G3a-G5) without the need for RRT Patients 18−49 years with CKD (G3a-G5) without the need for RRT and one of the following: Coronary heart disease Diabetes mellitus Stroke Estimated CVR>10% Carrier patients kidney transplant | No monitoring required (fire and forget) |
| ESC/EAS 2019 | Depending on cardiovascular risk: High: eGFR 30−55ml/min/m2 Very high: eGFR <30ml/min/m2 | LDL reduction ≥50% and: LDL<70mg/dl LDL<55mg/dl |
| SEA 2019 | Primary prevention in patients with CKD≥grade 3b Secondary prevention in CKD patients≥grade 3b | LDL<130mg/dlaLDL<70mg/dl |
CKD, chronic kidney disease; EGFR, estimated glomerular filtration rate; KDIGO, Kidney Disease, Improving Global Outcomes; LDL, low-density lipoprotein cholesterol; NLA, National Lipid Association; SEA, Spanish Society of Arteriosclerosis.
The Spanish Society of Atherosclerosis has just published the recommendations in which, in addition to the above, it includes patients with CKD grade 3b or higher in the subgroup of patients who are candidates for treatment with iPCSK9 in primary prevention if they do not reach LDL below 130mg/dl with statins.25
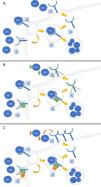
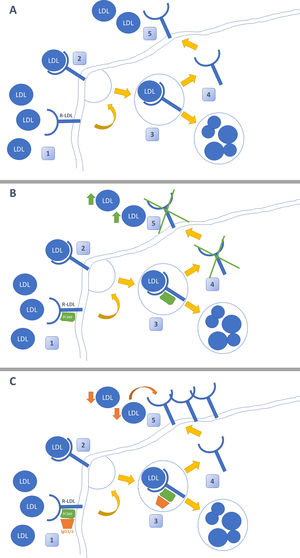
PCSK-9 inhibitors: evidence in the renal patientThe emergence of new lipid- lowering drugs has led to a revolution in the treatment of dyslipidemia. Its mechanism of action lies in blocking the enzyme protein PCSK9 by means of a monoclonal antibody, whose main function is to block the recycling of LDL receptors (LDL-R). When iPCSK9 antibodies are administered, the R-LDLs are internalized into the hepatocyte along with an LDL molecule. Once inside it, in the absence of PCSK9, they separate so that the LDL molecule is degraded and R-LDL is recycled and can return to the surface to continue capturing LDL molecules (Fig. 1).26 The current indication for the use of iPCSK9 is summarized in the Table 3.27,28
Mechanism of action of iPCSK9. Figure A shows the typical pathway for LDL cholesterol metabolism in the liver. This reaches the hepatocyte, where it is internalized after binding to its LDL receptor (LDL-R). Subsequently, it is separated so that LDL is degraded in the lysosome and the R-LDL can be recycled to be re-exposed to the surface and reuptake more LDL molecules. Figure B demonstrates the action of PCSK-9. This enzyme binds non-covalently to R-LDL and LDL, forcing the degradation of both in lysosomes and preventing the recycling of R-LDL. The effect of PCSK-9 inhibitors is shown in Figure C. These are monoclonal antibodies (IgG1 / 2) that bind to PCSK-9 and prevent it from exerting its function on R-LDL and LDL. The use of iPCSK9 favors the continuous recycling of R-LDL and the uptake of LDL molecules in a more pronounced way.
Indication of iPCSK9 usage according to positioning report.
| Criterion | LDL required | IPCSK9 drug | |
|---|---|---|---|
| Patients with homozygous familial hypercholesterolemia | Age ≥12 years and one of the following: Intolerance to statins Maximum dose statins | 100mg/dl | Evolocumab (Repatha ®) |
| Patients with heterozygous familial hypercholesterolemia | One of the following: Intolerance to statins Maximum dose statins | 100mg/dl | Evolocumab (Repatha ®) Alirocumab (Praulent ®) |
| Patients with non-familial primary hypercholesterolemia or mixed dyslipidemia | Vascular disease ateroescerótica and one of the following: Intolerance to statins Maximum dose statins | 100mg/dl | Evolocumab (Repatha ®) Alirocumab (Praulent ®) |
The iPCSK9 marketed (evolocumab and alirocumab) have been able to demonstrate their ability to lower LDL cholesterol levels in two clinical trials (DESCARTES and ODYSSEY).29,30 In both, the results have been published in the population with kidney disease (eGFR> 30ml/min/1.73m2), showing an efficacy similar to that of the rest of the subjects in the studies and exceeding a 50% reduction in the LDL cholesterol numbers.31,32 The effect of iPCSKs on lipid metabolism goes beyond mere control of LDL. In the LAPLACE-2 study, the addition of evolocumab to a medium or high potency statin in patients with primary hypercholesterolemia resulted in an improvement in non-HDL cholesterol, apolipoprotein B, triglycerides, and lipoprotein (a) and even a modest increase HDL cholesterol (5–10%).33
Reduction of cardiovascular eventsRegarding its potential benefit in reducing major cardiovascular events, the FOURIER and ODYSSEY-OUTCOMES clinical trials demonstrated the effectiveness of evolocumab and alirocumab, respectively, in secondary prevention in patients with high or very high cardiovascular risk with LDL cholesterol levels due to above 70mg/dl.34,35 In both studies, patients with CKD were included, being an exclusion criterion in the FOURIER an eGFR <20ml/min/1.73m 2 and less than 30ml/min/1.73m 2 in the ODYSSEY OUTCOMES. Recently, an efficacy and safety analysis of evolocumab in patients with CKD at baseline has been published in the FOURIER study. Of the 27,564 patients included in this study, 15,034 had grade 2 CKD and 4,443 grade 3 or higher CKD. The results showed that evolocumab was at the least as effective in reducing major cardiovascular events in patients with CKD against those with preserved renal function with an absolute reduction in the highest risk with decreasing eGFR.31 A higher rate of adverse events (including changes in renal function) in patients with CKD, except in cases of not objectified rhabdomyolysis that appeared more frequently in patients with grade 3 or higher.31
At present, we do not have any specific clinical trial for the population with kidney disease that evaluates the efficacy of iPCSK9 neither in cardiovascular events nor in renal function parameters. Furthermore, no study has sub-analyzed patients with eGFR <20ml/min/1.73m 2 or those with kidney transplantation, so there are still gaps in these groups of patients.36
Very low LDL safetyThe strategy of reducing LDL cholesterol levels to the minimum possible (« the lower the better ») with the aim of minimizing the risk of cardiovascular events has raised doubts about its safety.37 From a physiological standpoint, cholesterol is necessary for the production of cell membranes and the potential effect hypolipidemic end of iPCSK9 has highlighted the need to assess the effects of extremely low figures on cell metabolism.
The most studied adverse effects in relation to low LDL levels have been myopathy, cognitive impairment, the appearance of cataracts, de novo diabetes and, above all, the incidence of hemorrhagic strokes.
Two studies (one sub - analysis of Fourier and meta - analysis based on 14 clinical trials with alirocumab) have issued specific data on the effects of iPCSK9 in patients who achieve very low levels of LDL cholesterol.38,39 In both, the safety of iPCSK9 is demonstrated even when LDL values are reached below 20mg/dl in the case of evolocumab and 15mg/dl for alirocumab. It should only be noted that there was an increase in the incidence of cataracts in patients with LDL below 25mg/dl compared to the rest of the groups, although not to a greater extent when compared with placebo.37
The most feared adverse effect derives from the epidemiological association between very low LDL values and hemorrhagic stroke.40 The pathophysiological explanation lies in the need for high amounts of cholesterol for the structure of the cerebral vascular walls. However, the data regarding this association are merely observational and none of the subanalyses published with iPCSK9 have been able to confirm them.38,39 Furthermore, when special populations that genetically have very low LDL levels have been analyzed (due to alterations in genes that increase the expression of RLDL such as PCSK9 or HMGCoA), the lack of association with hemorrhagic strokes is confirmed.41,42
Cognitive impairment was studied following an alert from the Food and Drug Administration published in 2012 on the use of statins and low LDL levels.43 The EBBINGHAUS study analyzed, using a cognitive test, Cambridge Neuropsychological Test Automated Battery, the cognitive evolution over 19 months of the patients in the FOURIER study without finding any difference between the groups that received the drug and the placebo group.44
Finally, the OSLER study with a 5-year follow-up (the longest experience with iPSCK9 published to date) has demonstrated the efficacy and safety of evolocumab without increased adverse reactions (including the development of antibodies) compared to placebo, which confirms the safety of the maintained low LDL levels and the iPCSK9 itself.45
ConclusionsThe use of iPCSK9 in patients with kidney disease is safe and effective in improving the lipid profile. Clinical trials are required in a population with kidney failure to assess the impact of its use on overall survival and on the reduction of cardiovascular events.
Conflict of interestsBQ and VAC have made paid presentations for Amgen SA®.
Please cite this article as: Quiroga B, Ramos PM, Chiva VÁ. Efectividad y seguridad del uso de inhibidores de PCSK9 en el tratamiento de la dislipidemia en el paciente con insuficiencia renal. Nefrologia. 2020;40:499–505.